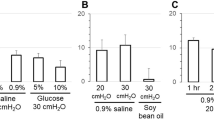
Summary
The mammalian urinary bladder epithelium accommodates volume changes by the insertion and withdrawal of cytoplasmic vesicles. Both apical membrane (which is entirely composed of fused vesicles) and the cytoplasmic vesicles contain three types of ionic conductances, one amiloride sensitive, an-other a cation-selective conductance and the third a cation conductance which seems to partition between the apical membrane and the mucosal solution. The transport properties of the apical membrane (which has been exposed to urine in vivo) differ from the cytoplasmic vesicles by possessing a lower density of amiloride-sensitive channels and a variable level of leak conductance. It was previously shown that glandular kallikrein was able to hydrolyze epithelial sodium channels into the leak conductance and that this leak conductance was further degraded into a channel which partitioned between the apical membrane and the mucosal solution. This report investigates whether kallikrein is the only urinary constituent capable of altering the apical membrane ionic permeability or whether other proteases or ionic conditions also irreversible modify apical membrane permeability.
Alterations of mucosal pH, urea concentrations, calcium concentrations or osmolarity did not irreversible affect the apical membrane ionic conductances. However, urokinase and plasmin (both serine proteases found in mammalian urine) were found to cause an irreversible loss of amiloride-sensitive current, a variable change in the leak current as well as the appearance of a third conductance which was unstable in the apical membrane and appears to partition between the apical membrane and the mucosal solution. Amiloride protects the amiloride-sensitive conductance from hydrolysis but does not protect the leak pathway. Neither channel is protected by sodium. Fluctuation analysis demonstrated that the loss of amiloride-sensitive current was due to a decrease in the sodium-channel density and not a change in the single-channel current. Assuming a simple model of sequential degradation, estimates of single-channel currents and conductances for both the leak channel and unstable leak channel are determined.
Similar content being viewed by others
References
Brown, K.M., Dennis, J.E. 1972. Derivative free analogues of the Levenberg-Marquardt algorithms for non-linear least squares approximation.Numer. Math. 18:289–297
Christensen, O., Bindslev, N. 1982. Fluctuation analysis of short-circuit current in a warm-blooded sodium-retaining epithelium: Site, current density and interaction with triamterene.J. Membrane Biol. 65:19–30
Fuch, W.E., Hviid Larsen, E., Lindemann, B. 1977. Current-voltage curve of sodium channels and concentration dependence of sodium permeability in frog skin.J. Physiol. 267:137–166
Garty, H., Edelman, I.S. 1983. Amiloride-sensitive trypsinization of apical sodium channels.J. Gen. Physiol. 81:785–803
Holmberg, L., Astedt, B. 1982. Radioimmunoassays of high and low molecular weight urokinase.In: Urokinase: Basic and Clinical Aspects, P.M. Mannuccii and A. D'Angelo, editors, Academic, London
Lewis, S.A. 1977. A reinvestigation of the function of the mamma lian urinary bladder.Am. J. Physiol. 232:F187-F195
Lewis, S.A., Alles, W.P. 1986. Urinary kallikrein: A physiological regulator of epithelial Na+ absorption.Proc. Natl. Acad. Sci. USA 83:5345–5348
Lewis, S.A., de Moura, J.L.C. 1982. Incorporation of cytoplasmic vesicles into apical membrane of mammalian urinary bladder epithelium.Nature 297:685–688
Lewis, S.A., de Moura, J.L.C. 1984. Apical membrane area of rabbit urinary bladder increases by fusion of intracellular vesicles: An electrophysiological study.J. Membrane Biol. 82:123–136
Lewis, S.A., Diamond, J.M. 1976. Na+ transport by rabbit urinary bladder, a tight epithelium.J. Membrane Biol. 28:1–40
Lewis, S.A., Ifshin, M.S., Loo, D.D.F., Diamond, J.M. 1984. Studies of sodium channels in rabbit urinary bladder by noise analysis.J. Membrane Biol. 80:135–151
Lewis, S.A., Wills, N.K. 1983. Apical membrane permeability and kinetic properties of the sodium pump in rabbit urinary bladder.J. Physiol. 341:169–184
Li, J.H.-Y., Palmer, L.G., Edelman, I.S., Lindemann, B. 1982. The role of sodium-channel density in the natriferic response of the toad urinary bladder to an antidiuretic hormone.J. Membrane Biol. 64:77–89
Lindemann, B., Van Driessche, W. 1977. Sodium-specific membrane channels of frog skin are pores: Current fluctuations reveal high turnover.Science 195:292–294
Loo, D.D.F., Lewis, S.A., Ifshin, M.S., Diamond, J.M. 1983. Turnover, membrane insertion and degradation of sodium channels in rabbit urinary bladder.Science 221:1288–1290
Rabiner, L.R., Schafer, R.W., Dlugos, D. 1979. Periodogram method for power spectral estimation.In Programs for digital signal processing. pp. 2.1—1—2.1—10. IEEE, New York
Sansom, S.C., O'Neil, R.G. 1985. Mineralocorticoid regulation of apical membrane Na+ and K+ transport of the cortical collecting duct.Am. J. Physiol. 248:F858-F868
Scott, W.N., Goodman, D.B.P. (editors), 1981. Hormonal regulation of epithelial transport of ions and water.Ann. N. Y. Acad. Sci. 372
Wills, N.K., Lewis, S.A. 1980. Intracellular Na+ activity as a function of Na+ transport rate across a tight epithelium.Biophys. J. 30:181–186
Wills, N.K., Lewis, S.A., Eaton, D.C. 1979. Active and Passive properties of rabbit descending colon: A microelectrode and nystatin study.J. Membrane Biol. 45:81–108
Zweifach, A., Lewis, S.A. 1988. Characterization of a partially degraded Na+ channel from urinary tract epithelium.J. Membrane Biol. 101:49–56
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lewis, S.A., Clausen, C. Urinary proteases degrade epithelial sodium channels. J. Membrain Biol. 122, 77–88 (1991). https://doi.org/10.1007/BF01872741
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01872741