Abstract
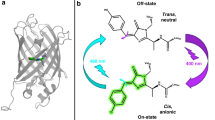
Several studies have led to the conclusion that, in the green fluorescent protein (GFP) of the jellyfish Aequorea victoria, a photoconversion involving excited-state proton transfer occurs from an A- to a B-form, while an intermediate I-form was held responsible for the green fluorescence. Here we have identified the I-form of wild-type GFP in absorption, located the 0-0 transitions of all three forms A, B and I, and determined vibrational frequencies of the ground and excited states. The intrinsically narrow 0-0 transitions are revealed by the wavelengths at which holes can be burnt. The pathways of photointerconversion are unraveled by excitation, emission and hole-burning spectroscopy. We present an energy-level scheme that has significant implications for GFP-mutants, which likewise can occur in the three photointerconvertible forms.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Chalfie, M., Tu, Y., Euskirchen, G., Ward, W.W. & Prasher, D.C. Science 263, 802– 805 (1994).
Prasher, D.C., Eckenrode, V.K., Ward, W.W., Prendergast, F.G. & Cormier, M.J. Gene 111, 229–233 (1992).
Tsien, R.Y. Annu. Rev. Biochem. 67, 509–544 (1998).
Misteli, T. & Spector, D.L. Nature Biotechnol. 15, 961–964 (1997).
Yang, F., Moss, L.G. & Philips, G.N. Jr. Nature Biotechnol. 14, 1246–1251 (1996).
Ormö, M. et al. Science 273, 1392– 1395 (1996).
Heim, R., Prasher, D.C. & Tsien R.Y. Proc. Natl. Acad. Sci. USA 91, 12501–12504 (1994).
Cubitt, A.B. et al. Trends Biochem. Sci. 20, 448– 455 (1995).
Niwa, H. et al. Proc. Natl. Acad. Sci. USA 93, 13617– 13622 (1996).
Chattoraj, M., King, B.A., Bublitz, G.U. & Boxer, S.G. Proc. Natl. Acad. Sci. USA 93, 8362– 8367 (1996).
Lossau, H. et al. Chem. Phys. 213, 1– 16 (1996).
Ward, W.W., Cody, C.W., Hart, R.C. & Cormier, M.J. Photochem. Photobiol. 31, 611–615 (1980).
Brejc, K. et al. Proc. Natl. Acad. Sci. USA 94, 2306– 2311 (1997).
Palm, G.J. et al. Nature Struct. Biol. 4, 361– 365 (1997).
Voityuk, A.A., Michel-Beyerle, M.E. & Rösch, N. Chem. Phys. Lett. 272, 162–167 (1997).
Heim, R. & Tsien, R.Y. Curr. Biol. 6, 178–182 (1996).
Völker, S. Annu. Rev. Phys. Chem. 40, 499–530 (1989).
Dickson, R.M., Cubitt, A.B., Tsien, R.Y. & Moerner, W.E. Nature 388, 355–358 (1997).
Acknowledgements
We thank J.H. van der Waals for critical remarks and helpful suggestions. The high-resolution spectroscopy experiments, carried out in Leiden, were financially supported by the Netherlands Foundation for Physical Research (FOM) and the Council for Chemical Research of the Netherlands Organization for Scientific Research (CW-NWO). V.S is the recipient of a Human Frontiers Science Program long-term fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Creemers, T., Lock, A., Subramaniam, V. et al. Three photoconvertible forms of green fluorescent protein identified by spectral hole-burning. Nat Struct Mol Biol 6, 557–560 (1999). https://doi.org/10.1038/9335
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/9335
This article is cited by
-
Cathodoluminescence of green fluorescent protein exhibits the redshifted spectrum and the robustness
Scientific Reports (2020)
-
Characterization of Split Fluorescent Protein Variants and Quantitative Analyses of Their Self-Assembly Process
Scientific Reports (2018)
-
Stochastic dynamic study of optical transition properties of single GFP-like molecules
Journal of Biological Physics (2016)
-
Light driven ultrafast electron transfer in oxidative redding of Green Fluorescent Proteins
Scientific Reports (2013)
-
Thermal Effect on Aequorea Green Fluorescent Protein Anionic and Neutral Chromophore Forms Fluorescence
Journal of Fluorescence (2012)