Abstract
Purpose
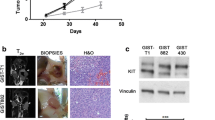
Preclinical imaging offers a useful tool for monitoring cancer biological behavior and therapy in vivo without the necessity of animal surgery. The following paper describes our examination of tumor progress and anti-angiogenic therapy with Bevacizumab on colon cancer subtypes (SW480 and SW620) by using different non-invasive real-time in vivo imaging techniques.
Procedures
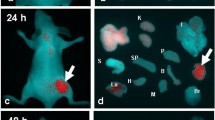
Color Doppler ultrasound imaging (CDUI) was used to observe the formation of new blood vessels; a homemade fluorescence reflectance imaging (FRI) apparatus was mainly used to test the difference in VEGFR2 expression between the tumor subtypes. Briefly, 15 Balb/c nude mice bearing subcutaneous SW480 and SW620 xenografts were randomly divided into Control and Drug groups. Bevacizumab treatment lasted for 3 weeks. All images were captured pre- and post-treatment. At the end of experiment, all mice were euthanized, and tumor tissue was collected and analyzed by immunohistochemical staining.
Results
Expression of VEGFR2 was found to be slightly (10 %) but significantly higher for the SW620 cells than for SW480 cells. In addition, SW620 has shown to be more vascularized than SW480 subtype. After 3-week Bevacizumab therapy, no blood vessels were found within 83 % of SW620, while it was 67 % in SW480; the increase of SW620 tumor volume post-treatment was only 3.17-fold compared with the tumor volume pre-treatment, and 4.51-fold higher in SW480.
Conclusion
Our data suggest that SW480 and SW620 cell lines respond differently to Bevacizumab therapy in vivo. Because of higher vascularization, and subsequently higher reduction by drug of new blood vessels and tumor growth rate, xenografts derived from the metastatic SW620 cell line have a better chance of being successfully treated with Bevacizumab compared with those derived from the primary tumor SW480 cell line.





Similar content being viewed by others
Abbreviations
- CDUI:
-
Color Doppler ultrasound imaging
- FRI:
-
Fluorescence reflectance imaging
- NBV:
-
New blood vessels
- CRC:
-
Colorectal cancer
- IHC:
-
Immunohistochemistry
References
Leibovitz A, Stinson JC, McCombs WB et al (1976) Classification of human colorectal adenocarcinoma cell lines. Cancer Res 36:4562–4569
Hewitt R, McMarlin A, Kleiner D et al (2000) Validation of a model of colon cancer progression. J Pathol 192:446–454
Wang H, Jinming Z, Jiahe T et al (2009) Using dual-tracer PET to predict the biologic behavior of human colorectal cancer. J Nucl Med 50:1857–1864
Sukhdeo K, Paranmban RI, Vidal JG et al (2013) Multiplex flow cytometry barcoding and antibody arrays identify surface antigen profiles of primary and metastatic colon cancer cell lines. PLoS One 8:e53015
Mousa L, Salem M, Mikhail S (2015) Biomarkers of angiogenesis in colorectal cancer. Biomarkers in Cancer 7(s1):13–19
Wang H, Liu B, Tian J et al (2010) Monitoring early responses to irradiation with dual-tracer micro-PET in dual-tumor bearing mice. World J Gastroenterol 16(43):5416–5423
Heenan SD (2004) Magnetic resonance imaging in prostate cancer. Nat Publ Group 7:282–288
Cherry S (2006) Multimodality in vivo imaging systems: twice the power or double the trouble? Annu Rev Biomed Eng 8:35–62
Dort M, Rehemtulla A, Ross B (2008) PET and SPECT imaging of tumor biology: new approaches towards oncology drug discovery and development. Curr Comput Aided Drug Des 4(1):46–45
Ji S, Zheng Y, Shao G (2013) Integrin αvβ3–targeted radiotracer 99mTc-3P-RGD2Useful for noninvasive monitoring of breast tumor response to antiangiogenic linifanib therapy but not anti-integrin αvβ3RGD2 therapy. Theranostics 3(11):816–830
Vuletic I, Liu J, Wu H et al (2015) Establishment of an mKate2-expressing cell line for non-invasive real-time breast cancer in vivo imaging. Mol Imaging Biol 17:811–818
Huang D, Lan H, Liu et al (2015) Anti-angiogenesis or pro-angiogenesis for cancer treatment: focus on drug distribution. Int J Clin Exp Med 8(6):8369–8376
Hasan M, Ho S, Owen D et al (2011) Inhibition of VEGF induces cellular senescence in colorectal cancer cells. Int J Cancer 129:2115–2123
Wang Y, Fei D, Vanderlaan M et al (2004) Biological activity of bevacizumab, a humanized anti-VEGF antibody in vitro. Angiogenesis 7:335–345
Kim T, Landen C, Lin Y et al (2009) Combined anti-angiogenic therapy against VEGF and integrin alphaVbeta3 in an orthotopic model of ovarian cancer. Cancer Biol Ther 8(23):2263–2272
Thaker A, Razjouyan F, Woods D et al (2012) Combination therapy of radiofrequency ablation and bevacizumab monitored with power Doppler ultrasound in a murine model of hepatocellular carcinoma. Int J Hyperth 28(8):766–775
Zhu H, Li Z, Mao S et al (2011) Antitumor effect of sFlt-1 gene therapy system mediated by Bifidobacterium Infantis on Lewis lung cancer in mice. Cancer Gene Ther 18:884–896
Roskoski RJ (2008) VEGF receptor protein–tyrosine kinases: structure and regulation. Biochem Biophys Res Commun 375:287–291
Hao Y, Yang J, Yin S et al (2014) The synergistic regulation of VEGF-mediated angiogenesis through miR-190 and target genes. RNA 20(8):1328–1336
Yancopoulos G, Davis S, Gale N et al (2000) Vascular-specific growth factors and blood vessel formation. Nature 407(6801):242–248
Saif MS (2013) Anti-VEGF agents in metastatic colorectal cancer (mCRC): are they all alike? Cancer Manag Res 5:103–111
He K, Cui B, Li G et al (2012) The effect of anti-VEGF drugs (bevacizumab and aflibercept) on the survival of patients with metastatic colorectal cancer (mCRC). OncoTargets and Therapy 5:59–65
Acknowledgements
We acknowledge the financial support by the National Key Instrumentation Development Project (No. 2011YQ030114), the Natural Science Foundation of China (No. 81421004), and the Beijing Natural Science Foundation (No. 7162113). We would like also to thank Prof. Ming Fan and Prof. Lingling Zhu from the Institute of Basic Medical Sciences (Beijing 100850, China) for their assistance and advice regarding our hypoxia experiment.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interests.
Electronic Supplementary Material
.
ESM 1
(PDF 662 kb)
Rights and permissions
About this article
Cite this article
Vuletic, I., Zhou, K., Li, H. et al. Validation of Bevacizumab Therapy Effect on Colon Cancer Subtypes by Using Whole Body Imaging in Mice. Mol Imaging Biol 19, 847–856 (2017). https://doi.org/10.1007/s11307-017-1048-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-017-1048-z