Abstract
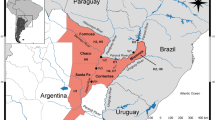
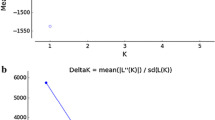
Brycon amazonicus is widely distributed in the Amazon basin. The species has traditionally been the focus of subsistence and commercial fisheries, and recently has become an important aquaculture species. Aquaculture relies on the removal of individuals from nature which form the basis of breeding stocks. The breeding stocks are often derived from local populations, but equally often are a mix of fishes from different regions or from other aquaculture stations. In this study, we found that B. amazonicus forms just one population in the central Amazon basin, and most animals in the aquaculture stations originated from this group. However, fishes from the Balbina aquaculture station represent another biological group, while the fishes in the experimental station of the Federal University of Amazonas are an admixed group. Fishes of the aquaculture stations are differentiated from each other and from the wild fish. Genetic diversity of the aquaculture fishes was not different from the wild fishes, and thus, inbreeding is unlikely to be a concern. Outbreeding depression, however, should be of concern given the observed levels of admixture in the aquaculture stocks. We conclude the article with recommendations for good practices to minimize the likelihood of inbreeding and outbreeding depression.


Similar content being viewed by others
References
Aguiar, J., H. Schneider, F. Gomes, J. Carneiro, S. Santos, L. R. Rodrigues & I. Sampaio, 2013. Genetic variation in native and farmed populations of Tambaqui (Colossoma macropomum) in the Brazilian Amazon: Regional discrepancies in farming systems. Anais da Academia Brasileira de Ciencias 85: 1439–1447.
Amado, M. V., I. P. Farias & T. Hrbek, 2011. A molecular perspective on systematics, taxonomy and classification amazonian discus fishes of the genus Symphysodon. International Journal of Evolutionary Biology 2011: 360654. doi:10.4061/2011/360654.
Ashikaga, F. Y., M. L. Orsi, C. Oliveira, J. A. Senhorini & F. Foresti, 2015. The endangered species Brycon orbignyanus: Genetic analysis and definition of priority areas for conservation. Environmental Biology of Fishes 98: 1845–1855.
Azevedo-Santos, V. M., O. Rigolin-Sá & F. M. Pelicice, 2011. Growing, losing or introducing? Cage aquaculture as a vector for the introduction of non-native fish in Furnas Reservoir, Minas Gerais, Brazil. Neotropical Ichthyology 9: 915–919.
Barroso, R. M., A. W. S. Hilsdorf, H. L. M. Moreira, A. M. Mello, S. E. F. Guimarães, P. H. Cabello & Y. M. Traub-Cseko, 2003. Identification and characterization of microsatellites loci in Brycon opalinus (Cuvier 1819)(Characiforme, Characidae, Bryconiae). Molecular Ecology Notes 3: 297–298.
Barroso, R. M., A. W. S. Hilsdorf, H. L. M. Moreira, P. H. Cabello & Y. M. Traub-Cseko, 2005. Genetic diversity of wild and cultured populations of Brycon opalinus (Cuvier, 1819) (Characiforme, Characidae, Bryconiae) using microsatellites. Aquaculture 247: 51–65.
da Batista, V. S., 1999. Biologia e administração pesqueira de alguns Characiformes explorados na Amazônia Central. Instituto Nacional de Pesquisas da Amazônia (INPA), Manaus.
da Batista, J., S. da & J. A. Alves-Gomes, 2006. Phylogeography of Brachyplatystoma rousseauxii (Siluriformes—Pimelodidae) in the Amazon Basin offers preliminary evidence for the first case of “homing” for an Amazonian migratory catfish. Genetics and Molecular Research 5: 723–740.
Bernardino, R. M., J. A. Senhorini & C. L. Bock, 1993. Propagação artificial do matrinxã Brycon cephalus (Günther, 1869) (Teleostei, Characidae). Boletim Técnico CEPTA 6: 1–9.
Castagnolli, N., 1992. Criação de peixes de água doce. FUNEP, Jaboticabal.
Chistiakov, D. A., B. Hellemans & F. A. M. Volckaert, 2006. Microsatellites and their genomic distribution, evolution, function and applications: A review with special reference to fish genetics. Aquaculture 255: 1–29.
DeWoody, J. A., J. Schupp, L. Kenefic, J. Busch, L. Murfitt & P. Keim, 2004. Universal method for producing ROX-labeled size standards suitable for automated genotyping. Biotechniques 37: 348–352.
Do, C., R. S. Waples, D. Peel, G. M. Macbeth, B. J. Tillett & J. R. Ovenden, 2014. NeEstimator v2: re-implementation of software for the estimation of contemporary effective population size (Ne) from genetic data. Molecular Ecology Resources 14: 209–214. doi:10.1111/1755-0998.12157.
Earl, D. A. & B. M. VonHoldt, 2012. STRUCTURE HARVESTER: a website and program for visualizing STRUCTURE output and implementing the Evanno method. Conservation Genetics Resources 4: 359–361.
