Abstract
Purpose
Burning mouth syndrome (BMS) is a chronic intraoral pain syndrome featuring idiopathic oral pain and burning discomfort despite clinically normal oral mucosa. The etiology of chronic pain syndrome is unclear, but preliminary neuroimaging research has suggested the alteration of volume, metabolism, blood flow, and diffusion at multiple brain regions. According to the neuromatrix theory of Melzack, pain sense is generated in the brain by the network of multiple pain-related brain regions. Therefore, the alteration of pain-related network is also assumed as an etiology of chronic pain. In this study, we investigated the brain network of BMS brain by using probabilistic tractography and graph analysis.
Methods
Fourteen BMS patients and 14 age-matched healthy controls underwent 1.5T MRI. Structural connectivity was calculated in 83 anatomically defined regions with probabilistic tractography of 60-axis diffusion tensor imaging and 3D T1-weighted imaging. Graph theory network analysis was used to evaluate the brain network at local and global connectivity.
Results
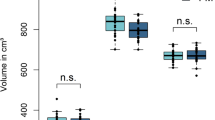
In BMS brain, a significant difference of local brain connectivity was recognized at the bilateral rostral anterior cingulate cortex, right medial orbitofrontal cortex, and left pars orbitalis which belong to the medial pain system; however, no significant difference was recognized at the lateral system including the somatic sensory cortex. A strengthened connection of the anterior cingulate cortex and medial prefrontal cortex with the basal ganglia, thalamus, and brain stem was revealed.
Conclusion
Structural brain network analysis revealed the alteration of the medial system of the pain-related brain network in chronic pain syndrome.




Similar content being viewed by others
Abbreviations
- BMS:
-
Burning mouth syndrome
- ACC:
-
Anterior cingulate cortex
- PFC:
-
Prefrontal cortex
References
Headache Classification Committee of the International Headache Society (IHS) (2013) The International Classification of Headache Disorders, 3rd edition (beta version). Cephalalgia 33:629–808. doi:10.1177/0333102413485658
Gao J, Chen L, Zhou J, Peng J (2008) A case-control study on etiological factors involved in patients with burning mouth syndrome. J Oral Pathol Med 38:24–28. doi:10.1111/j.1600-0714.2008.00708.x
Gorsky M, Silverman S, Chinn H (1991) Clinical characteristics and management outcome in the burning mouth syndrome. An open study of 130 patients. Oral Surg Oral Med Oral Pathol 72:192–195
Gurvits GE, Tan A (2013) Burning mouth syndrome. World J Gastroenterol 19:665–672. doi:10.3748/wjg.v19.i5.665
Kohorst JJ, Bruce AJ, Torgerson RR et al (2014) A population-based study of the incidence of burning mouth syndrome. Mayo Clin Proc 89:1545–1552. doi:10.1016/j.mayocp.2014.05.018
Renton T (2011) Burning mouth syndrome. Rev Pain 5:12–17. doi:10.1177/204946371100500403
Adamo D, Celentano A, Ruoppo E et al (2015) The relationship between sociodemographic characteristics and clinical features in burning mouth syndrome. Pain Med 16:2171–2179. doi:10.1111/pme.12808
Forssell H, Jääskeläinen S, Tenovuo O, Hinkka S (2002) Sensory dysfunction in burning mouth syndrome. Pain 99:41–47. doi:10.1016/S0304-3959(02)00052-0
Grushka M, Epstein JB, Gorsky M (2003) Burning mouth syndrome and other oral sensory disorders: a unifying hypothesis. Pain Res Manag 8:133–135
Albuquerque RJC, de Leeuw R, Carlson CR et al (2006) Cerebral activation during thermal stimulation of patients who have burning mouth disorder: an fMRI study. Pain 122:223–234. doi:10.1016/j.pain.2006.01.020
Forssell H, Jääskeläinen S, List T et al (2015) An update on pathophysiological mechanisms related to idiopathic oro-facial pain conditions with implications for management. J Oral Rehabil 42:300–322. doi:10.1111/joor.12256
Lauria G, Majorana A, Borgna M et al (2005) Trigeminal small-fiber sensory neuropathy causes burning mouth syndrome. Pain 115:332–337. doi:10.1016/j.pain.2005.03.028
Yilmaz Z, Renton T, Yiangou Y et al (2007) Burning mouth syndrome as a trigeminal small fibre neuropathy: increased heat and capsaicin receptor TRPV1 in nerve fibres correlates with pain score. J Clin Neurosci 14:864–871. doi:10.1016/j.jocn.2006.09.002
