Abstract
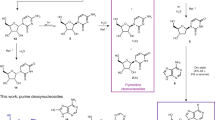
The RNA world hypothesis proposes that RNA once functioned as the principal genetic material and biological catalyst. However, RNA is a complex molecule made up of phosphate, ribose, and nucleobase moieties, and its evolution is unclear. Yakhnin has proposed a period of prebiotic chemical evolution prior to the advent of replication and Darwinian evolution, in which macromolecules containing polyols joined by phosphodiester linkages underwent spontaneous transesterification reactions with selection for stability. Although he proposes that the nucleobases were obtained during this stage from less stable macromolecules, the ultimate source of the nucleobases is not addressed. We propose that the purine nucleobases arose in situ from simpler precursors attached to a ribose-phosphate backbone, and that the weaker and less specific intra- and interstrand interactions between these precursors were the forerunners to the base pairing and base stacking interactions of the modern RNA nucleobases. Further, in line with Granick’s hypothesis of biosynthetic pathways recapitulating evolution, we propose that these simpler precursors were the same or similar to intermediates of the modern de novo purine biosynthetic pathway. We propose that successive nucleobase precursors formed progressively stronger interactions that stabilized the ribose-phosphate polymer, and that the increased stability of the parent polymer drove the selection and further chemical evolution of the purine nucleobases. Such interactions may have included hydrogen bonding between ribose hydroxyls, hydrogen bonding between carbonyl oxygens and protonated amine side groups, the intra- and interstrand coordination of metal cations, and the stacking of imidazole rings. Five of the eleven steps of the modern de novo purine biosynthetic pathway have previously been shown to have alternative nonenzymatic syntheses, while a sixth step has also been proposed to occur nonenzymatically, supporting a prebiotic origin for the pathway.




Similar content being viewed by others
Abbreviations
- R5P:
-
Ribose-5-phosphate
- R-2,4-BP:
-
Ribose-2,4-bisphosphate
- PRPP:
-
5-Phosphoribosylpyrophosphate
- PRA:
-
5-Phospho-d-ribosylamine
- GAR:
-
Glycinamide ribonucleotide
- FGAM:
-
N-Formylglycinamidine ribonucleotide
- AIR:
-
Aminoimidazole ribonucleotide
- CAIR:
-
Carboxyaminoimidazole ribonucleotide
- AICAR:
-
Aminoimidazole-4-carboxamide ribonucleotide
- FAICAR:
-
5-Formamido-4-imidazolecarboxamide ribonucleotide
- GAP:
-
Glycolaldehyde phosphate
- dICAR:
-
1-(2-Deoxy-β-d-ribofuranosyl)-imidazole-4-carboxamide
References
Athavale SS, Petrov AS, Hsiao C, Watkins D, Prickett CD, Gossett JJ, Lie L, Bowman JC, O’Neill E, Bernier CR, Hud NV, Wartell RM, Harvey SC, Williams LD (2012) RNA folding and catalysis mediated by iron (II). PLoS One 7:e38024. doi:10.1371/journal.pone.0038024
Atkins JF, Gesteland RF, Cech TR (2011) RNA worlds: from life’s origins to diversity in gene regulation. Cold Spring Harbor Laboratory Press, Plainview. ISBN 978-0-879699-46-8
Benner SA, Ellington AD, Tauer A (1989) Modern metabolism as a palimpsest of the RNA world. Proc Natl Acad Sci USA 86:7054–7058
Benner SA, Kim HJ, Carrigan MA (2012) Asphalt, water, and the prebiotic synthesis of ribose, ribonucleosides, and RNA. Acc Chem Res 45:2025–2034. doi:10.1021/ar200332w
Berg JM, Tymoczko JL, Stryer L (2012) Biochemistry, 7th edn. W H Freeman, New York
