Summary
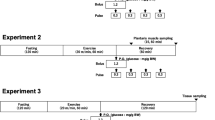
In the rat, muscle glycogen is mobilized during the first stage of exercise, despite normoglycaemia. The aim of the present study was to examine if this process could be prevented or reduced by hyperglycaemia. Three experiments were carried out: in the first, rats were forced to run on a treadmill; in the second the gastrocnemius muscle group was made to contract by stimulation of the sciatic nerve and in the third adrenaline was administered subcutaneously. Each group was divided into two subgroups: control and enriched with glucose (hyperglycaemic). It was shown that hyperglycaemia has no effect on running-induced glycogen mobilization in hind-limb muscles of different fibre composition but prevented it totally in diaphragm muscle. Hyperglycaemia also did not affect the glycogen mobilization induced by stimulation of the sciatic nerve. However, it delayed and reduced markedly the glycogenolytic effect of adrenaline. It is concluded that increased glycogenolysis in muscles at the beginning of exercise may be a consequence of a delay in the activation of glucose transporting mechanisms in muscle cells.
Similar content being viewed by others
References
Addington EE, Grunewald KK (1987) Preexercise feedings of glucose, fructose or sucrose: effects on substrate depletion in rats. J Nutr 117:593–597
Armstrong RB, Ianuzzo CD (1977) Exercise-induced muscle glycogen depletion and repletion in diabetic rats. Life Sci 20:301–308
Armstrong RB, Laughlin H (1984) Exercise blood flow patterns within and among rat muscles after training. Am J Physiol 246 (Heart Circ Physiol 15):H59-H68
Armstrong RB, Saubert IV CW, Sembrowich WL, Shepherd RE, Gollnick PD (1974) Glycogen depletion in rat skeletal muscle fibers at different intensities and durations of exercise. Pflü-gers Arch 352:243–256
Baldwin KM, Reitman JS, Terjung RL, Winder WW, Holloszy JO (1973) Substrate depletion in different types of muscle and in liver during prolonged running. Am J Physiol 225:1045–1050
Carroll NV, Longley RM, Roe JH (1956) The determination of glycogen in liver and muscle by use of anthrone reagent. J Biol Chem 220:583–593
Chasiotis D (1983) The regulation of glycogen phosphorylase and glycogen breakdown in human skeletal muscle. Acta Physiol Scand [Suppl] 518:1–68
Chasiotis D, Hultman E (1985) The effect of adrenalin infusion on the regulation of glycogenolysis in human muscle during isometric contraction. Acta Physiol Scand 123:55–60
Chasiotis D, Sahlin K, Hultman E (1982) Regulation of glycogenolysis in human muscle at rest and during exercise. J Appl Physiol 53:708–715
Chasiotis D, Sahlin K, Hultman E (1983) Regulation of glycogenolysis in human muscle in response to epinephrine infusion. J Appl Physiol 54:45–50
Cohen P (1981) The role of calmodulin troponin, and cyclic AMP in the regulation of glycogen metabolism in mammalian skeletal muscle. Adv Cyclic Nucleotide Res 14:345–359
Conlee RK, McLane JA, Rennie MJ, Winder WW, Holloszy JO (1979) Reversal of phosphorylase activation in muscle despite continued contractile activity. Am J Physiol 237 (Regulatory Integrative Comp Physiol 6):R281-R296
Drummond GJ (1971) Microenvironment and enzyme function: control of energy metabolism during muscle work. Am Zool 11:83–97
Erickson MA, Schwarzkopf RJ, Mackenzie RD (1987) Effect of caffeine, fructose, and glucose ingestion on muscle glycogen utilization during exercise. Med Sci Sports Exerc 19:579–583
Fielding RA, Costill DL, Fink WJ, King DS, Kovaleski JE, Kinwan JP (1987) Effect of pre-exercise carbohydrate feedings on muscle glycogen use during exercise in well-trained runners. Eur J Appl Physiol 56:225–229
