Abstract
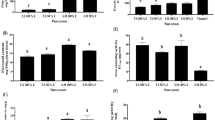
Colorectal cancer is one of the leading causes of death worldwide, and healthy diet is critical in preventing it. This study intends to investigate phenolic profile, antioxidant, and anti-proliferative activity as well as Glutathione S-transferase (GST) inhibition activity of water extract of Salvia fruticosa Mill. (SF.) HPLC analysis revealed that the main bioactive ingredients of SF were rosmarinic and caffeic acid. Total phenolic and flavonoid contents were 425.1 mg GAE/g extract and 94.7 mg CE/g extract, respectively. SF exhibited high antioxidant activity through DPPH and ABTS assays. The extract exhibited time and dose-dependent anti-proliferative activity on colorectal adenocarcinoma cell lines (Caco-2 and HT-29). The extract also inhibited GST activity in a mixed-type manner, with an IC50 value of 6.8 µg/mL. To conclude, the results suggest that SF may be considered as a medicinal plant which can be used in theprevention and treatment of colon cancer, besides chemotherapy drugs.




Similar content being viewed by others
References
G. Ku, I.B. Tan, T. Yau, N. Boku, S. Laohavinij, A.L. Cheng, Y.K. Kang, G. de, Lima lopes, management of colon cancer: resource-stratified guidelines from the Asian oncology summit 2012. Lancet Oncol. 13, 470–481 (2012)
A.A. Plumb, S. Halligan, Colorectal cancer screening. Semin. Roentgenol. 50, 101–110 (2015)
M.T. Ayseli, Y. Ipek Ayseli, Flavors of the future: Health benefits of flavor precursors and volatile compounds in plant foods. Trends Food Sci. Technol. 48, 69–77 (2016)
D.X. Yang, C.P. Gross, P.R. Soulos, J.B. Yu, Estimating the magnitude of colorectal cancers prevented during the era of screening 1976 to 2009. Cancer. 120, 2893–2901 (2014)
S. Enayat, M. Ceyhan, A.A. Başaran, M. Gürsel, S. Banerjee, Anticarcinogenic effects of the ethanolic extract of salix aegyptiaca in colon cancer cells: Involvement of Akt/PKB and MAPK pathways. Nutr. Cancer. 65, 1045–1058 (2013)
V. Papageorgiou, C. Gardeli, A. Mallouchos, M. Papaioannou, M. Komaitis, Variation of the chemical profile and antioxidant behavior of Rosmarinus officinalis L. and Salvia fruticosa Miller Grown in Greece. J. Agric. Food Chem. Agric. Food Chem. 56, 7254–7264 (2008)
B. Gürdal, Ş Kültür, An ethnobotanical study of medicinal plants in Marmaris (Muǧla, Turkey). J. Ethnopharmacol. 146, 113–126 (2013)
S. Akbulut, M.M. Bayramoglu, The trade and use of some medical and aromatic herbs in Turkey. Stud. Ethno Med. 7, 67–77 (2013)
F.S. Şenol, I. Orhan, F. Celep, A. Kahraman, M. Doǧan, G. Yilmaz, B. Şener, Survey of 55 Turkish Salvia taxa for their acetylcholinesterase inhibitory and antioxidant activities. Food Chem. 120, 34–43 (2010)
Species, RP-HPLC/MS/MS analysis of the phenolic compounds, antioxidant and antimicrobial activities of Salvia L. Antioxidants. 5, 1–15 (2016)
M. Adams, F. Gmünder, M. Hamburger, Plants traditionally used in age related brain disorders-A survey of ethnobotanical literature. J. Ethnopharmacol. 113, 363–381 (2007)
M. Marouf, M. Batal, S. Moledor, S.N. Talhouk, Exploring the practice of traditional wild plant collection in lebanon. Food, Cult. Soc. 18, 355–378 (2015)
M. Er, O. Tugay, M.M. Özcan, D. Ulukuş, F. Al-Juhaimi, Biochemical properties of some Salvia L. species. Environ. Monit. Assess. 185, 5193–5198 (2013)
A. Al-Aboudi, F.U. Afifi, Plants used for the treatment of diabetes in Jordan: a review of scientific evidence. Pharm. Biol. 49, 221–239 (2011)
J. Dai, R.J. Mumper, Plant phenolics: extraction, analysis and their antioxidant and anticancer properties. Molecules 15, 7313–7352 (2010)
L. Bravo, Polyphenols: Chemistry, dietary sources, metabolism, and nutritional significance. Nutr. Rev. 56, 317–333 (2009)