Eliav E, Kamran B, Schaham R et al (2007) Evidence of chorda tympani dysfunction in patients with burning mouth syndrome. J Am Dent Assoc 138:628–633
Nasri-Heir C, Gomes J, Heir GM et al (2011) The role of sensory input of the chorda tympani nerve and the number of fungiform papillae in burning mouth syndrome. Oral Surg Oral Med Oral Pathol Oral Radiol Endodontol 112:65–72. doi:10.1016/j.tripleo.2011.02.035
Svensson P, Bjerring P, Arendt-Nielsen L, Kaaber S (1993) Sensory and pain thresholds to orofacial argon laser stimulation in patients with chronic burning mouth syndrome. Clin J Pain 9:207–215
Sinding C, Gransjøen AM, Schlumberger G et al (2016) Grey matter changes of the pain matrix in patients with burning mouth syndrome. Eur J Neurosci 43:997–1005. doi:10.1111/ejn.13156
Shinozaki T, Imamura Y, Kohashi R et al (2016) Spatial and temporal brain responses to noxious heat thermal stimuli in burning mouth syndrome. J Dent Res 95:1138–1146. doi:10.1177/0022034516653580
Khan SA, Keaser ML, Meiller TF, Seminowicz DA (2014) Altered structure and function in the hippocampus and medial prefrontal cortex in patients with burning mouth syndrome. Pain 155:1472–1480. doi:10.1016/j.pain.2014.04.022
Kupers R, Kehlet H (2006) Brain imaging of clinical pain states: a critical review and strategies for future studies. Lancet Neurol 5:1033–1044. doi:10.1016/S1474-4422(06)70624-X
Seifert F, Maihöfner C (2009) Central mechanisms of experimental and chronic neuropathic pain: findings from functional imaging studies. Cell Mol Life Sci 66:375–390. doi:10.1007/s00018-008-8428-0
Price DD (2000) Psychological and neural mechanisms of the affective dimension of pain. Science 288:1769–1772. doi:10.1126/science.288.5472.1769
Apkarian AV, Bushnell MC, Treede R-D, Zubieta J-K (2005) Human brain mechanisms of pain perception and regulation in health and disease. Eur J Pain 9:463–484. doi:10.1016/j.ejpain.2004.11.001
Melzack R (2001) Pain and the neuromatrix in the brain. J Dent Educ 65:1378–1382
Van Dijk KRA, Hedden T, Venkataraman A et al (2010) Intrinsic functional connectivity as a tool for human connectomics: theory, properties, and optimization. J Neurophysiol 103:297–321. doi:10.1152/jn.00783.2009
Whitfield-Gabrieli S, Nieto-Castanon A (2012) Conn: a functional connectivity toolbox for correlated and anticorrelated brain networks. Brain Connectivity 2:125–141. doi:10.1089/brain.2012.0073
Behrens TEJ, Woolrich MW, Jenkinson M et al (2003) Characterization and propagation of uncertainty in diffusion-weighted MR imaging. Magn Reson Med 50:1077–1088. doi:10.1002/mrm.10609
Nucifora PGP, Verma R, Lee S-K, Melhem ER (2007) Diffusion-tensor MR imaging and tractography: exploring brain microstructure and connectivity. Radiology 245:367–384. doi:10.1148/radiol.2452060445
Behrens TEJ, Berg HJ, Jbabdi S et al (2007) Probabilistic diffusion tractography with multiple fibre orientations: what can we gain? NeuroImage 34:144–155. doi:10.1016/j.neuroimage.2006.09.018
Cao Q, Shu N, An L et al (2013) Probabilistic diffusion tractography and graph theory analysis reveal abnormal white matter structural connectivity networks in drug-naive boys with attention deficit/hyperactivity disorder. J Neurosci 33:10676–10687. doi:10.1523/JNEUROSCI.4793-12.2013
Koike K, Shinozaki T, Hara K et al (2014) Immune and endocrine function in patients with burning mouth syndrome. Clin J Pain 30:168–173. doi:10.1097/AJP.0b013e31828c4bf1
Yamada H, Abe O, Shizukuishi T et al (2014) Efficacy of distortion correction on diffusion imaging: comparison of FSL eddy and eddy_correct using 30 and 60 directions diffusion encoding. PLoS One. doi:10.1371/journal.pone.0112411.s009
Daducci A, Gerhard S, Griffa A et al (2012) The connectome mapper: an open-source processing pipeline to map connectomes with MRI. PLoS One 7:e48121. doi:10.1371/journal.pone.0048121
Desikan RS, Ségonne F, Fischl B et al (2006) An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. NeuroImage 31:968–980. doi:10.1016/j.neuroimage.2006.01.021
