Abstract
Rationale
Acute naloxone-precipitated morphine withdrawal (MWD) produces a conditioned place aversion (CPA) in rats even after one or two exposures to high-dose (20 mg/kg, sc) morphine followed 24-h later by naloxone (1 mg/kg, sc). However, the somatic withdrawal reactions produced by acute naloxone-precipitated MWD in rats have not been investigated. A recently discovered fatty acid amide, N-oleoylglycine (OlGly), which has been suggested to act as a fatty acid amide hydrolase (FAAH) inhibitor and as a peroxisome proliferator-activated receptor alpha (PPARα) agonist, was previously shown to interfere with a naloxone-precipitated MWD-induced CPA in rats.
Objectives
The aims of these studies were to examine the somatic withdrawal responses produced by acute naloxone-precipitated MWD and determine whether OlGly can also interfere with these responses.
Results
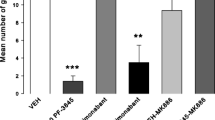
Here, we report that following two exposures to morphine (20 mg/kg, sc) each followed by naloxone (1 mg/kg, sc) 24 h later, rats display nausea-like somatic reactions of lying flattened on belly, abdominal contractions and diarrhea, and display increased mouthing movements and loss of body weight. OlGly (5 mg/kg, ip) interfered with naloxone-precipitated MWD-induced abdominal contractions, lying on belly, diarrhea and mouthing movements in male Sprague–Dawley rats, by both a cannabinoid 1 (CB1) and a PPARα mechanism of action. Since these withdrawal reactions are symptomatic of nausea, we evaluated the potential of OlGly to interfere with lithium chloride (LiCl)-induced and MWD-induced conditioned gaping in rats, a selective measure of nausea; the suppression of MWD-induced gaping reactions by OlGly was both CB1 and PPARα mediated.
Conclusion
These results suggest that the aversive effects of acute naloxone-precipitated MWD reflect nausea, which is suppressed by OlGly.





Similar content being viewed by others
Abbreviations
- AEA:
-
anandamide
- AM:
-
AM251
- ANOVA:
-
analysis of variance
- CB1 :
-
cannabinoid 1
- CB2 :
-
cannabinoid 2
- CPA:
-
conditioned place aversion
- FAAH:
-
fatty acid amide hydrolase
- ip:
-
intraperitoneal
- LiCl:
-
lithium chloride
- LOB:
-
lying on belly
- M:
-
morphine
- MK:
-
MK886
- MWD:
-
morphine withdrawal
- N:
-
naloxone
- OEA:
-
N-oleoylethanolamide
- OlGly:
-
N-oleoylglycine
- PEA:
-
N-palmitoylethanolamide
- PPARα:
-
peroxisome proliferator-activated receptor alpha
- S:
-
saline
- sc:
-
subcutaneous
- TR:
-
taste reactivity
- VEH:
-
vehicle
References
Contreras M, Ceric F, Torrealba F (2007) Inactivation of the interoceptive insula disrupts drug craving and malaise induced by lithium. Science 26:655–658. https://doi.org/10.1126/science.1145590
Donvito G, Piscitelli F, Muldoon P, Jackson A, Vitale RM, D’Aniello E et al (2019) N-Oleoyl-glycine reduces nicotine reward and withdrawal in mice. Neuropharmacology 148:320–331. https://doi.org/10.1016/j.neuropharm.2018.03.020
Eisenberg RM (1982) Further studies on the acute dependence produced by morphine in opiate naive rats. Life Sci 31:1531–1540. https://doi.org/10.1016/0024-3205(82)90043-1
Gellert VF, Sparber SB (1977) A comparison of the effects of naloxone upon body weight loss and suppression of fixed-ratio operant behavior in morphine-dependent rats. J Pharmacol ExpTher 201:44–54
Grill HJ, Norgren R (1978) The taste reactivity test: 1 Mimetic responses to gustatory stimuli in neurologically normal rats. Brain Res 143:263–279. https://doi.org/10.1016/0006-8993(78)90568-1
Heishman SJ, Stitzer ML, Bigelow GE, Liebson IA (1990) Acute opioid physical dependence in humans: effect of naloxone at 6 and 24 hours postmorphine. Pharmacol Biochem Behav 36:393–399. https://doi.org/10.1016/0091-3057(90)90421-D
