Abstract
Purpose
To evaluate the potential benefit of curative radiotherapy (RT) to the primary tumor in metastatic castration-resistant prostate cancer (mCRPC) patients treated with abiraterone.
Materials and methods
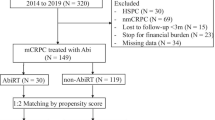
The clinical parameters of 106 mCRPC patients treated with abiraterone were retrospectively evaluated. Patients were either oligometastatic (≤5 metastases) at diagnosis or became oligometastatic after the systemic treatment was analyzed. Local RT to the primary tumor and pelvic lymphatics was delivered in 44 patients (41%), and 62 patients (59%) did not have RT to the primary tumor. After propensity match analysis, a total of 92 patients were analyzed.
Resultsn
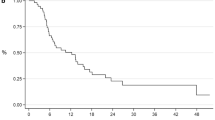
Median follow-up time was 14.2 months (range: 2.3–54.9 months). Median overall survival (OS) was higher in patients treated with local RT to the primary tumor than in those treated without local RT with borderline significance (24.1 vs. 21.4 months; p = 0.08). Local RT to the prostate and pelvic lymphatics significantly diminished the local recurrence rate (16 patients, 31% vs. 2 patients, 5%; p = 0.003). In multivariate analysis, the prostate specific antigen (PSA) response ≥50% of the baseline obtained 3 weeks after abiraterone therapy was the only significant prognostic factor for better OS and progression-free survival (PFS). Patients treated with primary RT to the prostate had significantly less progression under abiraterone and a longer abiraterone period than those treated without local prostate RT.
Conclusions
Local prostate RT significantly improved OS and local control in mCRPC patients treated with abiraterone. The patients treated with primary RT had significantly less progression under abiraterone and a longer abiraterone period than those treated without local prostate RT.
Zusammenfassung
Ziel
Bewertung des potenziellen Nutzens der kurativen Strahlentherapie (RT) für den Primärtumor bei Patienten mit metastasiertem kastrationsresistentem Prostatakrebs (mCRPC), die mit Abirateron behandelt werden.
Material und Methoden
Die klinischen Parameter von 106 mit Abirateron behandelten mCRPC-Patienten wurden retrospektiv bewertet. Die Patienten waren bei der Diagnose entweder oligometastatisch (≤5 Metastasen) oder wurden nach Analyse der systemischen Behandlung oligometastatisch. Eine lokale RT für Primärtumor und Beckenlymphknoten bekamen 44 Patienten (41%), 62 Patienten (59%) hatten keine RT des Primärtumors. Nach der Propensity-Match-Analyse wurden insgesamt 92 Patienten analysiert.
Ergebnisse
Die mediane Nachbeobachtungszeit betrug 14,2 Monate (Spanne 2,3–54,9 Monate). Das mediane Gesamtüberleben (OS) war bei den mit lokaler RT behandelten Patienten gegenüber dem Primärtumor höher als bei denjenigen ohne lokale RT, mit grenzwertiger Signifikanz (24,1 vs. 21,4 Monate; p = 0,08). Die lokale RT für die Prostata- und Beckenlymphatika verringerte signifikant die lokale Rezidivrate (16 Patienten, 31% vs. 2 Patienten, 5%; p = 0,003). In der multivariaten Analyse war die 3 Wochen nach Abiraterontherapie ermittelte PSA-Antwort ≥50 des Ausgangswerts der einzige signifikante Prognosefaktor für ein besseres OS und progressionsfreies Überleben (PFS). Patienten, die mit primärer Prostata-RT behandelt wurden, zeigten eine signifikant geringere Progression unter Abirateron und eine längere Abirateron-Periode als Patienten ohne lokale Prostata-RT.
Schlussfolgerung
Die lokale Prostata-RT verbesserte das OS und die lokale Kontrolle bei mit Abirateron behandelten mCRPC-Patienten signifikant. Die mit primärer RT behandelten Patienten hatten unter Abirateron eine deutlich geringere Progression und eine längere Abirateron-Periode als Patienten, die ohne lokale Prostata-RT behandelt wurden.


Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A (2017) Cancer Statistics, 2017. CA Cancer J Clin 67(1):7–30. https://doi.org/10.3322/caac.21387
Crawford ED, Higano CS, Shore ND, Hussain M, Petrylak DP (2015) Treating patients with metastatic castration resistant prostate cancer: a comprehensive review of available therapies. J Urol 194(6):1537–1547. https://doi.org/10.1016/j.juro.2015.06.106
de Bono JS, Logothetis CJ, Molina A, Fizazi K, North S, Chu L, Chi KN, Jones RJ, Goodman OB Jr., Saad F, Staffurth JN, Mainwaring P, Harland S, Flaig TW, Hutson TE, Cheng T, Patterson H, Hainsworth JD, Ryan CJ, Sternberg CN, Ellard SL, Flechon A, Saleh M, Scholz M, Efstathiou E, Zivi A, Bianchini D, Loriot Y, Chieffo N, Kheoh T, Haqq CM, Scher HI, COU-AA-301 Investigators (2011) Abiraterone and increased survival in metastatic prostate cancer. N Engl J Med 364(21):1995–2005. https://doi.org/10.1056/NEJMoa1014618
Scher HI, Fizazi K, Saad F, Taplin ME, Sternberg CN, Miller K, de Wit R, Mulders P, Chi KN, Shore ND, Armstrong AJ, Flaig TW, Flechon A, Mainwaring P, Fleming M, Hainsworth JD, Hirmand M, Selby B, Seely L, de Bono JS, AFFIRM Investigators (2012) Increased survival with enzalutamide in prostate cancer after chemotherapy. N Engl J Med 367(13):1187–1197. https://doi.org/10.1056/NEJMoa1207506
de Bono JS, Oudard S, Ozguroglu M, Hansen S, Machiels JP, Kocak I, Gravis G, Bodrogi I, Mackenzie MJ, Shen L, Roessner M, Gupta S, Sartor AO, TROPIC Investigators (2010) Prednisone plus cabazitaxel or mitoxantrone for metastatic castration-resistant prostate cancer progressing after docetaxel treatment: a randomised open-label trial. Lancet 376(9747):1147–1154. https://doi.org/10.1016/S0140-6736(10)61389-X
Kantoff PW, Higano CS, Shore ND, Berger ER, Small EJ, Penson DF, Redfern CH, Ferrari AC, Dreicer R, Sims RB, Xu Y, Frohlich MW, Schellhammer PF, IMPACT Study Investigators (2010) Sipuleucel-T immunotherapy for castration-resistant prostate cancer. N Engl J Med 363(5):411–422. https://doi.org/10.1056/NEJMoa1001294
Parker C, Nilsson S, Heinrich D, Helle SI, O’Sullivan JM, Fossa SD, Chodacki A, Wiechno P, Logue J, Seke M, Widmark A, Johannessen DC, Hoskin P, Bottomley D, James ND, Solberg A, Syndikus I, Kliment J, Wedel S, Boehmer S, Dall’Oglio M, Franzen L, Coleman R, Vogelzang NJ, O’Bryan-Tear CG, Staudacher K, Garcia-Vargas J, Shan M, Bruland OS, Sartor O, ALSYMPCA Investigators (2013) Alpha emitter radium-223 and survival in metastatic prostate cancer. N Engl J Med 369(3):213–223. https://doi.org/10.1056/NEJMoa1213755
Beer TM, Armstrong AJ, Rathkopf DE, Loriot Y, Sternberg CN, Higano CS, Iversen P, Bhattacharya S, Carles J, Chowdhury S, Davis ID, de Bono JS, Evans CP, Fizazi K, Joshua AM, Kim CS, Kimura G, Mainwaring P, Mansbach H, Miller K, Noonberg SB, Perabo F, Phung D, Saad F, Scher HI, Taplin ME, Venner PM, Tombal B, PREVAIL Investigators (2014) Enzalutamide in metastatic prostate cancer before chemotherapy. N Engl J Med 371(5):424–433. https://doi.org/10.1056/NEJMoa1405095
Ryan CJ, Smith MR, Fizazi K, Saad F, Mulders PF, Sternberg CN, Miller K, Logothetis CJ, Shore ND, Small EJ, Carles J, Flaig TW, Taplin ME, Higano CS, de Souza P, de Bono JS, Griffin TW, De Porre P, Yu MK, Park YC, Li J, Kheoh T, Naini V, Molina A, Rathkopf DE, COU-AA-302 Investigators (2015) Abiraterone acetate plus prednisone versus placebo plus prednisone in chemotherapy-naive men with metastatic castration-resistant prostate cancer (COU-AA-302): final overall survival analysis of a randomised, double-blind, placebo-controlled phase 3 study. Lancet Oncol 1(4):152–160. https://doi.org/10.1016/S1470-2045
Dy SM, Asch SM, Naeim A, Sanati H, Walling A, Lorenz KA (2008) Evidence-based standards for cancer pain management. J Clin Oncol 26(23):3879–3885. https://doi.org/10.1200/JCO.2007.15.9517
