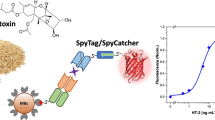
Abstract
Mycotoxins can be found as natural contaminants in many foods and feeds, and owing to their toxic effects, it is essential to detect them before they enter the food chain. An interesting approach for the analysis of mycotoxins by competitive immunoassays is the use of epitope-mimicking peptides, or mimotopes, which can replace the toxin conjugates traditionally used in such assays. Mimotopes can be selected from phage-displayed peptide libraries even without any prior knowledge of the antibody–antigen interaction, and after identifying the target specific clones, individual clones can be efficiently amplified in bacteria and used directly in the immunoassay. Following such approach, we have previously selected and identified a dodecapeptide which functions as a mimotope for the mycotoxin fumonisin B1. In this work, we present the development and comparison of various immunoassays based on this mimotope, named A2, which has been used in the phage-displayed format in which it was selected, but also as a fluorescent recombinant fusion protein or as a synthetic peptide. The highest sensitivity was obtained with a magnetic bead–based assay using the synthetic peptide and enzymatic detection which provided a detection limit of 0.029 ng mL−1. Analysis of the binding kinetics by surface plasmon resonance (SPR) further reinforced the suitability of the synthetic peptide for the competitive immunoassays, as this mimotope showed a slightly lower affinity for the target antibody in comparison with the recombinant fusion protein.

Graphical abstract



Similar content being viewed by others
References
Wild D. The immunoassay handbook: theory and applications of ligand binding, ELISA, and related techniques. 4th ed. Oxford: Elsevier; 2013.
Chauhan R, Singh J, Sachdev T, Basu T, Malhotra BD. Recent advances in mycotoxins detection. Biosens Bioelectron. 2016;81:532–45.
Qiu Y-L, He Q-H, Xu Y, Bhunia AK, Tu Z, Chen B, et al. Deoxynivalenol-mimic nanobody isolated from a naïve phage display nanobody library and its application in immunoassay. Anal Chim Acta. 2015;887:201–8.
Xiao H, Clarke JR, Marquardt RR, Frohlich AA. Improved methods for conjugating selected mycotoxins to carrier proteins and dextran for immunoassays. J Agric Food Chem. 1995;43:2092–7.
Peltomaa R, Benito-Peña E, Moreno-Bondi MC. Bioinspired recognition elements for mycotoxin sensors. Anal Bioanal Chem. 2018;410:747–71.
Berthiller F, Brera C, Crews C, Iha MH, Krska R, Lattanzio VMT, et al. Developments in mycotoxin analysis: an update for 2014-2015. World Mycotoxin J. 2016;9:5–30.
Freire FDCO, da Rocha MEB. Impact of mycotoxins on human health. In: Mérillon J-M, Ramawat KG, editors. Fungal metabolites. Cham: Springer International Publishing; 2016.
Goyal S, Ramawat KG, Mérillon JM. Different shades of fungal metabolites: an overview. In: Mérillon J-M, Ramawat KG, editors. Fungal metabolites. Cham: Springer International Publishing; 2016.
Bennett JW, Klich M. Mycotoxins. Clin Microbiol Rev. 2003;16:497–516.
European Commission Regulation (EC) No 1881/2006. Off J Eur Union. 2006;L364:5–24.
European Commission Regulation (EC) No 1126/2007. Off J Eur Union. 2007;L255:14–7.
European Commission Regulation (EC) No 105/2010. Off J Eur Union. 2010;L35:7–8.
European Commission Regulation (EC) No 165/2010. Off J Eur Union. 2010;L50:8–12.
Senyuva HZ, Gilbert J, Stroka J. Determination of fumonisins B1 and B2 in corn by LC/MS with immunoaffinity column cleanup: Interlaboratory study. J AOAC Int. 2010;93:211–21.