Rubinov M, Sporns O (2010) Complex network measures of brain connectivity: uses and interpretations. NeuroImage 52:1059–1069. doi:10.1016/j.neuroimage.2009.10.003
Rubinov M, Sporns O (2011) Weight-conserving characterization of complex functional brain networks. NeuroImage 56:2068–2079. doi:10.1016/j.neuroimage.2011.03.069
Boccaletti S, Latora V, Moreno Y, Chavez M (2006) Complex networks: structure and dynamics. Phys Rep 424:175–308. doi:10.1016/j.physrep.2005.10.009
Albert R, Barabási A-L (2002) Statistical mechanics of complex networks. Rev Mod Phys 74:47–97. doi:10.1103/RevModPhys.74.47
Borgatti SP, Mehra A, Brass DJ, Labianca G (2009) Network analysis in the social sciences. Science 323:892–895. doi:10.1126/science.1165821
Newman M (2003) The structure and function of complex networks. SIAM Rev 45:167–256
Bullmore E, Sporns O (2009) Complex brain networks: graph theoretical analysis of structural and functional systems. Nat Rev Neurosci 10:186–198. doi:10.1038/nrn2575
Achard S (2006) A resilient, low-frequency, small-world human brain functional network with highly connected association cortical hubs. J Neurosci 26:63–72. doi:10.1523/JNEUROSCI.3874-05.2006
Watts DJ, Strogatz SH (1998) Collective dynamics of small-world networks. Nature 393:440–442. doi:10.1038/30918
Woolf CJ (2010) What is this thing called pain? J Clin Invest 120:3742. doi:10.1172/JCI45178
Chen J (2011) History of pain theories. Neurosci Bull 27:343–350. doi:10.1007/s12264-011-0139-0
May A (2007) Neuroimaging: visualising the brain in pain. Neurol Sci 28:S101–S107. doi:10.1007/s10072-007-0760-x
Moisset X, Bouhassira D (2007) Brain imaging of neuropathic pain. NeuroImage 37(Suppl 1):S80–S88. doi:10.1016/j.neuroimage.2007.03.054
Luo C, Kuner T, Kuner R (2014) Synaptic plasticity in pathological pain. Trends Neurosci 37:343–355. doi:10.1016/j.tins.2014.04.002
Borsook D, Moulton EA, Schmidt KF, Becerra LR (2007) Neuroimaging revolutionizes therapeutic approaches to chronic pain. Mol Pain 3:1. doi:10.1186/1744-8069-3-25
Rainville P (1997) Pain affect encoded in human anterior cingulate but not somatosensory cortex. Science 277:968–971. doi:10.1126/science.277.5328.968
Peyron R, Laurent B, García-Larrea L (2000) Functional imaging of brain responses to pain. A review and meta-analysis (2000). Neurophysiol Clin 30:263–288
Ghatan PH, Hsieh JC, Wirsen-Meurling A et al (1995) Brain activation induced by the perceptual maze test: a PET study of cognitive performance. NeuroImage 2:112–124. doi:10.1006/nimg.1995.1014
Wager TD (2004) Placebo-induced changes in fMRI in the anticipation and experience of pain. Science 303:1162–1167. doi:10.1126/science.1093065
Amodio DM, Frith CD (2006) Meeting of minds: the medial frontal cortex and social cognition. Nat Rev Neurosci 7:268–277. doi:10.1038/nrn1884
May A (2008) Chronic pain may change the structure of the brain. Pain 137:7–15. doi:10.1016/j.pain.2008.02.034
Baliki MN, Geha PY, Jabakhanji R et al (2008) A preliminary fMRI study of analgesic treatment in chronic back pain and knee osteoarthritis. Mol Pain 4:47. doi:10.1186/1744-8069-4-47
Apkarian AV, Sosa Y, Sonty S et al (2004) Chronic back pain is associated with decreased prefrontal and thalamic gray matter density. J Neurosci 24:10410–10415. doi:10.1523/JNEUROSCI.2541-04.2004
Grushka M (1987) Clinical features of burning mouth syndrome. Oral Surg Oral Med Oral Pathol 63:30–36. doi:10.1016/0030-4220(87)90336-7
Kong J, Jensen K, Loiotile R et al (2013) Functional connectivity of the frontoparietal network predicts cognitive modulation of pain. Pain 154:459–467. doi:10.1016/j.pain.2012.12.004
Bushnell MC, Čeko M, Low LA (2013) Cognitive and emotional control of pain and its disruption in chronic pain. Nat Rev Neurosci 14:502–511. doi:10.1038/nrn3516
Catani M, Thiebaut de Schotten M, Slater D, Dell’Acqua F (2013) Connectomic approaches before the connectome. NeuroImage 80:2–13. doi:10.1016/j.neuroimage.2013.05.109
Talbot JD, Marrett S, Evans AC et al (1991) Multiple representations of pain in human cerebral cortex. Science 251:1355–1358
Apkarian VA (1995) Functional imaging of pain: new insights regarding the role of the cerebral cortex in human pain perception. Semin Neurosci 7:279–293. doi:10.1006/smns.1995.0031