Mickisch GH, Garin A, van Poppel H, de Prijck L, Sylvester R, European Organisation for Research and Treatment of Cancer (EORTC) Genitourinary Group (2001) Radical nephrectomy plus interferon-alfa-based immunotherapy compared with interferon alfa alone in metastatic renal-cell carcinoma: a randomised trial. Lancet 358(9286):966–970
Temple LK, Hsieh L, Wong WD, Saltz L, Schrag D (2004) Use of surgery among elderly patients with stage IV colorectal cancer. J Clin Oncol 22(17):3475–3484. https://doi.org/10.1200/JCO.2004.10.218
Alicikus ZA, Yamada Y, Zhang Z, Pei X, Hunt M, Kollmeier M, Cox B, Zelefsky MJ (2011) Ten-year outcomes of high-dose, intensity-modulated radiotherapy for localized prostate cancer. Cancer 117(7):1429–1437. https://doi.org/10.1002/cncr.25467
Culp SH, Karam JA, Wood CG (2014) Population-based analysis of factors associated with survival in patients undergoing cytoreductive nephrectomy in the targeted therapy era. Urol Oncol 32(5):561–568. https://doi.org/10.1016/j.urolonc.2013.12.003
Fossati N, Trinh QD, Sammon J, Sood A, Larcher A, Sun M, Karakiewicz P, Guazzoni G, Montorsi F, Briganti A, Menon M, Abdollah F (2015) Identifying optimal candidates for local treatment of the primary tumor among patients diagnosed with metastatic prostate cancer: a SEER-based study. Eur Urol 67(1):3–6. https://doi.org/10.1016/j.eururo.2014.08.056
Satkunasivam R, Kim AE, Desai M, Nguyen MM, Quinn DI, Ballas L, Lewinger JP, Stern MC, Hamilton AS, Aron M, Gill IS (2015) Radical prostatectomy or external beam radiation therapy vs no local therapy for survival benefit in metastatic prostate cancer: a SEER-medicare analysis. J Urol 194(2):378–385. https://doi.org/10.1016/j.juro.2015.02.084
Scher HI, Halabi S, Tannock I, Morris M, Sternberg CN, Carducci MA, Eisenberger MA, Higano C, Bubley GJ, Dreicer R, Petrylak D, Kantoff P, Basch E, Kelly WK, Figg WD, Small EJ, Beer TM, Wilding G, Martin A, Hussain M, Prostate Cancer Clinical Trials Working Group (2008) Design and end points of clinical trials for patients with progressive prostate cancer and castrate levels of testosterone: recommendations of the Prostate Cancer Clinical Trials Working Group. J Clin Oncol 26(7):1148–1159. https://doi.org/10.1200/JCO.2007.12.4487
Aizawa R, Takayama K, Nakamura K, Inoue T, Kobayashi T, Akamatsu S, Yamasaki T, Ogawa O, Mizowaki T (2018) Long-term outcomes of definitive external-beam radiotherapy for non-metastatic castration-resistant prostate cancer. Int J Clin Oncol. https://doi.org/10.1007/s10147-018-1265-8
Lankford SP, Pollack A, Zagars GK (1995) Radiotherapy for regionally localized hormone refractory prostate cancer. Int J Radiat Oncol Biol Phys 33(4):907–912. https://doi.org/10.1016/0360-3016(95)02005-0
Botticella A, Guarneri A, Filippi AR, Levra NG, Munoz F, Ragona R, Gontero P, Ricardi U (2013) May non-metastatic clinically localized castration-resistant prostate cancer after primary androgen ablation benefit from salvage prostate radiotherapy? J Cancer Res Clin Oncol 139(11):1955–1960. https://doi.org/10.1007/s00432-013-1520-3
Tannock IF, de Wit R, Berry WR, Horti J, Pluzanska A, Chi KN, Oudard S, Theodore C, James ND, Turesson I, Rosenthal MA, Eisenberger MA, TAX 327 Investigators (2004) Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med 351(15):1502–1512. https://doi.org/10.1056/NEJMoa040720
Fidler IJ, Poste G (2008) The “seed and soil” hypothesis revisited. Lancet Oncol 9(8):808. https://doi.org/10.1016/S1470-2045(08)70201-8
Kaplan RN, Rafii S, Lyden D (2006) Preparing the “soil”: the premetastatic niche. Cancer Res 66(23):11089–11093. https://doi.org/10.1158/0008-5472.CAN-06-2407
Kim MY, Oskarsson T, Acharyya S, Nguyen DX, Zhang XH, Norton L, Massague J (2009) Tumor self-seeding by circulating cancer cells. Cell 139(7):1315–1326. https://doi.org/10.1016/j.cell.2009.11.025
Resel Folkersma L, San Jose Manso L, Galante Romo I, Moreno Sierra J, Olivier Gomez C (2012) Prognostic significance of circulating tumor cell count in patients with metastatic hormone-sensitive prostate cancer. Urology 80(6):1328–1332. https://doi.org/10.1016/j.urology.2012.09.001
