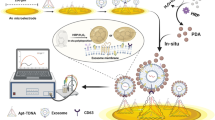
Abstract
Exosomes have attracted significant attention as cancer diagnostic targets and therapeutic agents due to their unique biogenesis and structure. To clarify the biological activities of exosomes, it is important to obtain a picture of their intracellular distribution and how they evolve over time. In this work, a new kind of intracellular exosome imaging and concurrent pH sensing method is demonstrated by using the surface-enhanced Raman scattering (SERS) technique. Specifically, 4-mercaptobenzoic acid (4MBA)–tagged silver nanoparticles are attached onto the outer surfaces of exosomes, in which silver nanoparticles are employed as SERS generators. Raman agents 4MBA are susceptible to a specific intracellular stimulus, that is, undergo a protonation or deprotonation in response to intracellular pH variation, which correspondingly exhibit different vibrational spectra features. By using the SERS spectroscopy, tracking of the intracellular distribution of exosomes and the concurrent quantitative sensing of environmental pH were achieved, which demonstrated that, as time prolonged, exosomes first attached with the tumor cell surfaces, and then entered into the cells and accumulated in lysosomes. Such SERS-active hybridized exosomes, that are sensitive to discrete variations in intracellular pH, have proved their capability for the investigation of interactions between exosomes and cells. The spectral diversity and flexible surface modification of these hybridized exosomes are also highly expected in developing multifunctional exosome-based nanoplatforms, which offers great potential to promote the exosome-based therapeutics forward into an advanced stage.
Graphical abstract








Similar content being viewed by others
References
El Andaloussi S, Maeger I, Breakefield XO, Wood MJA. Extracellular vesicles: biology and emerging therapeutic opportunities. Nat Rev Drug Discov. 2013;12:348–58.
Raposo G, Stoorvogel W. Extracellular vesicles: exosomes, microvesicles, and friends. J Cell Biol. 2013;200:373–83.
Azmi AS, Bao B, Sarkar FH. Exosomes in cancer development, metastasis, and drug resistance: a comprehensive review. Cancer Metastasis Rev. 2013;32:623–42.
Alvarez-Erviti L, Seow Y, Yin H, Betts C, Lakhal S, Wood MJA. Delivery of siRNA to the mouse brain by systemic injection of targeted exosomes. Nat Biotechnol. 2011;29:341–U179.
Ferguson SW, Nguyen J. Exosomes as therapeutics: the implications of molecular composition and exosomal heterogeneity. J Control Release. 2016;228:179–90.
Huang T, Deng C-X. Current progresses of exosomes as cancer diagnostic and prognostic biomarkers. Int J Biol Sci. 2019;15:1–11.
Koliha N, Heider U, Ozimkowski T, Wiemann M, Bosio A, Wild S. Melanoma affects the composition of blood cell-derived extracellular vesicles. Front Immunol. 2016;7:282.
Li T, Long S, Gu M, Guo J, Liu Y, Zhang W, et al. Serum exosomal microRNAs as potent circulating biomarkers for melanoma. Melanoma Res. 2018;28:295–303.
Savvateeva EN, Tikhonov AA, Butvilovskaya VI, Tsybulskaya MV, Rubina AY. Exosomal surface protein markers in diagnosis of colorectal cancer. Mol Biol. 2017;51:659–65.
Xiao Y, Zhong J, Zhong B, Huang J, Jiang L, Jiang Y, et al. Exosomes as potential sources of biomarkers in colorectal cancer. Cancer Lett. 2020;476:13–22.
Meng Y, Sun J, Wang X, Hu T, Ma Y, Kong C, et al. Exosomes: a promising avenue for the diagnosis of breast cancer. Technol Cancer Res Treat. 2019;18:1533033818821421.
Sohn Y-S, Jang D-h, Na W. Immunoaffinity characteristics of exosomes from breast cancer cells using surface plasmon resonance spectroscopy. J Sens Sci Technol. 2019;28:355–9.
Tang Y, Huang Y, Li J, Qin S, Xu Y, An T, et al. Alterations in exosomal miRNA profile upon epithelial-mesenchymal transition in human lung cancer cell lines. BMC Genomics. 2018;19:802.
Wang N, Song X, Liu L, Niu L, Wang X, Song X, et al. Circulating exosomes contain protein biomarkers of metastatic non-small-cell lung cancer. Cancer Sci. 2018;109:1701–9.