Jaffee JH, Martin WR (1990) Opioid analgesics and antagonists. In: Gilman AG, Rall TW, Nies AS, Taylor P (eds) The pharmacological basis of therapeutics (8th ed), Pergamon, New York pp 485-521
June HL, Stitzer ML, Cone E (1995) Acute physical dependence: time course and relation to human plasma morphine concentrations. Clin Pharmacol Ther 57:270–280. https://doi.org/10.1016/0009-9236(95)90152-3
Koob GF (2009) Dynamics of neuronal circuits in addiction: reward, antireward, and emotional memory. Pharmacopsychiatry 42:S32–S41. https://doi.org/10.1055/s-0029-1216356
Koob GF, Wall TL, Bloom FE (1989) Nucleus accumbens as a substrate for the aversive stimulus effects of opiate withdrawal. Psychopharmacology 98:530–534
Limebeer CL, Vemuri VK, Bedard H, Lang ST, Ossenkopp KP, Makriyannis A et al (2010) Inverse agonism of cannabinoid CB1 receptors potentiates LiCl-induced nausea in the conditioned gaping model in rats. Br J Pharmacol 161:336–349. https://doi.org/10.1111/j.1476-5381.2010.00885.x
Maldonado R, Stinus L, Koob GF (1996) Neurobiological mechanisms of opiate withdrawal. R.G. Landes Company, Austin
Martin WR, Eades CG (1964) A comparison between acute and chronic physical dependence in the chronic spinal dog. J Pharmacol Exp Ther 146:385–394
Martin BR, Compton DR, Thomas BF, Prescott WR, Little PJ, Razdan RK et al (1991) Behavioral, biochemical, and molecular modeling evaluations of cannabinoid analogs. Pharmacol Biochem Behav 40:471–478. https://doi.org/10.1016/0091-3057(91)90349-7
McDonald RV, Parker LA, Siegel S (1997) Conditioned sucrose aversions produced by naloxone-precipitated withdrawal from acutely administered morphine. Pharmacol Biochem Behav 58:1003–1008. https://doi.org/10.1016/S0091-3057(97)00313-4
Meachum C, Bernstein IL (1992) Behavioral conditioned responses to contextual andodor stimuli paired with LiCl administration. Physiol Behav 52:895–899. https://doi.org/10.1016/0031-9384(92)90368-C
Parker LA (2014) Conditioned flavor avoidance and conditioned gaping: rat models of conditioned nausea. Eur J Pharmacol 722:122–133. https://doi.org/10.1016/j.ejphar.2013.09.070
Parker LA, Cyr JA, Santi AN, Burton PD (2002) The aversive properties of acute morphine dependence persist 48 h after a single exposure to morphine: evaluation by taste and place conditioning. Pharmacol Biochem Behav 72:87–92. https://doi.org/10.1016/S0091-3057(01)00724-9
Parker LA, Hills K, Jensen K (1984) Behavioral CRs elicited by a lithium- or an amphetamine-paired contextual test chamber. Anim Learn Behav 12:307–315. https://doi.org/10.3758/BF03199972
Parker LA, Joshi A (1998) Naloxone-precipitated morphine withdrawal induced place aversions: effect of naloxone at 24 hours postmorphine. Pharmacol Biochem Behav 61:331–333. https://doi.org/10.1016/S0091-3057(98)00104-X
Parker LA, Kwiatkowska M, Burton P, Mechoulam R (2004) Effect of cannabinoids on lithium-induced vomiting in the Suncus murinus (house musk shrew). Psychopharmacology 171:156–161. https://doi.org/10.1007/s00213-003-1571-2
Parker LA, Niphakis MJ, Downey R, Limebeer CL, Rock EM, Sticht MA, Morris H, Abdullah R, Lichtman AH, Cravatt BF (2014) A new MAGL inhibitor, MJN110, reduces acute and anticipatory nausea in rats and vomiting in Suncus murinus. Psychopharmacology 232:583–593. https://doi.org/10.1007/s00213-016-4277-y
Parker LA, Rennie M (1992) Naltrexone-induced aversions: Assessment by the placeConditioning, taste reactivity and taste avoidance paradigms. Pharmacol Biochem Behav 41:559–565. https://doi.org/10.1016/0091-3057(92)90373-n