C. Dincer, A. Topuz, H. Sahin-Nadeem, K.S. Ozdemir, I.B. Cam, I. Tontul, R.S. Gokturk, S.T. Ay, A comparative study on phenolic composition, antioxidant activity and essential oil content of wild and cultivated sage (Salvia fruticosa Miller) as influenced by storage. Ind. Crops Prod. 39, 170–176 (2012)
E.C. Quadrangularis, A. Marmelos, R. Prema, D. Sekaran, S. Sekar, K. Bannoth, C. Sekhar, In vitro Antioxidant Study On Combined Plants. Int. J. Pharm. Toxicol. 3, 9–13 (2013)
M.F. Azevedo, C.F. Lima, M. Fernandes-Ferreira, M.J. Almeida, J.M. Wilson, C. Pereira-Wilson, Rosmarinic acid, major phenolic constituent of Greek sage herbal tea, modulates rat intestinal SGLT1 levels with effects on blood glucose. Mol. Nutr. Food Res. 55, 15–25 (2011)
A. Altay, F. Bozoğlu, Salvia fruticosa Modulates mRNA expressions and activity levels of xenobiotic metabolizing CYP1A2, CYP2E1, NQO1, GPx, and GST enzymes in human colorectal adenocarcinoma HT-29 Cells. Nutr. Cancer. 69, 1–12 (2017)
M. Kartal-Yandim, A. Adan-Gokbulut, Y. Baran, Molecular mechanisms of drug resistance and its reversal in cancer. Crit. Rev. Biotechnol. 8551, 1–11 (2015)
V.L. Singleton, J.A. Rossi, Colorimetry of total phenolics with phosphomolybdic phosphotungstic acid reagents. Am. J. Enol. Vitic. 16, 144–158 (1965)
J. Zhishen, T. Mengcheng, W. Jianming, The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem. 64, 555–559 (1999)
B. Li, C. Zhang, L. Peng, Z. Liang, X. Yan, Y. Zhu, Y. Liu, Comparison of essential oil composition and phenolic acid content of selected Salvia species measured by GC-MS and HPLC methods, Ind. Crops Prod. 69, 329–334 (2015)
M.S. Blois, Antioxidant determinations by the use of a stable free radical. Nature. 181, 1199–1200 (1958)
R. Re, N. Pellegrini, A. Proteggente, A. Pannala, M. Yang, C. Evans, Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 26, 1231–1237 (1999)
N. Çoruh, A.G. Sağdiçoğlu Celep, F. Özgökçe, Antioxidant properties of Prangos ferulacea (L.) Lindl., Chaerophyllum macropodum Boiss. and Heracleum persicum Desf. from Apiaceae family used as food in Eastern Anatolia and their inhibitory effects on glutathione-S-transferase. Food Chem. 100, 1237–1242 (2007)
W.H. Habig, M.J. Pabst, W.B. Jakoby, Glutathione Stransferase: the first enzymatic step in mercapturic acid formation. J. Biol. Chem. 249, 7130–7139 (1974)
O. Lowry, N.J. Rosebrough, A.L. Farr, R.J. Randall, Protein measurement with the folin phenol reagent. J. Biol. Chem. 193, 265–275 (1951)
T. Mosmann, Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J. Immunol. Methods. 65, 55–63 (1983)
A. Altay, G.S. Celep, A.E. Yaprak, I. Baskose, F. Bozoglu, Glassworts as possible anticancer agents against human colorectal adenocarcinoma cells with their nutritive, antioxidant and phytochemical profiles, Chem. Biodivers. 14 (2017)
M.S. Kocak, C. Sarikurkcu, M. Cengiz, S. Kocak, M.C. Uren, B. Tepe, Salvia cadmica: Phenolic composition and biological activity. Ind. Crops Prod. 85, 204–212 (2016)
S. Krimat, T. Dob, M. Toumi, A. Kesouri, A. Noasri, Assessment of phytochemicals, antioxidant, antimicrobial and cytotoxic properties of Salvia chudaei Batt. et Trab. endemic medicinal plant from Algeria. J. Mater. Environ. Sci. 6, 70–78 (2015)
O. Firuzi, R. Miri, M. Asadollahi, S. Eslami, A.R. Jassbi, Cytotoxic, antioxidant and antimicrobial activities and phenolic contents of eleven Salvia species from Iran. Iran. J. Pharm. Res. 12, 801–810 (2013)
M. Kostić, B. Zlatković, B. Miladinović, S. Živanović, T. Mihajilov-Krstev, D. Pavlović, D. Kitić, Rosmarinic acid levels, phenolic contents, antioxidant and antimicrobial activities of the extracts from Salvia verbenaca L. obtained with different solvents and procedures. J. Food Biochem. 39, 199–208 (2015)
O. Martínez-Cruz, O. Paredes-López, Phytochemical profile and nutraceutical potential of chia seeds (Salvia hispanica L.) by ultra high performance liquid chromatography. J. Chromatogr. A. 1346, 43–48 (2014)
