Abstract
Propose
To study CCDC103 expression profiles and understand how pathogenic variants in CCDC103 affect its expression profile at mRNA and protein level.
Methods
To increase the knowledge about the CCDC103, we attempted genotype-phenotype correlations in two patients carrying novel homozygous (missense and frameshift) CCDC103 variants. Whole-exome sequencing, quantitative PCR, Western blot, electron microscopy, immunohistochemistry, immunocytochemistry, and immunogold labelling were performed to characterize CCDC103 expression profiles in reproductive and somatic cells.
Results
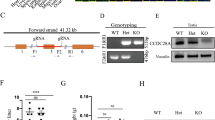
Our data demonstrate that pathogenic variants in CCDC103 gene negatively affect gene and protein expression in both patients who presented absence of DA on their axonemes. Further, we firstly report that CCDC103 is expressed at different levels in reproductive tissues and somatic cells and described that CCDC103 protein forms oligomers with tissue-specific sizes, which suggests that CCDC103 possibly undergoes post-translational modifications. Moreover, we reported that CCDC103 was restricted to the midpiece of sperm and is present at the cytoplasm of the other cells.
Conclusions
Overall, our data support the CCDC103 involvement in PCD and suggest that CCDC103 may have different assemblies and roles in cilia and sperm flagella biology that are still unexplored.











Similar content being viewed by others
References
Fliegauf M, Benzing T, Omran H. When cilia go bad: cilia defects and ciliopathies. Nat Rev Mol Cell Biol. 2007;8:880–93.
Mitchison HM, Valente EM. Motile and non-motile cilia in human pathology: from function to phenotypes. J Pathol. 2017;241(2):294–309.
Fawcett D. Cilia and flagella. In: Fawcett D, editor. The cell: biochemistry, physiology, morphology. 2nd ed. Phyladelphia, USA: WB Saunders company; 1981. p. 575–603.
Pereira R, Sá R, Barros A, Sousa M. Major regulatory mechanisms involved in sperm motility. Asian J Androl. 2017;19(1):5–14.
King SM. The dynein microtubule motor. Biochim Biophys Acta (BBA) - Mol Cell Res. 2000;1496(1):60–75.
Leigh MW, Pittman JE, Carson JL, Ferkol TW, Dell SD, Davis SD, et al. Clinical and genetic aspects of primary ciliary dyskinesia/Kartagener syndrome. Genet Med. 2009;11:473–87.
Jackson CL, Behan L, Collins SA, Goggin PM, Adam EC, Coles JL, et al. Accuracy of diagnostic testing in primary ciliary dyskinesia. Eur Respir J. 2016;47:837–48.
Ishikawa H, Marshall WF. Ciliogenesis: building the cell’s antenna. Nat Rev Mol Cell Biol. 2011;12:222–34.
Pereira R, Oliveira J, Sousa M. A molecular approach to sperm immotility in humans: a review. Med Reprod Embriol Clín. 2014;01(1):15–25.
Kurkowiak M, Ziętkiewicz E, Witt M. Recent advances in primary ciliary dyskinesia genetics. J Med Genet. 2015;52:1–9.
Pereira R, Oliveira J, Ferraz L, Barros A, Santos R, Sousa M. Mutation analysis in patients with total sperm immotility. J Assist Reprod Genet. 2015;32(6):893–902.
Panizzi JR, Becker-heck A, Castleman VH, Al-mutairi D, Liu Y, Loges NT, et al. CCDC103 mutations cause primary ciliary dyskinesia by disrupting assembly of ciliary dynein arms. Nat Genet. 2012;44:714–9.
King SM, Patel-King RS. The oligomeric outer dynein arm assembly factor CCDC103 is tightly integrated within the ciliary axoneme and exhibits periodic binding to microtubules. J Biol Chem. 2015;290:7388–401.
Miller SA, Dykes DD, Polesky HF. A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res. 1988;16:1215.
