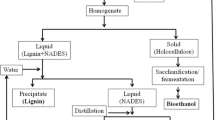
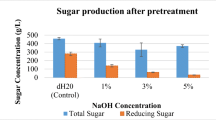
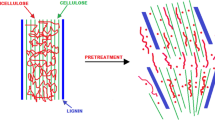
Summary
Enzymatic conversion of lignocellulosic material to fuels and chemicals depends on a initial pretreatment to render the cellulose more susceptible to enzymatic attack. Biological delignification of aspenwood with the fungus Phlebia tremellosus was compared to steaming as a pretreatment method.
The biologically delignified aspenwood (BDA) had a high pentosan content and did not contain inhibitors of enzymatic hydrolysis or subsequent fermentation. In contrast, the steamed aspenwood required a water extraction step to remove the inhibitory material and this step also removed most of the pentosan. The yield of treated material was 90% from biological delignification and 70% from steaming.
The cellulose in the BDA was less accessible to the cellulase enzymes than the steamed aspenwood. Combined hydrolysis and fermentation with Saccharomyces cerevisiae gave a lower yield of ethanol from BDA than from the steamed aspenwood, but the yields based on the weight of substrate before pretreatment were comparable. Combined hydrolysis and fermentation with Klebsiella pneumoniae gave higher yields of butanediol from BDA than from steamed aspenwood, because Klebsiella can ferment the xylose which was present in the biologically treated aspenwood. Trichoderma harzianum produced lower levels of cellulase enzymes when grown on BDA than when grown on steamed aspenwood and this was related to the xylan found in the biologically treated material.
Article PDF
Similar content being viewed by others
Abbreviations
- BDA:
-
biologically delignified aspenwood
- SEA-WI:
-
steam-exploded, water-extracted aspenwood
- AI-SEA-WI:
-
acid-impregnated, steam-exploded, water-extracted aspenwood
- CHF:
-
combined hydrolysis and fermentation
- FP:
-
filter paper
References
Ackman RS (1972) Porous polymer bead packings and formic acid vapour in the GLC of volatile free fatty acids. J Chromatogr Sci 10:560–565
Blanchette RA, Reid ID (1986) Ultrastructural aspects of wood delignification by Phlebia (Merulins) tremellosus. Appl Environm Microbiol 52:239–245
Dale BE (1985) Cellulose pretreatments: technology and techniques. Ann Rept Ferm Proc 8:299–323
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356
Eriksson K-E (1982) A Panoramic paper — advances in enzymatic degradation of lignocellulosic materials. In: Duckworth HE (ed) Ethanol from Biomass. Proc Roy Soc Can Symp, Winnipeg, pp 345–370
Hatakka AI (1983) Pretreatment of wheat straw by white-rot fungi for enzymatic saccharification of cellulose. Eur J Appl Microbiol Biotechnol 18:350–357
Jeffries TW (1985) Emerging technology for fermenting d-xylose. Trends in Biotechnology 3:208–212
Koehler LH (1952) Differentiation of carbohydrates by ananthrone reaction rate and color intensity. Anal Chem 24:1576–1579
Linden BE, Dale JC (1984) Fermentation substrates and economics. Ann Rep Ferm Proc 7:107–134
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurements with the Folin phenol reagent. J Biol Chem 193:265–275
Mandels M, Andreotti RE, Roche C (1976) Measurement of saccharifying cellulase. Biotechnol Bioeng Symp 6:21–33
Mes-Hartree M, Saddler JN (1983) The nature of inhibitory materials present in pretreated lignocellulosic substrates which inhibit the enzymatic hydrolysis of cellulose. Biotechnol Lett 5:531–536
Miller GL (1959) Use of dinitrosalicylic reagent for the determination of reducing sugars. Anal Chem 31:426–442
Nakasone KK, Burdsall HH (1984) Merulius, a synonym of Phlebia. Mycotaxon 21:241–246
Parekh SR, Yu S, Wayman M (1986) Use of recycled Candida shehatae and Pichia stipitis for enhanced ethanol production. Appl Microbiol Biotechnol (in press)
Raabo E, Terkildsen TC (1960) On the determination of blood glucose. Scand J Clin Lab Invest 12:402–406
Reid ID (1985) Biological delignification of aspenwood by solid-state fermentation with the fungus Merulius tremellosus. Appl Environm Microbiol 50:133–139
Reid ID, Abrams GD, Pepper JM (1982) Water-soluble products from the degradation of aspen lignin by Phanerochaete chrysosporium. Can J Bot 60:2357–2364
Reid ID, Seifert KA (1982) Effect of an atmosphere of oxygen on growth, respiration, and lignin degradation by white-rot fungi. Can J Bot 60:252–260
Saddler JN (1982) Screening of highly cellulolytic fungi and the action of their cellulase enzyme systems. Enzyme Microbial Technol 4:414–418
Saddler JN, Brownell HH (1982) Pretreatment of wood cellulosics to enhance enzymatic hydrolysis to glucose. Proceedings of the International Symposium on Ethanol from Biomass. Winnipeg, Canada
Saddler JN, Brownell HH, Clermont LP, Levitin N (1982) Enzymatic hydrolysis of cellulose and various pretreated wood fractions. Biotechnol Bioeng 24:1389–1402
Saddler JN, Hogan CM, Louis-Seize G (1985) A comparison between the cellulase systems of Trichoderma harzianum E58 and Trichoderma reesei C30. Appl Microbiol Biotechnol 22:139–145
Yu EKC, Saddler JN (1982) Enhanced production of 2,3-butanediol by Klebsiella pneumoniae grown on high sugar concentrations in the presence of acetic acid. Appl Environm Microbiol 44:777–784
Yu EKC, Deschatelets L, Saddler JN (1984) The combined enzymatic hydrolysis and fermentation of hemicellulose to 2,3-butanediol. Appl Microbiol Biotechnol 19:365–372
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mes-Hartree, M., Yu, E.K.C., Reid, I.D. et al. Suitability of aspenwood biologically delignified with Pheblia tremellosus for fermentation to ethanol or butanediol. Appl Microbiol Biotechnol 26, 120–125 (1987). https://doi.org/10.1007/BF00253894
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00253894