Petrie GN, Wills KL, Piscitelli F, Smoum R, Limebeer CL, Rock EM et al (2019) Oleoyl glycine: interference with the aversive effects of acute naloxone-precipitated MWD, but not morphine reward, in male Sprague-Dawley rats. Psychopharmacology 236:2623–2633. https://doi.org/10.1007/s00213-019-05237-9
Ramesh D, Ross GR, Schlosburg JE, Owens R, Abdullah R, Kinsey SG et al (2011) Blockade of endocannabinoid hydrolytic enzymes attenuates precipitated opioid withdrawal symptoms in mice. J Pharmacol Exp Ther 339:173–185. https://doi.org/10.1124/jpet.111.181370
Rock EM, Limebeer CL, Ward JM, Cohen A, Grove K, Niphakis MJ, Cravatt BF, Parker LA. (2015).Fatty acid amide hydrolase (FAAH) inhibition interferes with acute nausea by a PPARα mechanism and anticipatory nausea by a CB1 receptor mechanism in a double dissociation. Psychopharmacology, 232: 3841-3848. https://doi.org/10.1007/s00213-015-4050-7
Rock EM, Moreno-Sanz G, Limebeer CL, Petrie G, Angelini R, Piomelli D et al (2017) Suppression of acute and anticipatory nausea by peripherally restricted FAAH inhibitor in animal models: role of PPARa and CB1 receptors. Br J Pharmacol 174:3837–3847. https://doi.org/10.1111/bph.13980
Rock EM, Limebeer CL, Aliasi-Sanai L, Parker LA (2019) The ventral pallidum as a critical region regulating fatty acid amide hydrolase inhibition of nausea-induced conditioned gaping in male Sprague-Dawley rats. Neuropharmacology 155:142–149. https://doi.org/10.1016/j.neuropharm.2019.05.031
Shoblock JR, Maidment NT (2006) Constitutively active mu opioid receptors mediate the enhanced conditioned aversvie effect of naloxone in morphine-dependent mice. Neuropsychopharmacology 31:171–177. https://doi.org/10.1038/sj.npp.1300782
Sticht MA, Limebeer CL, Rafla BR, Abdullah RA, Poklis JL, Ho W, Niphakis MJ, Cravatt BF, Sharkey KA, Lichtman AH, Parker LA (2016) Endocannabinoid regulation of nausea is mediated by 2-arachidonolyglycerol (2-AG) in the rat visceral insular cortex. Neuropharmacology 102:92–102. https://doi.org/10.1016/j.neuropharm.2015.10.039
Stinus L, Le Moal M, Koob GF (1990) Nucleus accumbens and amygdala are possible substrates for the aversive stimulus effects of opiate withdrawal. Neuroscience 37:767–773. https://doi.org/10.1016/0306-4522(90)90106-E
Tuerke KJ, Winters BD, Parker LA (2012) Ondansetron interferes with unconditioned lying-on belly and acquisition of conditioned gaping induced by LiCl as models of nausea-induced behaviors in rats. Physiol Behav 105:856–860. https://doi.org/10.1016/j.physbeh.2011.10.017
Wills KL, Petrie GN, Millett G, Limebeer CL, Rock EM, Niphakis MJ et al (2016) Double dissociation of monoacylglycerol lipase inhibition and CB1 antagonism in the central amygdala, basolateral amygdala, and the interoceptive insular cortex on the affective properties of acute naloxone-precipitated morphine withdrawal in rats. Neuropsychopharmacology 41:1865–1873. https://doi.org/10.1038/npp.2015.356
Wills KL, Vemuri K, Kalmar A, Lee A, Limebeer CL, Makriyannis A et al (2014) CB1 antagonism: interference with affective properties of acute naloxone-precipitated morphine withdrawal in rats. Psychopharmacology 231:4291–4300. https://doi.org/10.1007/s00213-014-3575-5
Funding
The research described here was funded by research grants from the Natural Sciences and Engineering Council of Canada (NSERC: 03629), Canadian Institutes of Health Research (CIHR:388239) and a research contract from PlantExt to Linda A Parker.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rock, E.M., Ayoub, S.M., Limebeer, C.L. et al. Acute naloxone-precipitated morphine withdrawal elicits nausea-like somatic behaviors in rats in a manner suppressed by N-oleoylglycine. Psychopharmacology 237, 375–384 (2020). https://doi.org/10.1007/s00213-019-05373-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-019-05373-2