Oliveira J, Negrao L, Fineza I, Taipa R, Melo-Pires M, Fortuna AM, et al. New splicing mutation in the choline kinase beta (CHKB) gene causing a muscular dystrophy detected by whole-exome sequencing. J Hum Genet. 2015;60:305–12.
Rutland J, Dewar A, Cox T, Cole P. Nasal brushing for the study of ciliary ultrastructure. J Clin Pathol. 1982;35:357–9.
Bernardino RL, Alves MG, Oliveira PF. Establishment of primary culture of Sertoli cells. In: Alves MG, Oliveira PF, editors. Sertoli Cells. New York, NY: Springer, Humana Press; 2018. p. 1–8.
Afzelius BA, Srurgess JM. The immotile-cilia syndrome: a microtubule-associated defect. Crit Rev Biochem Mol Biol. 1985;19:63–87.
Sousa M, Oliveira E, Alves Â, Gouveia M, Figueiredo H, Ferraz L, et al. Ultrastructural analysis of five patients with total sperm immotility. Zygote. 2015;23(6):1–8.
De Iongh R, Rutland J. Orientation of respiratory tract cilia in patients with primary ciliary dyskinesia, bronchiectasis, and in normal subjects. J Clin Pathol. 1989;42:613–9.
Pfaffl MW. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001;29:2002–7.
Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 2015;17:405–23.
Betts MJ, Russell RB. Amino acid properties and consequences of substitutions. In: Barnes MR, Gray IC, editors. Bioinformatics for Geneticists. New York: John Wiley & Sons, Ltd; 2003. p. 289–316.
Venselaar H, Te Beek T A, Kuipers RK, Hekkelman ML, Vriend G. Protein structure analysis of mutations causing inheritable diseases. An e-Science approach with life scientist friendly interfaces. BMC Bioinforma. 2010;11:548.
Choksi SP, Lauter G, Swoboda P, Roy S. Switching on cilia: transcriptional networks regulating ciliogenesis. Development. 2014;141(7):1427–41.
Maurizy C, Quinternet M, Abel Y, Verheggen C, Santo PE, Bourguet M, et al. The RPAP3-Cterminal domain identifies R2TP-like quaternary chaperones. Nat Commun. 2018;9(1):2093.
Wayne N, Mishra P, Bolon DN. Hsp90 and client protein maturation. In: Calderwood SK, Prince TL, editors. Molecular chaperones: methods and protocols. Totowa, NJ: Humana Press; 2011. p. 33–44.
Mcmanus CJ, Graveley BR, Treisman J, Richter J. RNA structure and the mechanisms of alternative splicing. Curr Opin Genet Dev. 2011;21:373–9.
Thomas J, Morlé L, Soulavie F, Laurençon A, Sagnol S, Durand B. Transcriptional control of genes involved in ciliogenesis: a first step in making cilia. Biol Cell. 2010;102(9):499–513.
Geremek M, Ziętkiewicz E, Bruinenberg M, Franke L, Pogorzelski A, Wijmenga C, et al. Ciliary genes are down-regulated in bronchial tissue of primary ciliary dyskinesia patients. PLoS One. 2014;9(2):e88216.
Lewis BP, Burge CB, Bartel DP. Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA Targets. Cell. 2005;120(1):15–20.
Catalanotto C, Cogoni C, Zardo G. MicroRNA in control of gene expression: an overview of nuclear functions. Int J Mol Sci. 2016;17(10):1712.
Marcet B, Chevalier B, Luxardi G, Coraux C, Zaragosi L-E, Cibois M, et al. Control of vertebrate multiciliogenesis by miR-449 through direct repression of the Delta/Notch pathway. Nat Cell Biol. 2011;13:693–9.
Song R, Walentek P, Sponer N, Klimke A, Lee JS, Dixon G, et al. miR-34/449 miRNAs are required for motile ciliogenesis by repressing cp110. Nature. 2014;510(7503):115–20.
