Abstract
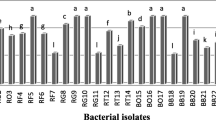
Rhizosphere bacteria were isolated from roots of young and mature plants with known antagonism to phytopathogenic nematodes, including velvet bean (Mucuna deeringiana), castor bean (Ricinus communis), sword bean (Cannavalia ensiformis), and Abruzzi rye (Secale cereale). Isolates from antagonistic plants were compared to soybean isolates for the frequency of antagonism to the root-knot (Meloidogyne incognita) and soybean cyst (Heterodera schachtii) nematodes in a disease assay with soybean. Bacterial isolates were identified using fatty acid analysis, and isolates which exhibited a significant reduction in incidence of soybean damage from both nematodes were characterized physiologically. The bacterial taxa associated with antagonistic plants were markedly different from soybean bacteria. Isolates from soybean were predominantly Bacillus spp., while those from antagonistic plants included more coryneform and Gram-negative genera. Pseudomonas cepacia and Pseudomonas gladioli were predominant among Gram-negative bacteria on antagonistic plants but were not isolated from soybean. Four to six times the number of bacteria from antagonistic plants, compared to soybean, significantly reduced disease incidence of both nematodes. No single pattern of physiological reactions was common among all these bacteria, suggesting that multiple mechanisms accounted for the observed biological control. The results suggest that rhizospheres of antagonistic plants may be useful sources of potential biological control agents for phytopathogenic nematodes.
Similar content being viewed by others
References
Alam M M Siddiqui M A and Ahmad A 1990 Antagonistic plants. In Nematode Bio-Control: Aspects and Prospects. Eds. M S Jairajpuri, M M Alam and I Ahmad. pp 41–50. CBS Publishers & Distributors, Delhi, India.
Becker J O, Savaleta-Mejia E, Colbert S F, Schroth M N, Weinhold A R, Hancock J G and VanGundy S D 1988 Effects of rhizobacteria on root-knot nematodes and gall formation. Phytopathology 78, 1466–1469.
DeBoer S H and Sasser M 1986 Differentiation of Erwinia carotovora spp. carotovora and E. carotovora spp. atroseptica on the basis of fatty acid composition. Can J. Microbiol. 3, 796–800.
deFreitas J R and Germida J J 1990 A root tissue culture system to study winter wheat-rhizobacteria interactions. Appl. Microbiol. Biotechnol. 33, 589–595.
Dropkin V H 1989 Introduction to Plant Nematology. Wiley, New York, 304 p.
Gitaitis R D and Beaver R W 1990 Characterization of fatty acid methyl ester content of Clavibacter michiganensis subsp. michiganensis. Phytopathology 80, 318–321.
Grandados-Alvarez N 1989 La rotación con leguminosas como alternativa para reducir el dano causado por fitopatogenos del suelo y elevar la producción del agrosistema maiz en el tropico humedo. M.S. Thesis. Colegio de Postgraduados, Montecillo, Mexico.
Hagedorn C, Gould W D, Bardinelli R and Dustavson D R 1987 A selective medium for enumeration and recovery of Pseudomonas cepacia biotypes from soil. Appl. Environ. Microbiol. 53, 2265–2268.
Hildebrand D C, Schroth M N and Sands D C 1988 Pseudomonas. In Laboratory Guide for Identification of Plant Pathogenic Bacteria, 2nd Edition. Ed. N W Schaad. pp 60–80. APS Press, St. Paul, MN.
Jantzen E and Hofstad T 1985 Fatty acid composition of oral isolates of Selenomonas. J. Gen. Microbiol. 131, 175–179.
Jatala P 1986 Biological control of plant-parasitic nematodes. Annu. Rev. Phytopathol. 24, 453–89.
Kawamoto S O and Lorbeer J W 1976 Protection of onion seedlings from Fusarium oxyporum f. sp. cepae by seed and soil infestation with Pseudomonas cepacia. Plant Dis. Reptr. 60, 180–191.
Kloepper J W 1991 Plant growth-promoting rhizobacteria as biological control agents. In Soil Microbial Technologies. Ed. B Metting. Marcel Dekker, New York. (In press).
Kloepper J W, Rodríguez-Kábana R, McInroy J A and Collins D J 1991 Analysis of populations and physiological characterization of microorganisms in rhizospheres of plants with antagonistic properties to phytopathogenic nematodes. Plant and Soil 136, 95–102.