Flanigan RC, Salmon SE, Blumenstein BA, Bearman SI, Roy V, McGrath PC, Caton JR Jr., Munshi N, Crawford ED (2001) Nephrectomy followed by interferon alfa-2b compared with interferon alfa-2b alone for metastatic renal-cell cancer. N Engl J Med 345(23):1655–1659. https://doi.org/10.1056/NEJMoa003013
Rusthoven CG, Jones BL, Flaig TW, Crawford ED, Koshy M, Sher DJ, Mahmood U, Chen RC, Chapin BF, Kavanagh BD, Pugh TJ (2016) Improved survival with prostate radiation in addition to androgen deprivation therapy for men with newly diagnosed metastatic prostate cancer. J Clin Oncol 34(24):2835–2842. https://doi.org/10.1200/jco.2016.67.4788
Kirby M, Hirst C, Crawford ED (2011) Characterising the castration-resistant prostate cancer population: a systematic review. Int J Clin Pract 65(11):1180–1192. https://doi.org/10.1111/j.1742-1241.2011.02799.x
Ryan CJ, Smith MR, de Bono JS, Molina A, Logothetis CJ, de Souza P, Fizazi K, Mainwaring P, Piulats JM, Ng S, Carles J, Mulders PF, Basch E, Small EJ, Saad F, Schrijvers D, Van Poppel H, Mukherjee SD, Suttmann H, Gerritsen WR, Flaig TW, George DJ, Yu EY, Efstathiou E, Pantuck A, Winquist E, Higano CS, Taplin ME, Park Y, Kheoh T, Griffin T, Scher HI, Rathkopf DE, COU-AA-302 Investigators (2013) Abiraterone in metastatic prostate cancer without previous chemotherapy. N Engl J Med 368(2):138–148. https://doi.org/10.1056/NEJMoa1209096
O’Donnell A, Judson I, Dowsett M, Raynaud F, Dearnaley D, Mason M, Harland S, Robbins A, Halbert G, Nutley B, Jarman M (2004) Hormonal impact of the 17alpha-hydroxylase/C(17,20)-lyase inhibitor abiraterone acetate (CB7630) in patients with prostate cancer. Br J Cancer 90(12):2317–2325. https://doi.org/10.1038/sj.bjc.6601879
Luu-The V, Belanger A, Labrie F (2008) Androgen biosynthetic pathways in the human prostate. Best Pract Res Clin Endocrinol Metab 22(2):207–221. https://doi.org/10.1016/j.beem.2008.01.008
Mostaghel EA, Cho E, Zhang A, Alyamani M, Kaipainen A, Green S, Marck BT, Sharifi N, Wright JL, Gulati R, True LD, Loda M, Matsumoto AM, Tamae D, Penning TN, Balk SP, Kantoff PW, Nelson PS, Taplin ME, Montgomery RB (2017) Association of tissue Abiraterone levels and SLCO genotype with Intraprostatic steroids and pathologic response in men with high-risk localized prostate cancer. Clin Cancer Res 23(16):4592–4601. https://doi.org/10.1158/1078-0432.CCR-16-2245
Rescigno P, Lorente D, Bianchini D, Ferraldeschi R, Kolinsky MP, Sideris S, Zafeiriou Z, Sumanasuriya S, Smith AD, Mehra N, Jayaram A, Perez-Lopez R, Mateo J, Parker C, Dearnaley DP, Tunariu N, Reid A, Attard G, de Bono JS (2016) Prostate-specific antigen decline after 4 weeks of treatment with abiraterone acetate and overall survival in patients with metastatic castration-resistant prostate cancer. Eur Urol 70(5):724–731. https://doi.org/10.1016/j.eururo.2016.02.055
Sridhara R, Eisenberger MA, Sinibaldi VJ, Reyno LM, Egorin MJ (1995) Evaluation of prostate-specific antigen as a surrogate marker for response of hormone-refractory prostate cancer to suramin therapy. J Clin Oncol 13(12):2944–2953. https://doi.org/10.1200/JCO.1995.13.12.2944
Kelly WK, Scher HI, Mazumdar M, Vlamis V, Schwartz M, Fossa SD (1993) Prostate-specific antigen as a measure of disease outcome in metastatic hormone-refractory prostate cancer. J Clin Oncol 11(4):607–615. https://doi.org/10.1200/JCO.1993.11.4.607
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
B.A. Yildirim, C. Onal, F. Kose, E. Oymak, A.M. Sedef, A.A. Besen, S. Aksoy, O.C. Guler, A.T. Sumbul, S. Muallaoglu, H. Mertsoylu, and G. Ozyigit declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Yildirim, B.A., Onal, C., Kose, F. et al. Outcome of loco-regional radiotherapy in metastatic castration-resistant prostate cancer patients treated with abiraterone acetate. Strahlenther Onkol 195, 872–881 (2019). https://doi.org/10.1007/s00066-019-01429-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-019-01429-6