King SM. Axonemal dynein arms. Cold Spring Harb Perspect Biol. 2016;8(11):a028100.
Inaba K. Sperm flagella: comparative and phylogenetic perspectives of protein components Unicellular algae Chlamydomonas. Mol Hum Reprod. 2011;17(8):524–38.
Neely MD, Boekelheide K. Sertoli cell processes have axoplasmic features: an ordered microtubule distribution and an abundant high molecular weight microtubule-associated protein (cytoplasmic dynein). J Cell Biol. 1988;107(5):1767–76.
Blackburn K, Bustamante-Marin X, Yin W, Goshe MB, Ostrowski LE. Quantitative proteomic analysis of human airway cilia identifies previously uncharacterized proteins of high abundance. J Proteome Res. 2017;16(4):1579–92.
Shoemark A, Moya E, Hirst RA, Patel MP, Robson E, Hayward J, et al. A high prevalence CCDC103 p.His154Pro mutation causing primary ciliary dyskinesia is associated with normal diagnostic investigations. Eur Respir J. 2017;50(suppl 61):PA1851.
Matoulkova E, Michalova E, Vojtesek B, Hrstka R. The role of the 3′ untranslated region in post-transcriptional regulation of protein expression in mammalian cells. RNA Biol. 2012;9(5):563–76.
Sabila M, Kundu N, Deana S, Ullah H. Tyrosine phosphorylation based homo-dimerization of Arabidopsis RACK1A proteins regulates oxidative stress signaling pathways in yeast. Front Plant Sci. 2016;7:176.
Mali GR, Yeyati PL, Mizuno S, Dodd DO, Tennant PA, Keighren MA, et al. ZMYND10 functions in a chaperone relay during axonemal dynein assembly. eLife. 2018;7:e34389.https://doi.org/10.7554/eLife.34389.
Acknowledgments
The authors would like to thank Ana Rita Gonçalves, MSc, Center of Medical Genetics Dr. Jacinto Magalhães (CGMJM-CHUP) for Sanger sequencing samples processing; Ângela Alves, MSc, Institute of Biomedical Sciences Abel Salazar-University of Porto (ICBAS-UP) for technical electron microscopy assistance; Raquel Bernardino, PhD, Tânia Dias, Msc, and Ana Maria, MSc for Sertoli cell culture assistance (ICBAS-UP); Fátima Ferreirinha, MSc (ICBAS-UP) for confocal microscopy assistance; and Rui Fernandes, MSc, Institute of Health Research and Innovation (IBMC/i3S-UP) for immunogold labelling assistance.
Funding
UMIB (Pest-OE/SAU/UI0215/2014) is funded by the National Funds through FCT-Foundation for Science and Technology, and FCT Grant ref.: PD/BD/105767/2014 (R.P.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Ethics approval and consent to participate
The authors declare that they have followed all the rules of ethical conduct regarding originality, data processing and analysis, duplicate publication, and biological material.
Consent for publication
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplemental Video 1
Immunocytochemical detection of CCDC103 from control sperm (SZ) obtained from a confocal Z-stack. CCDC103 is present, with a helix-like shape, at sperm midpiece (green). Sperm flagella stained with axoneme-specific acetylated α-tubulin (red) and nuclei stained with DAPI (blue). (AVI 23 kb)
Supplementary Video 2
(AVI 27 kb)
Supplemental Table S1.
List of primers used in this study. (DOCX 16 kb)
Supplemental File S1.
Immunogold staining (DOCX 1360 kb)
Rights and permissions
About this article
Cite this article
Pereira, R., Oliveira, M.E., Santos, R. et al. Characterization of CCDC103 expression profiles: further insights in primary ciliary dyskinesia and in human reproduction. J Assist Reprod Genet 36, 1683–1700 (2019). https://doi.org/10.1007/s10815-019-01509-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-019-01509-7