Hoshino A, Costa-Silva B, Shen T-L, Rodrigues G, Hashimoto A, Mark MT, et al. Tumour exosome integrins determine organotropic metastasis. Nature. 2015;527:329–35.
Maji S, Chaudhary P, Akopova I, Nguyen PM, Hare RJ, Gryczynski I, et al. Exosomal annexin II promotes angiogenesis and breast cancer metastasis. Mol Cancer Res. 2017;15:93–105.
Fernandez A, Vendrell M. Smart fluorescent probes for imaging macrophage activity. Chem Soc Rev. 2016;45:1182–96.
Gao W, Yuan J, Liu H, Wu C, Huang D, Zhu J, et al. Dendritic cells derived exosomes migration to spleen and induction of inflammation are regulated by CCR7. Sci Rep. 2017;7:42996–6.
He F, Ye Z, Zhao L, Yin B, Ye B. Probing exosome internalization pathways through confocal microscopy imaging. Chem Commun. 2019;55:14015–8.
Suetsugu A, Honma K, Saji S, Moriwaki H, Ochiya T, Hoffman RM. Imaging exosome transfer from breast cancer cells to stroma at metastatic sites in orthotopic nude-mouse models. Adv Drug Deliv Rev. 2013;65:383–90.
Fan Z, Xiao K, Lin J, Liao Y, Huang X. Functionalized DNA enables programming exosomes/vesicles for tumor imaging and therapy. Small. 2019;15:e1903761.
Vegh RB, Bravaya KB, Bloch DA, Bommarius AS, Tolbert LM, Verkhovsky M, et al. Chromophore photoreduction in red fluorescent proteins is responsible for bleaching and phototoxicity. J Phys Chem B. 2014;118:4527–34.
Liu X, Braun GB, Zhong H, Hall DJ, Han W, Qin M, et al. Tumor-targeted multimodal optical imaging with versatile cadmium-free quantum dots. Adv Funct Mater. 2016;26:267–76.
Wang Z, Zong S, Li W, Wang C, Xu S, Chen H, et al. SERS-fluorescence joint spectral encoding using organic-metal-QD hybrid nanoparticles with a huge encoding capacity for high-throughput biodetection: putting theory into practice. J Am Chem Soc. 2012;134:2993–3000.
Wang Z, Zong S, Chen H, Wang C, Xu S, Cui Y. SERS-fluorescence joint spectral encoded magnetic nanoprobes for multiplex cancer cell separation. Adv Healthc Mater. 2014;3:1889–97.
Wang Y, Yan B, Chen L. SERS tags: novel optical nanoprobes for bioanalysis. Chem Rev. 2013;113:1391–428.
Doering WE, Nie SM. Spectroscopic tags using dye-embedded nanoparticles and surface-enhanced Raman scattering. Anal Chem. 2003;75:6171–6.
Hada A-M, Potara M, Suarasan S, Vulpoi A, Nagy-Simon T, Licarete E, et al. Fabrication of gold-silver core-shell nanoparticles for performing as ultrabright SERS-nanotags inside human ovarian cancer cells. Nanotechnology. 2019;30:315701.
Dharmalingam P, Venkatakrishnan K, Tan B. Probing cancer metastasis at a single-cell level with a Raman-functionalized anionic probe. Nano Lett. 2020;20:1054–66.
Kuku G, Saricam M, Akhatova F, Danilushkina A, Fakhrullin R, Culha M. Surface-enhanced Raman scattering to evaluate nanomaterial cytotoxicity on living cells. Anal Chem. 2016;88:9813–20.
Sheena TS, Devaraj V, Lee J-M, Balaji P, Gnanasekar P, Oh J-W, et al. Sensitive and label-free shell isolated Ag NPs@Si architecture based SERS active substrate: FDTD analysis and in-situ cellular DNA detection. Appl Surf Sci. 2020;515:145955.
Chen H, Wang Z, Zong S, Wu L, Chen P, Zhu D, et al. SERS-fluorescence monitored drug release of a redox-responsive nanocarrier based on graphene oxide in tumor cells. ACS Appl Mater Interfaces. 2014;6:17526–33.
Zong S, Wang Z, Chen H, Cui Y. Assessing telomere length using surface enhanced Raman scattering. Sci Rep. 2014;4:6977.
Chen H, Wang Z, Ma X, Zong S, Cui Y. Magnetically controllable dual-mode nanoprobes for cell imaging with an onion-liked structure. Talanta. 2013;116:978–84.