Solfrizzo M, Girolamo AD, Gambacorta L, Visconti A, Stroka J, van Egmond HP. Determination of fumonisins B1 and B2 in corn-based foods for infants and young children by LC with immunoaffinity column cleanup: Interlaboratory validation study. J AOAC Int. 2011;94:900–8.
Goryacheva IY, Saeger SD, Eremin SA, Peteghem CV. Immunochemical methods for rapid mycotoxin detection: evolution from single to multiple analyte screening: a review. Food Addit Contam. 2007;24:1169–83.
Köppen R, Koch M, Siegel D, Merkel S, Maul R, Nehls I. Determination of mycotoxins in foods: current state of analytical methods and limitations. Appl Microbiol Biotechnol. 2010;86:1595–612.
Wang Y, Wang H, Li P, Zhang Q, Kim HJ, Gee SJ, et al. Phage-displayed peptide that mimics aflatoxins and its application in immunoassay. J Agric Food Chem. 2013;61:2426–33.
Yuan Q, Pestka JJ, Hespenheide BM, Kuhn LA, Linz JE, Hart LP. Identification of mimotope peptides which bind to the mycotoxin deoxynivalenol-specific monoclonal antibody. Appl Environ Microbiol. 1999;8:65.
Peltomaa R, Benito-Peña E, Barderas R, Sauer U, González Andrade M, Moreno-Bondi MC. Microarray-based immunoassay with synthetic mimotopes for the detection of fumonisin B1. Anal Chem. 2017;89:6216–23.
Xu Y, Chen B, He Q, Qiu Y-L, Liu X, He Z, et al. New approach for development of sensitive and environmentally friendly immunoassay for mycotoxin fumonisin B1 based on using peptide-MBP fusion protein as substitute for coating antigen. Anal Chem. 2014;86:8433–40.
Liu X, Xu Y, He Q, He Z, Xiong Z. Application of mimotope peptides of fumonisin B1 in peptide ELISA. J Agric Food Chem. 2013;61:4765–70.
Liu R, Yu Z, He Q, Xu Y. An immunoassay for ochratoxin A without the mycotoxin. Food Control. 2007;18:872–7.
Lai W, Fung DYC, Yang X, Renrong L, Xiong Y. Development of a colloidal gold strip for rapid detection of ochratoxin A with mimotope peptide. Food Control. 2009;20:791–5.
He Z, He Q, Xu Y, Li Y, Liu X, Chen B, et al. Ochratoxin A mimotope from second-generation peptide library and its application in immunoassay. Anal Chem. 2013;85:10304–11.
Xu Y, He Z, He Q, Qiu Y, Chen B, Chen J, et al. Use of cloneable peptide–MBP fusion protein as a mimetic coating antigen in the standardized immunoassay for mycotoxin ochratoxin A. J Agric Food Chem. 2014;62:8830–6.
Zou X, Chen C, Huang X, Chen X, Wang L, Xiong Y. Phage-free peptide ELISA for ochratoxin A detection based on biotinylated mimotope as a competing antigen. Talanta. 2016;146:394–400.
He Q-H, Xu Y, Huang Y-H, Liu R-R, Huang Z-B, Li Y-P. Phage-displayed peptides that mimic zearalenone and its application in immunoassay. Food Chem. 2011;126:1312–5.
Barbas CFI, Burton DR, Scott JK, Silverman GJ. Phage display: a laboratory manual. Cold Spring Harbor: Cold Spring Harbor Laboratory Press; 2001.
Smith GP. Filamentous fusion phage: novel expression vectors that display cloned antigens on the virion surface. Science. 1985;228:1315–7.
McCafferty J, Griffiths AD, Winter G, Chiswell DJ. Phage antibodies: filamentous phage displaying antibody variable domains. Nature. 1990;348:552–4.
Peltomaa R, López-Perolio I, Benito-Peña E, Barderas R, Moreno-Bondi MC. Application of bacteriophages in sensor development. Anal Bioanal Chem. 2016;408:1805–28.