Bernhardt HS (2012) The RNA world hypothesis: the worst theory of the early evolution of life (except for all the others). Biol Direct 7:23. doi:10.1186/1745-6150-7-23
Bernhardt HS, Tate WP (2012) Primordial soup or vinaigrette: did the RNA world evolve at acidic pH? Biol Direct 7:4. doi:10.1186/1745-6150-7-4
Bevilacqua PC, Turner DH (1991) Comparison of binding of mixed ribose-deoxyribose analogues of CUCU to a ribozyme and to GGAGAA by equilibrium dialysis: evidence for ribozyme specific interactions with 2′ OH groups. Biochemistry 30:10632–10640
Bowman JC, Lenz TK, Hud NV, Williams LD (2012) Cations in charge: magnesium ions in RNA folding and catalysis. Curr Opin Struct Biol 22:262–272. doi:10.1016/j.sbi.2012.04.006
Brown T, Leonard GA, Booth ED, Kneale G (1990) Influence of pH on the conformation and stability of mismatch base-pairs in DNA. J Mol Biol 212:437–440
Butcher SE, Pyle AM (2011) The molecular interactions that stabilize RNA tertiary structure: RNA motifs, patterns, and networks. Acc Chem Res 44:1302–1311. doi:10.1021/ar200098t
Caetano-Anollés K, Caetano-Anollés G (2013) Structural phylogenomics reveals gradual evolutionary replacement of abiotic chemistries by protein enzymes in purine metabolism. PLoS One 8:e59300. doi:10.1371/journal.pone.0059300
Cheng YS, Murray M, Schendel F, Otvos J, Wehrli S, Stubbe J (1987) Chemical characterization of phosphoribosylamine, a substrate for newly discovered trifunctional protein containing glycineamide ribonucleotide synthetase activity. Adv Enzyme Regul 26:319–333
Choi S, Personick ML, Bogart JA, Ryu D, Redman RM, Laryea-Walker E (2011) Oxidation of a guanine derivative coordinated to a Pt(IV) complex initiated by intermolecular nucleophilic attacks. Dalton Trans 40:2888–2897
Corfield PW, Hunter WN, Brown T, Robinson P, Kennard O (1987) Inosine adenine base pairs in a B-DNA duplex. Nucleic Acids Res 15:7935–7949
Crick FH (1968) The origin of the genetic code. J Mol Biol 38:367–379
Engelhart AE, Hud NV (2010) Primitive genetic polymers. Cold Spring Harb Perspect Biol 2:a002196. doi:10.1101/cshperspect.a002196
Engelhart AE, Powner MW, Szostak JW (2013) Functional RNAs exhibit tolerance for non-heritable 2′–5′ versus 3′–5′ backbone heterogeneity. Nat Chem 5:390–394. doi:10.1038/nchem.1623
Eschenmoser A (1999) Chemical etiology of nucleic acid structure. Science 284:2118–2124
Fani R, Fondi M (2009) Origin and evolution of metabolic pathways. Phys Life Rev 6:23–52. doi:10.1016/j.plrev.2008.12.003
Fernandez-Forner D, Eritja R, Bardella F, Ruiz-Perez C, Solans X, Giralt E, Pedroso E (1991) Preparation of oligonucleotides containing dAICA using an unexpected side-reaction observed on a protected derivative of 2-aza-2′-deoxyinosine. Tetrahedron 47:8917–8930
Granick S (1957) Speculations on the origins and evolution of photosynthesis. Ann N Y Acad Sci 69:292–308
Griffey RH, Monia BP, Cummins LL, Freier S, Greig MJ, Guinosso CJ, Lesnik E, Manalili SM, Mohan V, Owens S, Ross BR, Sasmor H, Wancewicz E, Weiler K, Wheeler PD, Cook PD (1996) 2′-O-aminopropyl ribonucleotides: a zwitterionic modification that enhances the exonuclease resistance and biological activity of antisense oligonucleotides. J Med Chem 39:5100–5109
Halmann M, Sanchez RA, Orgel LE (1969) Phosphorylation of d-ribose in aqueous solution. J Org Chem 34:3702–3703. doi:10.1021/jo01263a132.28
Han MJ, Yoo KS, Kim YH, Chang JY (2003) Polymeric enzyme mimics: catalytic activity of ribose-containing polymers for a phosphate substrate. Org Biomol Chem 1:2276–2282