Zong S, Wang Z, Chen H, Yang J, Cui Y. Surface enhanced Raman scattering traceable and glutathione responsive nanocarrier for the intracellular drug delivery. Anal Chem. 2013;85:2223–30.
Puppulin L, Hosogi S, Sun H, Matsuo K, Inui T, Kumamoto Y, et al. Bioconjugation strategy for cell surface labelling with gold nanostructures designed for highly localized pH measurement. Nat Commun. 2018;9:5278.
Lee PC, Meisel D. Adsorption and surface-enhanced Raman of dyes on silver and gold sols. J Phys Chem. 1982;86:3391–5.
Konnova SA, Danilushkina AA, Fakhrullina GI, Akhatova FS, Badrutdinov AR, Fakhrullin RF. Silver nanoparticle-coated “cyborg” microorganisms: rapid assembly of polymer-stabilised nanoparticles on microbial cells. RSC Adv. 2015;5:13530–7.
Wang Z, Zong S, Chen H, Wu H, Cui Y. Silica coated gold nanoaggregates prepared by reverse microemulsion method: dual mode probes for multiplex immunoassay using SERS and fluorescence. Talanta. 2011;86:170–7.
Zhu D, Wang Z, Zong S, Chen H, Wu X, Pei Y, et al. Ag@4ATP-coated liposomes: SERS traceable delivery vehicles for living cells. Nanoscale. 2014;6:8155–61.
Chen P, Wang Z, Zong S, Zhu D, Chen H, Zhang Y, et al. pH-sensitive nanocarrier based on gold/silver core-shell nanoparticles decorated multi-walled carbon manotubes for tracing drug release in living cells. Biosens Bioelectron. 2016;75:446–51.
Capocefalo A, Mammucari D, Brasili F, Fasolato C, Bordi F, Postorino P, et al. Exploring the potentiality of a SERS-active pH nano-biosensor. Front Chem. 2019;7.
Zhang Z, Bando K, Mochizuki K, Taguchi A, Fujita K, Kawata S. Quantitative evaluation of surface-enhanced Raman scattering nanoparticles for intracellular pH sensing at a single particle level. Anal Chem. 2019;91:3254–62.
Xu M, Ma X, Wei T, Lu Z-X, Ren B. In situ imaging of live-cell extracellular pH during cell apoptosis with surface-enhanced Raman spectroscopy. Anal Chem. 2018;90:13922–8.
Shen Y, Liang L, Zhang S, Huang D, Zhang J, Xu S, et al. Organelle-targeting surface-enhanced Raman scattering (SERS) nanosensors for subcellular pH sensing. Nanoscale. 2018;10:1622–30.
Pallaoro A, Braun GB, Reich NO, Moskovits M. Mapping local pH in live cells using encapsulated fluorescent SERS nanotags. Small. 2010;6:618–22.
Talley CE, Jusinski L, Hollars CW, Lane SM, Huser T. Intracellular pH sensors based on surface-enhanced Raman scattering. Anal Chem. 2004;76:7064–8.
Gong Y-J, Zhang M-L, Wang B-X, Lv Q, Wang Y, Dong W. A smart approach toward rhodamine spiro-ring derivatives sensing platform for lysosome-targetable imaging applications. Sens Actuators B Chem. 2019;283:239–46.
Kneipp J, Kneipp H, Wittig B, Kneipp K. Following the dynamics of pH in endosomes of live cells with SERS nanosensors. J Phys Chem C. 2010;114:7421–6.
Zinchuk V, Zinchuk O. Quantitative colocalization analysis of confocal fluorescence microscopy images. Curr Protoc Cell Biol. 2008;39:4.19. 11–14.19. 16.
Zinchuk V, Zinchuk O, Okada T. Quantitative colocalization analysis of multicolor confocal immunofluorescence microscopy images: pushing pixels to explore biological phenomena. Acta Histochem Cytochem. 2007;40:101–11.
Funding
This work was supported by the National Natural Science Foundation of China (NSFC) (No. 61805143).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 2865 kb)
Rights and permissions
About this article
Cite this article
Chen, H., Luo, C. & Zhang, S. Intracellular imaging and concurrent pH sensing of cancer-derived exosomes using surface-enhanced Raman scattering. Anal Bioanal Chem 413, 4091–4101 (2021). https://doi.org/10.1007/s00216-021-03365-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-021-03365-w