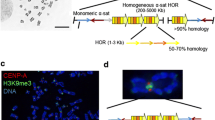
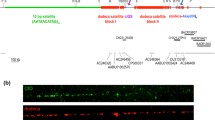
Abstract
Yeast artificial chromosomes (YACs) spanning the centromeric region of the human Y chromosome were introduced into mouse LA-9 cells by spheroplast fusion in order to determine whether they would form mammalian artificial chromosomes. In about 50% of the cell lines generated, the YAC DNA was associated with circular extrachromosomal structures. These episomes were only present in a proportion of the cells, usually at high copy number, and were lost rapidly in the absence of selection. These observations suggest that, despite the presence of centromeric sequences, the structures were not segregating efficiently and thus were not forming artificial chromosomes. However, extrachromosomal structures containing alphoid DNA appeared cytogenetically smaller than those lacking it, as long as yeast DNA was also absent. This suggests that alphoid DNA can generate the condensed chromatin structure at the centromere.
Similar content being viewed by others
References
Allshire RC, Cranston G, Gosden JR, Maule JC, Hastie ND, Fantes PA (1987) A fission yeast chromosome can replicate autonomously in mouse cells. Cell 50: 391–403
Baum M, Ngan VK, Clarke L (1994) The centromeric K-type repeat and the central core are together sufficient to establish a functionalSchizosaccharomyces pombe centromere. Mol Biol Cell 5: 747–761
Bayne RAL, Broccoli D, Taggart MH, Thomson EJ, Farr CJ, Cooke HJ (1994) Sandwiching of a gene within 12 kb of a functional telomere and alpha satellite does not result in silencing. Hum Mol Genet 3: 539–546
Blennow E, Telenius H, de Vos D, Larsson C, Herriksson P, Johansson O, Carter NP, Nordenskjold M (1994) Tetrasomy 15q: two marker chromosomes with no detectable alpha-satellite DNA. Am J Hum Genet 54: 877–883
Brown KE, Barnett MA, Burgtorf C, Shaw P, Buckle VJ, Brown WRA (1994) Dissecting the centromere of the human Y chromosome with cloned telomeric DNA. Hum Mol Genet 3: 1227–1237
Brown W, Tyler-Smith C (1995) Centromere activation. Trends Genet 11: 337–339
Burke DT, Carle GF, Olson MV (1987) Cloning of large segments of exogenous DNA into yeast by means of artificial chromosome vectors. Science 236: 806–12
Cohen D, Chumakov I, Weissenbach J (1993) A first-generation physical map of the human genome. Nature 366: 698–701
Cooke CA, Bernat RL, Earnshaw WC (1990) CENP-B: a major human centromere protein located beneath the kinetochore. J Cell Biol 110: 1475–1488
Cooper KF, Fisher RB, Tyler-Smith C (1992) Structure of the pericentric long arm region of the human Y chromosome. J Mol Biol 228: 421–432
Cooper KF, Fisher RB, Tyler-Smith C (1993) Structure of the sequences adjacent to the centromeric alphoid satellite DNA array on the human Y chromosome. J Mol Biol 230: 787–799
Cramer JH, Farrelly FW, Rownd RH (1976) Restriction endonuclease analysis of ribosomal DNA fromSaccharomyces cerevisiae. Mol Gen Genet 148: 233–241
Den Dunnen JT, Grootscholten PM, Dauwerse, JG, Walker AP, Monaco AP, Butler R, Anand R, Coffey AJ, Bentley DR, Steensma HY, Van Ommen GJB (1992) Reconstruction of the 2.4Mb DMD-gene by homologous recombination. Hum Mol Genet 1: 19–28
Earnshaw WC, Sullivan KF, Machlin PS, Cooke CA, Kaiser DA, Pollard TD, Rothfield NF, Cleveland DW (1987) Molecular cloning of cDNA for CENP-B, the major human centromere autoantigen. J Cell Biol 104: 817–829