C. Proestos, M. Komaitis, Analysis of naturally occurring phenolic compounds in aromatic plants by RP-HPLC coupled to diode array detector (DAD) and GC-MS after silylation. Foods. 2, 90–99 (2013)
P. Siddhuraju, A. Abirami, G. Nagarani, M. Sangeethapriya, Antioxidant capacity and total phenolic content of aqueous acetone and ethanol extract of edible parts of Moringa oleifera and Sesbania grandiflora. Int. J. Biol. Biomol. Agric. Food Biotechnol. Eng. 8, 1090–1098 (2014)
V. Farzaneh, I.S. Carvalho, A review of the health benefit potentials of herbal plant infusions and their mechanism of actions. Ind. Crops Prod. 65, 247–258 (2015)
M.R. Loizzo, M. Abouali, P. Salehi, A. Sonboli, M. Kanani, F. Menichini, R. Tundis, In vitro antioxidant and antiproliferative activities of nine Salvia species. Nat. Prod. Res. 28, 2278–2285 (2014)
R. Stocker, Antioxidant defenses in human blood plasma and extra-cellular fluids. Arch. Biochem. Biophys. 595, 136–139 (2016)
H. Göcer, A. Akıncıoğlu, S. Göksu, İ Gülçin, Carbonic anhydrase inhibitory properties of phenolic sulfonamides derived from dopamine related compounds. Arab. J. Chem. 10, 398–402 (2017)
O. Tusevski, A. Kostovska, A. Iloska, L. Trajkovska, S. Simic, Phenolic production and antioxidant properties of some Macedonian medicinal plants, Open Life Sci. 9 (2014)
S. Dudonné, X. Vitrac, P. Coutiére, M. Woillez, J.M. Mérillon, Comparative study of antioxidant properties and total phenolic content of 30 plant extracts of industrial interest using DPPH, ABTS, FRAP, SOD, and ORAC assays. J. Agric. Food Chem. 57, 1768–1774 (2009)
F. Bouaziz, M. Koubaa, F. Barba, S. Roohinejad, S. Chaabouni, Antioxidant properties of water-soluble gum from flaxseed hulls. Antioxidants. 5, 2–10 (2016)
N. Das, D. Goshwami, S. Hasan, Z. Al Mahmud, S.Z. Raihan, Evaluation of antioxidant, antimicrobial and cytotoxic activities of Terminalia citrina Leaves. J. Pharm. Res. 10 (2016) 8–15
S. Sabbobeh, R.; Hejaz, H.; Jahajha, A.; Al-Akhras, S.; Al-Jaas, H.; Abu-Lafi, Antioxidant an antimicrobial activities of the leaf extract of Salvia palaestina. J. Appl. Pharm. Sci. 6, 76–82 (2016)
G. Şeker Karatoprak, F. Göger, M.B. Yerer, M. Koşar, Chemical composition and biological investigation of Pelargonium endlicherianum root extracts. Pharm. Biol. 55, 1608–1618 (2017)
E. Arinç, D. Yilmaz, A. Bozcaarmutlu, mechanism of inhibition of CYP1A1 and glutathione S-transferase activities in Fish liver by quercetin, mechanism of inhibition of CYP1A1 and glutathione S-transferase activities in fish liver by quercetin. Nutr. Cancer (2014) 37–41
W. Harshbarger, S. Gondi, S.B. Ficarro, J. Hunter, D. Udayakumar, D. Gurbani, W.D. Singer, Y. Liu, L. Li, J.A. Marto, K.D. Westover, Structural and biochemical analyses reveal the mechanism of glutathione S-transferase Pi 1 inhibition by the anti-cancer compound piperlongumine. J. Biol. Chem. 292, 112–120 (2017)
R. Zhuo, K.M. Kosak, S. Sankar, E.T. Wiles, Y. Sun, J. Zhang, J. Ayello, G.D. Prestwich, P.J. Shami, M.S. Cairo, S.L. Lessnick, W. Luo, Targeting Glutathione S-transferase M4 in Ewing sarcoma. Front. Pediatr. 2, 83 (2014)
H.J. and P. DJ, The glutathione S-transferase supergene family: regulation of GST and the contribution of the isoenzymes to cancer chemoprotection and drug resistance, Crit. Rev. Biochem. Mol. 30 (1995) 445–600
Z. Wang, Z. Zhao, M.M. Abou-Zaid, J.T. Arnason, R. Liu, B. Walshe-Roussel, A. Waye, S. Liu, A. Saleem, L.A. Cáceres, Q. Wei, I.M. Scott, Inhibition Of insect glutathione S-transferase (GST) By Conifer Extracts, Arch. Insect Biochem. Physiol. 87, 234–249 (2014)
N. Çoruh, A.G. Saǧdiçoǧlu Celep, F. Özgökçe, M. Işcan, Antioxidant capacities of Gundelia tournefortii L. extracts and inhibition on glutathione-S-transferase activity. Food Chem 100, 1249–1253 (2007)