Kloepper J W, Zablotowicz R M, Tipping E M and Lifshitz R 1990 Plant growth-promotion mediated by bacterial rhizosphere colonizers. In The Rhizosphere and Plant Growth. Eds. D L Keister and P B Cregan. pp 315–326. Kluwer Academic Publishers, Dordrecht, The Netherlands.
Lambert B, Leyns F, vanRooyen L, Gosselé F, Papon Y and Swings J 1987 Rhizobacteria of maize and their antifungal activities. Appl. Environ. Microbiol. 53, 1866–1871.
Lear B 1959 Application of castor ponace and cropping of castor beans to soil to reduce nematode populations. Plant Dis. Reptr. 43, 459–460.
Moss C W, Shinoda T and Samuels J W 1982 Determination of cellular fatty acid compositions of various yeasts by gas-liquid chromatography. J. Clin. Microbiol. 6, 1073–1079.
Norris J R, Bekeley R C W, Logan N A and O'Donnell A G 1981 The genera Bacillus and Sporolactobacillus. In The Prokaryotes, Vol. 2. Eds. M P Starr, H Stolp, H G Trüper, A Balows and H G Schlegel. pp 1711–1742. Springer-Verlag, Berlin.
Oostendorp M and Sikora R A 1989 Seed treatment with antagonistic rhizobacteria for the suppression of Heterodera schachtii early root infection of sugar beet. Revue Nématol. 12, 77–83.
Palleroni N H 1984 Pseudomonadaceae. In Bergey's Manual of Systematic Bacteriology, Vol. 1. Eds. N R Krieg and J G Holt. pp 141–219. Williams and Wilkins. Baltimore, MD.
Palleroni N J, Kunisawa R, Contopoulou R and Doudoroff M 1973. Nucleic acid homologies in the genus Pseudomonas. Int. J. Syst. Bacteriol. 23, 333–339.
Pedersen J F and Rodríguez-Kábana R 1991 Winter grass cover crop effects on nematodes and yields of double cropped soybean. Plant and Soil 131, 287–291.
Rasoamananjara R, Peladen F, Turlot J C, Monteil J and Richard C 1986 Characterization of Flavobacterium species by analysis of volatile fatty acid production, J. Gen. Microbiol. 132, 2723–2732.
Rich J R, Rahi G S, Opperman C H and Davis E L 1989 Influence of the castor bean (Ricinus communis) on motility of Meloidogyne incognita. Nematropica 19, 99–103.
Rizzo A F, Korkeala H and Mononen I 1987 Gas chromatography analysis of cellular fatty acids and neutral monosacchrides in the identification of lactobacilli. Appl. Environ. Microbiol. 53, 2883–2888.
Rodríguez-Kábana R, King P S, Robertson D G and Weaver C F 1988 Potential of crops uncommon to Alabama for management of root-knot and soybean cyst nematodes. Ann. Appl. Nematol. 2, 116–120.
Rodríguez-Kábana R, King P S and Weaver C F 1990 Potential of some tropical and subtropical legumes for the management of soybean (Gycine max) nematodes. (Abstr.) Nematropica 20, 17.
Rodríguez-Kábana R and Morgan-Jones G 1987 Biological control of nematodes: Soil amendments and microbial antagonists. Plant and Soil 100, 237–247.
Rodríguez-Kábana R and Pope M H 1981 A simple incubation method for the extraction of nematodes from soil. Nematropcia 11, 175–186.
Sayre R M and Starr M P 1988 Bacterial disease and antagonism of nematodes. In Diseases of Nematodes, Vol. 1. Eds. G O Poinar and H-B Jansson. pp 69–101. CRC Press, Boca Raton FL.
Schroth M N and Hancock J G 1982 Disease-suppressive soil and root-colonizing bacteria. Science. 216, 1376–1381.
Vincente N E and Acosta N 1987 Effects of Mucuna deeringiana on Meloidogyne incognita. Nematropica 17, 99–102.
Zeck W M 1971 A rating scheme for field evaluation of root-knot nematode infestations. Pflanzenschutz-Nac. 24, 141–144.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kloepper, J.W., Rodríguez-Kábana, R., McInroy, J.A. et al. Rhizosphere bacteria antagonistic to soybean cyst (Heterodera glycines) and root-knot (Meloidogyne incognita) nematodes: Identification by fatty acid analysis and frequency of biological control activity. Plant Soil 139, 75–84 (1992). https://doi.org/10.1007/BF00012844
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00012844