Featherstone T, Huxley C (1993) Extrachromosomal maintenance and amplification of yeast artificial chromosome DNA in mouse cells. Genomics 17: 267–278
Feinberg AP, Vogelstein B (1984) A technique for radiolabelling DNA restriction fragments to high specific activity. Anal Biochem 137: 266–267
Gaff C, du Sart D, Kalitsis P, Iannello R, Nagy A, Choo KHA (1994) A novel nuclear protein binds centromeric alpha satellite DNA. Hum Mol Genet 3: 711–716
Green ED, Olson MV (1990) Chromosomal region of the cystic fibrosis gene in yeast artificial chromosomes: a model for human genome mapping. Science 250: 94–8
Haaf T, Warburton PE, Willard HF (1992) Integration of human α-satellite DNA into simian chromosomes: centromere protein binding and disruption of normal chromosome segregation. Cell 70: 681–696
Hahnenberger KM, Carbon J, Clarke L (1991) Identification of DNA regions required for mitotic and meiotic functions with-in the centromere ofSchizosaccharomyces pombe chromosome I. Mol Cell Biol 11: 2206–2215
Hegemann JH, Fleig UN (1993) The centromere of budding yeast. BioEssays 15: 451–460
Hill A, Bloom K (1987) Genetic manipulation of centromere function. Mol Cell Biol 7: 2397–2405
Huxley C, Green ED, Dunham I (1990) Rapid assessment ofS. cerevisiae mating type by PCR. Trends Genet 6: 236
Kipling D, Mitchell AR, Masumoto H, Wilson HE, Nicol L, Cooke HJ (1995) CENP-B binds a novel centromeric sequence in the Asian mouseMus caroli. Mol Cell Biol 15: 4009–4020
Koshland D, Rutledge L, Fitzgerlad-Hayes M, Hartwell LH (1987) A genetic analysis of dicentric minichromosomes inSaccharomyces cerevisiae. Cell 48: 801–812
Lanini L, McKeon F (1995) Domains required for CENP-C assembly at the kinetochore. Mol Biol Cell 6: 1049–1059
Larin Z, Lerach H (1990) Yeast artificial chromosomes: An alternative approach to the molecular analysis of mouse developmental mutations. Genet Res 56: 203–208
Larin Z, Fricker MD, Tyler-Smith C (1994) De novo formation of several features of a centromere following introduction of a Y alphoid YAC into mammalian cells. Hum Mol Genet 3: 689–695
Larin Z, Monaco AP, Lerach H (1995) Generation of large insert YAC libraries. In: Markie D (ed) Methods in molecular biology, vol 54: YAC protocols. Human Press, Totowa, NJ, pp 1–11
Lee JT, Murgia A, Sosnoski D, Olivos IM, Nussbaum RL (1992) Construction and characterization of a yeast artificial chromosome library for Xpter-Xq27.3: a systematic determination of cocloning rate and X-chromosome representation. Genomics 12: 526–533
Mann C, Davis RW (1983) Instability of dicentric plasmids in yeast. Proc Natl Acad Sci USA 80: 228–232
Marcus M, Tantravahi R, Dev VG, Miller DA, Miller OJ (1976) Human-mouse cell hybrid with human multiple Y chromosomes. Nature 262: 63–65
Masumoto H, Masukata H, Muro Y, Nozaki N, Okazaki T (1989) A human centromere antigen (CENP-B) interacts with a short specific sequence in alphoid DNA, a human centromeric satellite. J Cell Biol 109: 1963–1973
McManus J, Perry P, Sumner AT, Wright DM, Thomson EJ, Allshire RC, Hastie ND, Bickmore WA (1994) Unusual chromosome structure of fission yeast DNA in mouse cells. J Cell Sci 107: 469–486
Murakami S, Matsumoto T, Niw O, Yanagida M (1991) Structure of the fission yeast centromere cen3; direct analysis of the reiterated inverted region. Chromosoma 101: 214–221
Murray AW, Szostak JW (1983) Construction of artificial chromosomes in yeast. Nature 305: 189–193