Hsieh J-C, Ståhle-Bäckdahl M, Hägermark Ö et al (1996) Traumatic nociceptive pain activates the hypothalamus and the periaqueductal gray: a positron emission tomography study. Pain 64:303–314. doi:10.1016/0304-3959(95)00129-8
Forss N, Raij TT, Seppä M, Hari R (2005) Common cortical network for first and second pain. NeuroImage 24:132–142. doi:10.1016/j.neuroimage.2004.09.032
Mullins P, Rowland L, Jung R, Sibbittjr W (2005) A novel technique to study the brain’s response to pain: proton magnetic resonance spectroscopy. NeuroImage 26:642–646. doi:10.1016/j.neuroimage.2005.02.001
Petrovic P, Ingvar M (2002) Imaging cognitive modulation of pain processing. Pain 95:1–5
Gracely RH, Geisser ME, Giesecke T et al (2004) Pain catastrophizing and neural responses to pain among persons with fibromyalgia. Brain 127:835–843. doi:10.1093/brain/awh098
Schweinhardt P, Bushnell MC (2010) Pain imaging in health and disease—how far have we come? J Clin Invest 120:3788. doi:10.1172/JCI43498
Fields H (2004) State-dependent opioid control of pain. Nat Rev Neurosci 5:565–575. doi:10.1038/nrn1431
Tracey I (2005) Nociceptive processing in the human brain. Curr Opin Neurobiol 15:478–487. doi:10.1016/j.conb.2005.06.010
Hadjipavlou G, Dunckley P, Behrens TE, Tracey I (2006) Determining anatomical connectivities between cortical and brainstem pain processing regions in humans: a diffusion tensor imaging study in healthy controls. Pain 123:169–178. doi:10.1016/j.pain.2006.02.027
Tracey I, Mantyh PW (2007) The cerebral signature for pain perception and its modulation. Neuron 55:377–391. doi:10.1016/j.neuron.2007.07.012
Koch MA, Norris DG, Hund-Georgiadis M (2002) An investigation of functional and anatomical connectivity using magnetic resonance imaging. NeuroImage 16:241–250. doi:10.1006/nimg.2001.1052
Camchong J, MacDonald AW, Bell C et al (2011) Altered functional and anatomical connectivity in schizophrenia. Schizophr Bull 37:640–650. doi:10.1093/schbul/sbp131
Damoiseaux JS, Greicius MD (2009) Greater than the sum of its parts: a review of studies combining structural connectivity and resting-state functional connectivity. Brain Struct Funct 213:525–533. doi:10.1007/s00429-009-0208-6
Messé A, Rudrauf D, Benali H, Marrelec G (2014) Relating structure and function in the human brain: relative contributions of anatomy, stationary dynamics, and non-stationarities. PLoS Comp Biol 10:e1003530. doi:10.1371/journal.pcbi.1003530
Greicius MD, Supekar K, Menon V, Dougherty RF (2008) Resting-state functional connectivity reflects structural connectivity in the default mode network. Cereb Cortex 19:72–78. doi:10.1093/cercor/bhn059
Xiao H, Yang Y, Xi J-H, Chen Z-Q (2015) Structural and functional connectivity in traumatic brain injury. Neural Regen Res 10:2062–2071. doi:10.4103/1673-5374.172328
Ray S, Miller M, Karalunas S et al (2014) Structural and functional connectivity of the human brain in autism spectrum disorders and attention-deficit/hyperactivity disorder: a rich club-organization study. Hum Brain Mapp 35:6032–6048. doi:10.1002/hbm.22603
Betzel RF, Byrge L, He Y et al (2014) Changes in structural and functional connectivity among resting-state networks across the human lifespan. NeuroImage 102(Pt 2):345–357. doi:10.1016/j.neuroimage.2014.07.067
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded by a Japan Society for the Promotion of Science Grant-in-Aid for Scientific Research to OA (grant number 16K10330), Grants-in-Aid for Scientific Research to YI (grant number 15K11326), a Uemura Fund Research Grant to YI and Grants-in-Aid for Scientific Research to TS (grant number 16K11897).
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Nihon University Hospitals’ Joint Institutional Review Board and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Wada, A., Shizukuishi, T., Kikuta, J. et al. Altered structural connectivity of pain-related brain network in burning mouth syndrome—investigation by graph analysis of probabilistic tractography. Neuroradiology 59, 525–532 (2017). https://doi.org/10.1007/s00234-017-1830-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-017-1830-2