Smartt AE, Ripp S. Bacteriophage reporter technology for sensing and detecting microbial targets. Anal Bioanal Chem. 2011;400:991–1007.
Carlomagno M, Lassabe G, Rossotti M, González-Techera A, Vanrell L, González-Sapienza G. Recombinant streptavidin nanopeptamer anti-immunocomplex assay for noncompetitive detection of small analytes. Anal Chem. 2014;86:10467–73.
Ding Y, Hua X, Chen H, Liu F, González-Sapien G, Wang M. Recombinant peptidomimetic-nano luciferase tracers for sensitive single-step immunodetection of small molecules. Anal Chem. 2018;90:2230–7.
Ding Y, Hua X, Du M, Yang Q, Hou L, Wang L, et al. Recombinant, fluorescent, peptidomimetic tracer for immunodetection of imidaclothiz. Anal Chem. 2018;90:13996–4002.
Peltomaa R, Amaro-Torres F, Carrasco S, Orellana G, Benito-Peña E, Moreno-Bondi MC. Homogeneous quenching immunoassay for fumonisin B1 based on gold nanoparticles and an epitope-mimicking yellow fluorescent protein. ACS Nano. 2018;12:11333–42.
Matz MV, Fradkov AF, Labas YA, Savitsky AP, Zaraisky AG, Markelov ML, et al. Fluorescent proteins from nonbioluminescent Anthozoa species. Nat Biotechnol. 1999;17:969–73.
Clontech Living Colors User Manual, vol II. Red Fluorescent Protein, Protocol PT3404−1. www.clontech.com. Accessed 10 May 2018.
Marco M-P, Gee S, Hammock BD. Immunochemical techniques for environmental analysis I. Immunosensors. TrAC. 1995;14:341–50.
Herranz S, Marazuela MD, Moreno-Bondi MC. Automated portable array biosensor for multisample microcystin analysis in freshwater samples. Biosens Bioelectron. 2012;33:50–5.
Davies J, Roberts CJ, Dawkes AC, Sefton J, Edwards JC, Glasbey TO, et al. Use of scanning probe microscopy and surface plasmon resonance as analytical tools in the study of antibody-coated microtiter wells. Langmuir. 1994;10:2654–61.
Shen M, Rusling J, Dixit CK. Site-selective orientated immobilization of antibodies and conjugates for immunodiagnostics development. Methods. 2017;116:95–111.
He Q, Xu Y, Zhang C, Li Y, Huang Z. Phage-borne peptidomimetics as immunochemical reagent in dot-immunoassay for mycotoxin zearalenone. Food Control. 2014;39:56–61.
New England Biolabs (NEB) Ph.D. Phage Display Libraries - instruction manual. www.neb.com. Accessed 31 Jan 2019.
Ji Y, He Q, Xu Y, Tu Z, Yang H, Qiu Y, et al. Phage displayed anti-idiotypic nanobody mediated immuno-PCR for sensitive and environmentally friendly detection of mycotoxin ochratoxin A. Anal Methods. 2016;8:7824–31.
Shu M, Xu Y, Wang D, Liu X, Li Y, He Q, et al. Anti-idiotypic nanobody: a strategy for development of sensitive and green immunoassay for fumonisin B1. Talanta. 2015;143:388–93.
Acknowledgments
R.P. acknowledges UCM for a pre-doctoral grant and R.B. the PI17CIII/00045 grant from the AES-ISCIII program.
Funding
This research was funded by the Spanish Ministry of Science, Innovation and Universities, grant RTI2018-096410-B-C21.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Peltomaa, R., Agudo-Maestro, I., Más, V. et al. Development and comparison of mimotope-based immunoassays for the analysis of fumonisin B1. Anal Bioanal Chem 411, 6801–6811 (2019). https://doi.org/10.1007/s00216-019-02068-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-019-02068-7