Abstract
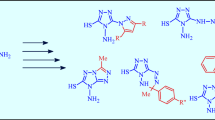
Reaction of 6-nitro-7-oxo-4,7-dihydroazolo[5,1-c][1,2,4]triazines with methyl iodide or dimethyl sulfate affords the N-methyl derivatives, no O-alkylation products being found. The effect of the alkylating agent, solvent, and the azole moiety on the course of the reaction has been examined.
Similar content being viewed by others
Literature Cited
T. L. Pilicheva, V. L. Rusinov, O. N. Chupakhin, N. A. Klyuev, G. G. Aleksandrov, and S. E. Esipov, Khim. Geterotsikl. Soedin., No. 11, 1544 (1986).
V. L. Rusinov, A. Yu. Petrov, and I. Ya. Postovskii, Khim. Geterotsikl. Soedin., No. 9, 1283 (1980).
P. Dea and R. K. Robins, in: Chemistry and Biology of Nucleosides and Nucleotides, R. E. Harmon (ed.), Academic Press, San Diego (1978), p. 301.
H. Gunther, Nuclear Magnetic Resonance Spectroscopy: An Introduction, Wiley, W. Sussex, (1983).
E. Breitnaier and W. Voelter, 13C NMR Spectroscopy, Verlag Chemie, Weinheim-New York (1978), p. 216.
V. L. Rusinov, L. G. Egorova, and A. Yu. Petrov, Khim. Geterotsikl. Soedin., No. 5, 697 (1984).
A. F. Pozharskii, Theoretical Principles of the Chemistry of Heterocycles [in Russian], Khimiya, Moscow (1985), p. 144.
Author information
Authors and Affiliations
Additional information
For Communication 6, see [1].
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 11, pp. 1543–1550, November, 1987.
Rights and permissions
About this article
Cite this article
Ulomskii, E.N., Rusinov, V.L., Chupakhin, O.N. et al. Nitroazines. 7. Alkylation of 6-nitro-7-oxo-4,7-dihydroazolo[5,1-c][1,2,4]-triazines and identification of the products. Chem Heterocycl Compd 23, 1236–1243 (1987). https://doi.org/10.1007/BF00479378
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00479378