Abstract
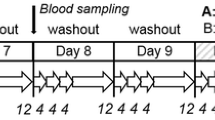
The present human intervention study investigated the relation between the intake of acrylamide (AA) in diets with minimized, low, and high AA contents and the levels of urinary exposure biomarkers. As biomarkers, the mercapturic acids, N-acetyl-S-(carbamoylethyl)-l-cysteine (AAMA), and N-acetyl-S-(1-carbamoyl-2-hydroxyethyl)-l-cysteine (GAMA) were monitored. The study was performed with 14 healthy male volunteers over a period of 9 days, under controlled conditions excluding any inadvertent AA exposure. Dietary exposure to AA was measured by determining AA contents in duplicates of all meals consumed by the volunteers. The study design included an initial washout period of 3 days on AA-minimized diet, resulting in dietary AA exposure not exceeding 41 ng/kg bw/d. Identical washout periods of 2 days each followed the AA exposure days (day 4, low exposure, and day 7, high exposure). At the respective AA intake days, volunteers ingested 0.6–0.8 (low exposure) or 1.3–1.8 (high exposure) μg AA/kg bw/d with their food. Both low and high AA intakes resulted in an AAMA output within 72 h corresponding to 58 % of the respective AA intake. At the end of the initial 3-day washout period, an AAMA baseline level of 93 ± 31 nmol/d was recorded, suggestive for an assumed net AA baseline exposure level of 0.2–0.3 μg AA/kg bw/d.




Similar content being viewed by others
References
Berger FI, Feld J, Bertow D, Eisenbrand G, Fricker G, Gerhardt N, Merz K-H, Richling E, Baum M (2011) Biological effects of acrylamide after daily ingestion of various foods in comparison to water: a study in rats. Mol Nutr Food Res 55:387–399
Boettcher MI, Bolt HM, Drexler H, Angerer J (2006) Excretion of mercapturic acids of acrylamide and glycidamide in human urine after single oral administration of deuterium-labelled acrylamide. Arch Toxicol 80:55–61
EFSA CONTAM Panel (EFSA panel on contaminants in the food chain) (2014) Draft scientific opinion on acrylamide in food. http://www.efsa.europa.eu/de/consultations/call/140701.pdf
Fennell TR, Sumner SC, Snyder RW, Burgess J, Spicer R, Bridson WE, Friedman MA (2005) Metabolism and hemoglobin adduct formation of acrylamide in humans. Toxicol Sci 85:447–459
Fuhr U, Boettcher MI, Kinzig-Schippers M, Weyer A, Jetter A, Lazar A, Taubert D, Tomalik-Scharte D, Pournara P, Jakob V (2006) Toxicokinetics of acrylamide in humans after ingestion of a defined dose in a test meal to improve risk assessment for acrylamide carcinogenicity. Cancer Epidemol Biomark 15:266–271
Gamboa da Costa G, Churchwell MI, Hamilton LP, Von Tungeln LS, Beland FA, Marques MM, Doerge DR (2003) DNA adduct formation from acrylamide via conversion to glycidamide in adult and neonatal mice. Chem Res Toxicol 16:1328–1337
Hartmann EC, Boettcher MI, Schettgen T, Fromme H, Drexler H, Jr Angerer (2008) Hemoglobin adducts and mercapturic acid excretion of acrylamide and glycidamide in one study population. J Agric Food Chem 56:6061–6068
Mottram DS, Wedzicha BL, Dodson AT (2002) Food chemistry: acrylamide is formed in the Maillard reaction. Nature 419:448–449
Stadler RH, Blank I, Varga N, Robert F, Hau J, Guy PA, Robert M-C, Riediker S (2002) Food chemistry: acrylamide from Maillard reaction products. Nature 419:449–450
Tareke E, Lyn-Cook B, Robinson B, Ali SF (2008) Acrylamide: a dietary carcinogen formed in vivo? J Agric Food Chem 56:6020–6023
Tareke E, Heinze TM, Gamboa da Costa G, Ali S (2009) Acrylamide formed at physiological temperature as a result of asparagine oxidation. J Agric Food Chem 57:9730–9733
Watzek N, Bohm N, Feld J, Scherbl D, Berger F, Merz KH, Lampen A, Reemtsma T, Tannenbaum SR, Skipper PL, Baum M, Richling E, Eisenbrand G (2012a) N7-glycidamide-guanine DNA adduct formation by orally ingested acrylamide in rats: a dose-response study encompassing human diet-related exposure levels. Chem Res Toxicol 25:381–390
Watzek N, Scherbl D, Feld J, Berger F, Doroshyenko O, Fuhr U, Tomalik-Scharte D, Baum M, Eisenbrand G, Richling E (2012b) Profiling of mercapturic acids of acrolein and acrylamide in human urine after consumption of potato crisps. Mol Nutr Food Res 56:1825–1837
Acknowledgments
We thank our volunteers for their participation in the study and the members of the research group as well as the study director Dr. Jens Galan for conducting the study. We also thank Dorothea Schipp for the statistical evaluation and Sylvia Schmidt, Patricia Podsiadly, Byron Baus, and Johannes Keller for supporting laboratory work. This study has been supported by Institute for Scientific Information on Coffee (ISIC), Entre deux Villes 10-1814 La Tour de Peliz-Switzerland.
Conflict of interest
G. Eisenbrand serves as scientific advisor of ISIC.
Ethical standards
The study was approved by the Ethics Commission of Rhineland-Palatinate, Mainz (No. 837.449.12 (8600-F)) and performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ruenz, M., Bakuradze, T., Eisenbrand, G. et al. Monitoring urinary mercapturic acids as biomarkers of human dietary exposure to acrylamide in combination with acrylamide uptake assessment based on duplicate diets. Arch Toxicol 90, 873–881 (2016). https://doi.org/10.1007/s00204-015-1494-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-015-1494-9