Hiller DA, Strobel SA (2011) The chemical versatility of RNA. Philos Trans R Soc Lond B Biol Sci 366:2929–2935. doi:10.1098/rstb.2011.0143
Hud NV, Jain SS, Li X, Lynn DG (2007) Addressing the problems of base pairing and strand cyclization in template-directed synthesis: a case for the utility and necessity of ‘molecular midwives’ and reversible backbone linkages for the origin of proto-RNA. Chem Biodivers 4:768–783
Hud NV, Cafferty BJ, Krishnamurthy R, Williams LD (2013) The origin of RNA and “my grandfather’s axe”. Chem Biol 20:466–474
Ingar AA, Luke RW, Hayter BR, Sutherland JD (2003) Synthesis of cytidine ribonucleotides by stepwise assembly of the heterocycle on a sugar phosphate. ChemBioChem 4:504–507
Johnson WT, Zhang P, Bergstrom DE (1997) The synthesis and stability of oligodeoxyribonucleotides containing the deoxyadenosine mimic 1-(2′-deoxy-beta-d-ribofuranosyl)imidazole-4-carboxamide. Nucleic Acids Res 25:559–567
Johnson KM, Price NE, Wang J, Fekry MI, Dutta S, Seiner DR, Wang Y, Gates KS (2013) On the formation and properties of interstrand DNA–DNA cross-links forged by reaction of an abasic site with the opposing guanine residue of 5′-CAp sequences in duplex DNA. J Am Chem Soc 135:1015–1025. doi:10.1021/ja308119q
Joshi PC, Aldersley MF, Zagorevskii DV, Ferris JP (2012) A nucleotide dimer synthesis without protecting groups using montmorillonite as catalyst. Nucleos Nucleot Nucl 31:536–566. doi:10.1080/15257770.2012.701787
Joyce GF (2002) The antiquity of RNA-based evolution. Nature 418:214–221
Keller MA, Turchyn AV, Ralser M (2014) Non-enzymatic glycolysis and pentose phosphate pathway-like reactions in a plausible Archean ocean. Mol Syst Biol 10:725. doi:10.1002/msb.20145228
Klein DJ, Moore PB, Steitz TA (2004) The roles of ribosomal proteins in the structure assembly, and evolution of the large ribosomal subunit. J Mol Biol 340:141–177
Klostermeier D, Millar DP (2002) Energetics of hydrogen bond networks in RNA: hydrogen bonds surrounding G+1 and U42 are the major determinants for the tertiary structure stability of the hairpin ribozyme. Biochemistry 41:14095–14102
Kopetzki D, Antonietti M (2011) Hydrothermal formose reaction. New J Chem 35:1787–1794
Krishnamurthy R, Pitsch S, Arrhenius G (1999a) Mineral induced formation of pentose-2,4-bisphosphates. Orig Life Evol Biosph 29:139–152
Krishnamurthy R, Arrhenius G, Eschenmoser A (1999b) Formation of glycolaldehyde phosphate from glycolaldehyde in aqueous solution. Orig Life Evol Biosph 29:333–354
Kua J, Bada JL (2011) Primordial ocean chemistry and its compatibility with the RNA world. Orig Life Evol Biosph 41:553–558. doi:10.1007/s11084-011-9250-5
Kua J, Krizner HE, De Haan DO (2011) Thermodynamics and kinetics of imidazole formation from glyoxal, methylamine, and formaldehyde: a computational study. J Phys Chem A 115:1667–1675. doi:10.1021/jp111527x
Kuruvilla E, Schuster GB, Hud NV (2013) Enhanced nonenzymatic ligation of homopurine miniduplexes: support for greater base stacking in a pre-RNA world. ChemBioChem 14:45–48. doi:10.1002/cbic.201200601
Lazcano A, Miller SL (1999) On the origin of metabolic pathways. J Mol Evol 49:424–431
Leontis NB, Stombaugh J, Westhof E (2002) The non-Watson-Crick base pairs and their associated isostericity matrices. Nucleic Acids Res 30:3497–3531
McGaughey GB, Gagné M, Rappé AK (1998) π–Stacking interactions. Alive and well in proteins. J Biol Chem 273:15458–15463
Mikkola S, Kaukinen U, Lönnberg H (2001) The effect of secondary structure on cleavage of the phosphodiester bonds of RNA. Cell Biochem Biophysics 34:95–119