Galbo H (1983) Hormonal and metabolic adaptation to exercise. Thieme-Stratton, Stuttgart
Górski J (1978) Exercise-induced changes of reactivity of different types of muscle on glycogenolytic effect of adrenaline. Pflügers Arch 373:1–7
Górski J, Namiot Z, Giedrojć J (1978) Effect of exercise on metabolism of glycogen and triglycerides in the respiratory muscles. Pflügers Arch 377:251–254
Górski J, Namiot Z, Krotkiewski M, Falkowska K (1984) In vivo effect of insulin on the glycogen content in different skeletal muscles of the rat. Horm Metab Res 16:680–680
Górski J, Hood DA, Terjung RL (1986) Blood flow distribution in tissues of perfused rat hindlimb preparations. Am J Physiol 250 (Endocrinol Metab 13):E441-E448
Hamilton N, Noble EG, Ianuzzo CD (1984) Glycogen repletion in different skeletal muscles from diabetic rats. Am J Physiol 247 (Endocrinol Metab 10):E740-E746
Hargreaves M, Briggs CA (1988) Effect of carbohydrate ingestion on exercise metabolism. J Appl Physiol 65:1553–1555
Holmes PA, Mansour TE (1968) Glucose as a regulator of glycogen phosphorylase in rat diaphragm. I. Effect of lucose and related compounds on phosphorylase and glycogen levels. Biochim Biophys Acta 156:266–274
Hutber CA, Bonen A (1989) Glycogenesis in muscle and liver during exercise. J Appl Physiol 66:2811–2817
Hultman E (1959) Rapid specific method for determination of aldosaccharides in body fluids. Nature 183:108–109
James DE, Kraegen EW, Chisholm DJ (1985) Muscle glucose metabolism in exercising rats: comparison with insulin stimulation. Am J Physiol 248 (Endocrinol Metab 11):E575-E580
Koivisto VA, Härkönen M, Karonen SL, Groop PH, Elovainio R, Ferrannini E, Sacca L, DeFronzo RA (1985) Glycogen depletion during prolonged exercise: influence of glucose, fructose, or placebo. J Appl Physiol 58:731–737
Kuipers H, Costill DL, Porter DA, Fink WJ, Morse WM (1986) Glucose feeding and exercise in trained rats: mechanisms for glycogen sparing. J Appl Physiol 61:859–863
Noakes TD, Lambert EV, Lambert MI, McArthur PS, Myburgh KH, Benade JS (1988) Carbohydrate ingestion and muscle glycogen depletion during marathon and ultramarathon racing. Eur J Appl Physiol 57:482–489
Rennie MJ, Fell RD, Ivy JL, Holloszy JO (1982) Adrenaline reaction of muscle phosphorylase after deactivation during phasic contractile activity. Biosci Rep 2:323–331
Roch-Norlund AE (1972) Muscle glycogen and glycogen synthase in diabetic man. Scand J Clin Lab Invest 29 [Suppl 125]:1–27
Saltin B, Gollnick PD (1988) Fuel for muscular exercise. Role of carbohydrate. In: Horton ES, Terjung RL (eds) Exercise, nutrition and energy metabolism. Macmillan, New York, pp 45–71
Tan MH, Bonen A, Watson-Wright W, Hood D, Supper M, Currie D, Belcastro AN, Pierce G (1984) Muscle glycogen repletion after exercise in trained normal and diabetic rats. J Appl Physiol 57:1404–1408
Wallberg-Henriksson H (1987) Glucose transport into skeletal muscle. Influence of contractile activity, insulin, catecholamines and diabetes mellitus. Acta Physiol Scand [Suppl] 564:180
Ziel FH, Wenkatesan N, Davidson MB (1988) Glucose transport is rate limiting for skeletal muscle glucose metabolism in normal and STZ-induced diabetic rats. Diabetes 37:885–890
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Górski, J., Żendzian-Piotrowska, M., Górska, M. et al. Effect of hyperglycaemia on muscle glycogen mobilization during muscle contractions in the rat. Europ. J. Appl. Physiol. 61, 408–412 (1990). https://doi.org/10.1007/BF00236060
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00236060