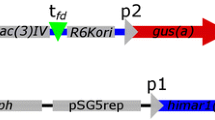
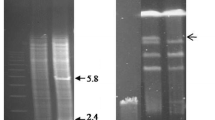
Abstract
The entire biosynthetic pathway for the synthesis of the antibiotic pigment violacein is encoded on a 14.5 kilobase (kb) fragment of theChromobacterium violaceum genome. When cloned intoEscherichia coli, pigment synthesis is strongly expressed, as it is in a wide range of Gram-negative bacteria. Transposon mutagenesis resulted in a number of different phenotypes that correlated with transposon insertion sites in the gene cluster.
Similar content being viewed by others
Literature Cited
Ballantine JA, Beer RJS, Crutchley DJ, Dodd GM, Palmer DR (1958) The synthesis of violacein and related compounds. Proc Chem Soc 232–233
Bolivar F, Rodriguez RL, Greene PJ, Betlach MC, Heynecker HL, Boyer HW, Crossa JH, Falkow S (1977) Construction and characterization of new cloning vehicles. II. A multipurpose cloning system. Gene 2:95–113
Chung CT, Niemela SL, Miller RH (1989) One-step preparation of competentEscherichia coli: transformation and storage of bacterial cells in the same solution. Proc Natl Acad Sci USA 86:2172–2175
De Ley J, Segers P, Gillis M (1978) Intra and intergeneric similarities ofChromobacterium andJanthinobacterium ribosomal ribonucleic acid cistrons. Int J Syst Bacteriol 28:154–168
DeMoss RD, Evans NR (1959) Physiological aspects of violacein biosynthesis in nonproliferating cells. J Bacteriol 78:583–588
DeMoss RD (1967) Violacein. In: Gottlieb D, Shaw P (eds) Mechanisms of action and biosynthesis of antibiotics, Vol 2. New York: Springer-Verlag, pp 77–81
DeMoss RD, Evans NR (1960) Incorporation of14C-labelled substrates into violacein. J Bacteriol 79:729–733
Dewhirst FE, Paster BJ, Bright PL (1989)Chromobacterium, Eikenella, Kingella, Neisseria, Simonsiella, andVitreoscilla species comprise a major branch of the beta group Proteobacteria by 16S ribosomal ribonucleic acid sequence comparison: transfer ofEikenella andSimonsiella to the familyNeisseriaceae (emend.). Int J Syst Bacteriol 39:258–266
Duran N, Campos V, Riveros R, Joyas A, Pereira MF, Haun M (1989) Bacterial chemistry: III. Preliminary studies on trypanosomal activities ofChromobacterium violaceum products. An Acad Bras Cienc 61:31–36
Hansen JB, Olsen RH (1978) Isolation of large bacterial plasmids and characterization of the P2 incompatibility group plasmids pMG1 and pMG5. J Bacteriol 135:227–238
Hoshino T, Ogasawara N (1990) Biosynthesis of violacein: evidence for the intermediacy of 5-hydroxy-l-tryptophan and the structure of a new pigment, oxyviolacein, produced by the metabolism of 5-hydroxytryptophan. Agric Biol Chem 54:2339–2347
Hoshino T, Kondo T, Uchiyama T, Ogasawara N (1987) Biosynthesis of violacein: a novel rearrangement in tryptophan metabolism with a 1,2-shift of the indole ring. Agric Biol Chem 51:965–968
Hoshino T, Yakano T, Hori S, Ogasawara N (1987) Biosynthesis of violacein: origins of hydrogen, nitrogen and oxygen atoms in the 2-pyrrolidone nucleus. Agric Biol Chem 51:2733–2741
Karn J, Brenner S, Barret L, Cesarini G (1980) Novel bacteriophage cloning vector. Proc Natl Acad Sci USA 77:5172–5176
Lichstein HC, van de Sand VF (1945) Violacein, an antibiotic pigment produced byChromobacterium violaceum. J Infect Dis 76:47–51
McCarthy SA, Sakata T, Kakimoto D, Johnson RM (1985) Production and isolation of purple pigment byAlteromonas luteoviolacea. Bull Jpn Soc Sci Fish 51:479–484
Mitoma C, Weissbach H, Udenfriend S (1956) 5-Hydroxytryptophan formation and tryptophan metabolism inChromobacterium violaceum. Arch Biochem Biophys 63:122–130
Moss MO, Ryall C (1981) The genusChromobacterium. In: Starr MP, Stolp H, Trüper HG, Balows A, Schlegel HG (eds) The prokaryotes, a handbook on habitats, isolation and identification of bacteria. Berlin: Springer-Verlag, pp 1355–1364
Moss MO, Ryall C, Logan NA (1978) The classification and characterization of chromobacteria from a lowland river. J Gen Microbiol 105:11–21
Parker WL, Rathnum ML, Johnson JH, Wells JS, Principe PA, Sykes RB (1988) Aerocyanidin: a new antibiotic produced byChromobacterium violaceum. J Antibiot 41:454–460
Pemberton JM, Harding CM (1987) Expression ofRhodopseudomonas sphaeroides carotenoid photopigment genes in phylogenetically related nonphotosynthetic bacteria. Curr Microbiol 15:67–71
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor, New York: Cold Spring Harbor Laboratory Press
Schmitt R, Altenbuchner J, Wiebauer K, Arnold W, Pühler A, Schoffl F (1981) Basis of transposition and gene amplification by Tn1721, and related tetracycline-resistance transposons. Cold Spring Harbor Symp Quant Biol 45:59–65
Sebek OK, Jager H (1962) Divergent pathways of indole metabolism inChromobacterium violaceum. Nature 196:793–794
Singh PD, Liu W, Gougoutas JZ, Malley MF, Poruban MA, Trejo WH, Wells JS, Sykes RB (1988) Aerocavin, a new antibiotic produced byChromobacterium violaceum. J Antbiot 41:446–453
Sivendra R, Tam SH (1977) Pathogenicity of nonpigmented cultures ofChromobacterium violaceum. J Clin Microbiol 5:514–516
Sneath PHA (1956) Cultural and biochemical characteristics of the genusChromobacterium. J Gen Microbiol 15:70–98
Sneath PHA (1960) A study of the bacterial genusChromobacterium. Iowa State Coll Sci 34:243–500
Sukhodolets VV (1988) Organisation and evolution of the bacterial genome. Microbiol Sci 5:202–206
Talmadge K, Gilbert W (1980) Construction of plasmid vectors with uniquePstI cloning sites in a signal sequence coding region. Gene 12:235–241
Ward JM, Grinsted J (1982) Physical and genetic analysis of the IncW group plasmids R388, Sa and R7K. Plasmid 7:239–250
Ward JM, Grinsted J (1982) Analysis of the IncP-1 group plasmids R906 and R751 and their relationship to RP1. Plasmid 8:244–252
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pemberton, J.M., Vincent, K.M. & Penfold, R.J. Cloning and heterologous expression of the violacein biosynthesis gene cluster fromChromobacterium violaceum . Current Microbiology 22, 355–358 (1991). https://doi.org/10.1007/BF02092154
Issue Date:
DOI: https://doi.org/10.1007/BF02092154