K. Zhang, N.P. Das, Inhibitory effects of plant polyphenols on rat liver glutathione S-transferases. Biochem. Pharmacol. 47, 2063–2068 (1994)
M. Canivenc-Lavier, M. Vernevaut, M. Totis, M. Siess, J. Magdalou, Comparative effects of flavonoids and model inducers on drugmetabolizing enzymes in rat liver. Toxicology. 114, 19–27 (1996)
P. Apáti, P.J. Houghton, G. Kite, G.B. Steventon, A. Kéry, In-vitro effect of flavonoids from Solidago canadensis extract on glutathione S-transferase. J. Pharm. Pharmacol. 58, 251–256 (2006)
M. Sengottuvelan, R. Senthilkumar, N. Nalini, Modulatory influence of dietary resveratrol during different phases of 1,2-dimethylhydrazine induced mucosal lipid-peroxidation, antioxidant status and aberrant crypt foci development in rat colon carcinogenesis, Biochim. Biophys. Acta Gen. Subj. 1760, 1175–1183 (2006)
S. Soltanian, M. Sheikhbahaei, N. Mohamadi, Cytotoxicity evaluation of methanol extracts of some medicinal plants on P19 embryonal carcinoma cells, 7 (2017) 142–149
B.R.T. Galani, M.E. Sahuc, G. Sass, F.N. Njayou, C. Loscher, P. Mkounga, G. Deloison, P. Brodin, Y. Rouillé, G. Tiegs, K. Séron, P.F. Moundipa, Khaya grandifoliola C.DC: a potential source of active ingredients against hepatitis C virus in vitro. Arch. Virol. 161, 1169–1181 (2016)
D.F.N. Pedro, A.A. Ramos, C.F. Lima, F. Baltazar, C. Pereira-Wilson, Colon Cancer Chemoprevention by Sage Tea Drinking: Decreased DNA Damage and Cell Proliferation. Phyther. Res. 30, 298–305 (2016)
C.P.R. Xavier, C.F. Lima, M. Fernandes-Ferreira, C. Pereira-Wilson, Salvia fruticosa, Salvia officinalis, and rosmarinic acid induce apoptosis and inhibit proliferation of human colorectal cell lines: the role in MAPK/ERK pathway. Nutr. Cancer. 61, 564–571 (2009)
K.R. Lawson, N.A. Ignatenko, G.A. Piazza, H. Cui, E.W. Gerner, Influence of K-ras activation on the survival responses of Caco-2 cells to the chemopreventive agents sulindac and difluoromethylornithine, Cancer Epidemiol. Biomark. Prev. 9, 1155–1162 (2000)
I. Dairaku, Y. Han, N. Yanaka, N. Kato, Inhibitory effect of curcumin on IMP dehydrogenase, the target for anticancer and antiviral chemotherapy agents. Biosci. Biotechnol. Biochem. 74, 185–187 (2010)
R. Zhou, M.G. Vander Heiden, C.M. Rudin, Genotoxic exposure is associated with alterations in glucose uptake and metabolism genotoxic exposure is associated with alterations in glucose uptake and metabolism. Cancer Res. 62, 3515–3520 (2002)
R.J. Gillies, R.A. Gatenby, Adaptive landscapes and emergent phenotypes: why do cancers have high glycolysis? J. Bioenerg. Biomembr. 39, 251–257 (2007)
A. Ramos, D. Pedro, A.R. Collins, C. Pereira-Wilson, Protection by Salvia extracts against oxidative and alkylation damage to DNA in human HCT15 and CO115 cells. J. Toxicol. Environ. Health. A. 75, 765–775 (2012)
R. Tundis, D. Iacopetta, M.S. Sinicropi, M. Bonesi, M. Leporini, N.G. Passalacqua, J. Ceramella, F. Menichini, M.R. Loizzo, Assessment of antioxidant, antitumor and pro-apoptotic effects of Salvia fruticosa Mill. subsp. thomasii (Lacaita) Brullo, Guglielmo, Pavone & Terrasi (Lamiaceae). Food Chem. Toxicol. 106, 155–164 (2017)
S.B. Hani, M. Bayachou, Salvia fruticosa reduces intrinsic cellular and H2O2-induced DNA oxidation in HEK 293 cells; assessment using flow cytometry, Asian Pac. J. Trop. Biomed. 4, 399–403 (2014)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflıct of ınterest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Altay, A., Kılıc Suloglu, A., Sagdıcoglu Celep, G. et al. Anatolıan sage Salvıa frutıcosa ınhıbıts cytosolıc glutathıone-s-transferase actıvıty and colon cancer cell prolıferatıon. Food Measure 13, 1390–1399 (2019). https://doi.org/10.1007/s11694-019-00055-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11694-019-00055-1