Mueller EJ, Meyer E, Rudolph J, Davisson VJ, Stubbe J (1994) N 5-carboxyaminoimidazole ribonucleotide: evidence for a new intermediate and two new enzymatic activities in the de novo purine biosynthetic pathway of Escherichia coli. Biochemistry 33:2269–2278
Munanairi A, O’Banion SK, Gamble R, Breuer E, Harris AW, Sandwick RK (2007) The multiple Maillard reactions of ribose and deoxyribose sugars and sugar phosphates. Carbohydr Res 342:2575–2592
Narlikar GJ, Khosla M, Usman N, Herschlag D (1997) Quantitating tertiary binding energies of 2′ OH groups on the P1 duplex of the tetrahymena ribozyme: intrinsic binding energy in an RNA enzyme. Biochemistry 36:2465–2477
Nierlich DP, Magasanik (1965) Phosphoribosylglycinamide synthetase of Aerobacter aerogenes. Purification and properties, and nonenzymatic formation of its substrate 5′-phosphoribosylamine. J Biol Chem 240:366–374
Orgel LE (2004) Prebiotic chemistry and the origin of the RNA world. Crit Rev Biochem Mol Biol 39:99–123
Ourisson G, Nakatani Y (1994) The terpenoid theory of the origin of cellular life: the evolution of terpenoids to cholesterol. Chem Biol 1:11–23
Pierce J, Serianni AS, Barker R (1985) Anomerization of furanose sugars and sugar phosphates. J Am Chem Soc 107:2448–2456. doi:10.1021/ja00294a041
Pochet S, D’Ari R (1990) Synthesis and enzymatic polymerisation of 5-amino-1-(2′-deoxy-beta-D-ribofuranosyl)imidazole-4-carboxamide-5′-triphosphate. Nucleic Acids Res 18:7127–7131
Powner MW, Gerland B, Sutherland JD (2009) Synthesis of activated pyrimidine ribonucleotides in prebiotically plausible conditions. Nature 459:239–242. doi:10.1038/nature08013
Prakash TP, Roberts C, Switzer C (1997) Activity of 2′,5′-linked RNA in the template-directed oligomerization of mononucleotides. Angew Chem Int Ed Engl 36:1522–1523
Price NE, Johnson KM, Wang J, Fekry MI, Wang Y, Gates KS (2014) Interstrand DNA–DNA cross-link formation between adenine residues and abasic sites in duplex DNA. J Am Chem Soc 136:3483–3490. doi:10.1021/ja410969x
Sacerdote MG, Szostak JW (2005) Semipermeable lipid bilayers exhibit diastereoselectivity favoring ribose. Proc Natl Acad Sci USA 102:6004–6008
Safaee N, Noronha AM, Rodionov D, Kozlov G, Wilds CJ, Sheldrick GM, Gehring K (2013) Structure of the parallel duplex of poly(A) RNA: evaluation of a 50 year-old prediction. Angew Chem Int Ed Engl 52:10370–10373. doi:10.1002/anie.201303461
Sagi VN, Punna V, Hu F, Meher G, Krishnamurthy R (2012) Exploratory experiments on the chemistry of the “glyoxylate scenario”: formation of ketosugars from dihydroxyfumarate. J Am Chem Soc 134:3577–3589. doi:10.1021/ja211383c
Sala M, Pezo V, Pochet S, Wain-Hobson S (1996) Ambiguous base pairing of the purine analogue 1-(2-deoxy-beta-d-ribofuranosyl)-imidazole-4-carboxamide during PCR. Nucleic Acids Res 24:3302–3306
Sanchez RA, Orgel LE (1970) Studies in prebiotic synthesis. V. Synthesis and photoanomerization of pyrimidine nucleosides. J Mol Biol 47:531–543
Sandwick R, Johanson M, Breuer E (2005) Maillard reactions of ribose 5-phosphate and amino acids. Ann N Y Acad Sci 1043:85–96
Schendel FJ, Cheng YS, Otvos JD, Wehrli S, Stubbe J (1988) Characterization and chemical properties of phosphoribosylamine, an unstable intermediate in the de novo purine biosynthetic pathway. Biochemistry 27:2614–2623
Sharma P, Mitra A, Sharma S, Singh H, Bhattacharyya D (2008) Quantum chemical studies of structures and binding in noncanonical RNA base pairs: the trans Watson–Crick:Watson–Crick family. J Biomol Struct Dyn 25:709–732