Neil DL, Villasante A, Fisher R, Vetrie D, Cox B, Tyler-Smith C (1990) Structural instability of human tandemly repeated DNA sequences cloned in yeast artificial chromosome vectors. Nucleic Acids Res 18: 1421–1428
Ohashi H, Wakui K, Ogawa K, Okano T, Niikawa N, Fukshima Y (1994) A stable acentric marker chromosome: possible existence of an intercalary ancient centromere at distal 8p. Am J Hum Genet 55: 1202–1208
Petrovic V, Nasioulas S, Chow CW, Voullaire L, Schmidt M, Dahl H (1992) Minute Y chromosome derived marker in a child with gonadoblastoma: cytogenetic and DNA studies. J Med Genet 29: 542–546
Saitoh H, Tomkiel J, Cooke CA, Ratrie H, Maurer M, Rothfield NF, Earnshaw WC (1992) CENP-C, an autoantigen in scleroderma, is a component of the human inner kinetochore plate. Cell 70: 115–125
Sherman F, Fink GR, Hicks JB (1986) Methods in yeast genetics: a laboratory course manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY
Southern EM (1975) Detection of specific DNA fragments among DNA fragments separated by gel electrophoresis. J Mol Biol 98: 503–517
Southern EM, Anand R, Brown WRA, Fletcher DS (1987) A model for the separation of large DNA molecules by crossed field gel electrophoresis. Nucleic Acids Res 15: 5925–5943
Steiner NC, Clarke L (1994) A novel epigenetic effect can alter centromere function in fission yeast. Cell 79: 865–874
Steiner NC, Hahnenberger KM, Clarke L (1993) Centormeres of the fission yeastSchizosaccharomyces pombe are highly variable genetic loci. Mol Cell Biol 13: 4578–4587
Sullivan KF, Glass CA (1991) CENP-B is a highly conserved mammalian centromere protein with homology to the helixloop-helix family of proteins. Chromosoma 100: 360–370
Taylor SS (1995) Manipulation of YACs to construct a Mammalian Artificial Chromosome. DPhil Thesis, University of Oxford
Taylor SS, Larin Z, Tyler-Smith C (1994) Addition of functional human telomeres to YAcs. Hum Mol Genet 3: 1383–1386
Tyler-Smith C, Brown WRA (1987) Structure of the major block of alphoid satellite DNA on the human Y chromosome. J Mol Biol 195: 457–470
Tyler-Smith C, Willard HF (1993) Mammalian chromosome structure. Curr Opin Genet Dev 3: 390–397
Tyler-Smith C, Oakey RJ, Larin Z, Fisher RB, Crocker M, Affara NA, Ferguson-Smith MA, Muenke M, Zuffardi O, Jobling MA (1993) Localization of DNA sequences required for human centromere function through an analysis of rearranged Y chromosomes. Nat Genet 5: 368–375
Vergnaud G, Page DC, Simmler MC, Brown L, Rouyer F, Noel B, Botstein D, de la Chapelle A, Weissenbach J (1986) A deletion map of the human Y chromosome based on DNA hybridization. Am J Hum Genet 38: 109–124
Voullaire LE, Slater HR, Petrovic V, Choo KHA (1993) A functional marker centromere with no detectable alpha-satellite, satellite III, or CENP-B protein: activation of a latent centromere? Am J Hum Genet 52: 1153–1163
Yoda K, Kitagawa K, Masumoto H, Muro Y, Okazaki T (1992) A human centromere protein, CENP-B, has a DNA binding domain containing four potential α helices at the NH2 terminus, which is separable from dimerizing activity. J Cell Biol 119: 1413–1427
Author information
Authors and Affiliations
Additional information
Edited by: H. F. Willard
Rights and permissions
About this article
Cite this article
Taylor, S.S., Larin, Z. & Tyler-Smith, C. Analysis of extrachromosomal structures containing human centromeric alphoid satellite DNA sequences in mouse cells. Chromosoma 105, 70–81 (1996). https://doi.org/10.1007/BF02509516
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02509516