Shilovsky GA, Khokhlov AN, Shram SI (2013) The protein poly(ADP-ribosyl)ation system: its role in genome stability and lifespan determination. Biochemistry (Mosc) 78:433–444. doi:10.1134/S0006297913050015
Silhavy TJ, Kahne D, Walker S (2010) The bacterial cell envelope. Cold Spring Harb Perspect Biol 2:a000414. doi:10.1101/cshperspect.a000414
Silverman SK, Cech TR (1999) Energetics and cooperativity of tertiary hydrogen bonds in RNA structure. Biochemistry 38:8691–8702
Soukup GA, Breaker RR (1999) Relationship between internucleotide linkage geometry and the stability of RNA. RNA 5:1308–1325
Su AA, Randau L (2011) A-to-I and C-to-U editing within transfer RNAs. Biochemistry (Mosc) 76:932–937. doi:10.1134/S0006297911080098
Tamura M, Holbrook SR (2002) Sequence and structural conservation in RNA ribose zippers. J Mol Biol 320:455–474
Tanaka K, Tengeiji A, Kato T, Toyama N, Shionoya M (2003) A discrete self-assembled metal array in artificial DNA. Science 299:1212–1213
Tinoco I Jr, Bustamante C (1999) How RNA folds. J Mol Biol 293:271–281
Trevino SG, Zhang N, Elenko MP, Lupták A, Szostak JW (2011) Evolution of functional nucleic acids in the presence of nonheritable backbone heterogeneity. Proc Natl Acad Sci USA 108:13492–13497. doi:10.1073/pnas.1107113108
Uesugi S, Oda Y, Ikehara M, Kawase Y, Ohtsuka E (1987) Identification of I:A mismatch base-pairing structure in DNA. J Biol Chem 262:6965–6968
Ulyanov NB, James TL (2010) RNA structural motifs that entail hydrogen bonds involving sugar-phosphate backbone atoms of RNA. New J Chem 34:910–917
Vail SL, Pierce AG (1972) Limitations for the addition of amides to formaldehyde and glyoxal. J Org Chem 37:391–393. doi:10.1021/jo00968a013
Yakhnin AV (2013) A model for the origin of life through rearrangements among prebiotic phosphodiester polymers. Orig Life Evol Biosph 43:39–47. doi:10.1007/s11084-012-9321-2
Yakovchuk P, Protozanova E, Frank-Kamenetskii MD (2006) Base-stacking and base-pairing contributions into thermal stability of the DNA double helix. Nucleic Acids Res 34:564–574
Zhang Y, Morar M, Ealick SE (2008) Structural biology of the purine biosynthetic pathway. Cell Mol Life Sci 65:3699–3724. doi:10.1007/s00018-008-8295-8
Zhang S, Wu Y, Zhang W (2014) G-quadruplex structures and their interaction diversity with ligands. ChemMedChem 9:899–911. doi:10.1002/cmdc.201300566
Zubay G (1998) Studies on the lead-catalyzed synthesis of aldopentoses. Orig Life Evol Biosph 28:13–26
Zubay G (2000a) Biochemical pathways may provide leads to prebiotic pathways. Chemtracts Biochem Mol Biol 13:357–363
Zubay G (2000b) Synthesis of the first nucleotides. Chemtracts: Org Chem 13:883–903
Zubay G, Mui T (2001) Prebiotic synthesis of nucleotides. Orig Life Evol Biosph 31:87–102
Acknowledgments
Bill Hawkins, Joel Tyndall, Alexander Yakhnin, Steve Benner, Ramanarayanan Krishnamurthy, Joe Kappock, Nicholas Hud and Jim Cleaves for helpful discussions, to Geremy Ralston for providing useful information, and to the anonymous reviewers for helpful suggestions that have greatly improved this manuscript. This work was supported by a Fast Start Grant from the Marsden Fund, New Zealand, and the Department of Biochemistry, University of Otago (HSB).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bernhardt, H.S., Sandwick, R.K. Purine Biosynthetic Intermediate-Containing Ribose-Phosphate Polymers as Evolutionary Precursors to RNA. J Mol Evol 79, 91–104 (2014). https://doi.org/10.1007/s00239-014-9640-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00239-014-9640-1