Evanno, G., S. Regnaut & J. Goudet, 2005. Detecting the number of clusters of individuals using the software STRUCTURE: A simulation study. Molecular Ecology 14: 2611–2620.
Excoffier, L. & H. E. L. Lischer, 2010. Arlequin suite ver 3.5: A new series of programs to perform population genetics analyses under Linux and Windows. Molecular Ecology Resources 10: 564–567.
Excoffier, L., P. E. Smouse & J. M. Quattro, 1992. Analysis of molecular variance inferred from metric distances among DNA haplotypes: Application to human mitochondrial DNA restriction data. Genetics 131: 479–491.
Farias, I. P., J. P. Torrico, C. García-Dávila, M. D. C. F. Santos, T. Hrbek & J. F. Renno, 2010. Are rapids a barrier for floodplain fishes of the Amazon basin? A demographic study of the keystone floodplain species Colossoma macropomum (Teleostei: Characiformes). Molecular Phylogenetics and Evolution 56: 1129–1135.
Filho, L. C. S. & V. D. S. Batista, 2009. Dinâmica populacional da matrinxã Brycon amazonicus (Characidae) na Amazônia Central. Zoologia (Curitiba) 26: 195–203.
Frankham, R., J. D. Ballou & D. A. Briscoe, 2002. Introduction to Conservation Genetics. Cambridge University Press, Cambridge.
Flowers, J. M., S. C. Schroeter & R. S. Burton, 2002. The recruitment sweepstakes has many winners: Genetic evidence from the sea urchin Strongylocentrotus purpuratus. Evolution 56: 1445–1453.
Frederico, R. G., I. P. Farias, M. L. G. Araújo, P. Charvet-Almeida & J. A. Alves-Gomes, 2012. Phylogeography and conservation genetics of the Amazonian freshwater stingray Paratrygon aiereba Müller & Henle, 1841 (Chondrichthyes: Potamotrygonidae). Neotropical Ichthyology 10: 71–80.
Gilk, S. E., I. A. Wang, C. L. Hoover, W. W. Smoker, S. G. Taylor, A. K. Gray & A. J. Gharrett, 2004. Outbreeding depression in hybrids between spatially separated pink salmon, Oncorhynchus gorbuscha, populations: Marine survival, homing ability, and variability in family size. Environmental Biology of Fishes 69: 287–297.
Gomes, L. C. & J. R. Urbinati, 2005. Criação de matrinxã. In Baldisserotto, B. & L. C. Gomes (eds), Especies nativas para piscicultura no Brasil. Editora UFSM, Santa Maria: 149–174.
Goulding, M., 1980. Fishes and the Forest: Explorations in Amazonian Natural History. University of California press, Los Angeles.
Hoshiba, M. A., 2007. Enriquecimento da alimentação das larvas de Matrinxã (Brycon amazonicus) com aminoácidos. Universidade Estadual Paulista, Jaboticabal, Influência no crescimento inicial e sobrevivência das larvas.
Hrbek, T., I. P. Farias, M. Crossa, I. Sampaio, J. I. R. Porto & A. Meyer, 2005. Population genetic analysis of Arapaima gigas, one of the largest freshwater fishes of the Amazon basin: Implications for its conservation. Animal Conservation 8: 297–308.
Hrbek, T., M. Crossa & I. P. Farias, 2007. Conservation strategies for Arapaima gigas (Schinz, 1822) and the Amazonian várzea ecosystem. Brazilian Journal of Biology 67: 909–917.
Huff, D. D., L. M. Miller, C. J. Chizinski & B. Vondracek, 2011. Mixed-source reintroductions lead to outbreeding depression in second-generation descendents of a native North American fish. Molecular Ecology 20: 4246–4258.
Hurtado-Alarcón, J. C., N. J. Mancera-Rodríguez & C. I. Saldamando-Benjumea, 2011. Genetic variability of Brycon henni (Characiformes: Characidae) in the middle basin of Nare and Guatapé Rivers, Magdalena River system, Colombia. Revista de Biología Tropical 59: 269–282.
Hutchings, J. A. & D. J. Fraser, 2008. The nature of fisheries- and farming-induced evolution. Molecular Ecology 17: 294–313.
Jakobsson, M. & N. A. Rosenberg, 2007. CLUMPP: A cluster matching and permutation program for dealing with label switching and multimodality in analysis of population structure. Bioinformatics 23: 1801–1806.
Junk, W. J., 1997. The Central Amazon System: Ecology of a Pulsing System. Springer, Berlin.
Kalinowski, S. T., 2005. Hp-Rare 1.0: a computer program for performing rarefaction on measures of allelic richness. Molecular Ecology Notes 5: 187–189. doi:10.1111/j.1471-8286.2004.00845.x.
Leberg, P. L., 2002. Estimating allelic richness: Effects of sample size and bottlenecks. Molecular Ecology 11: 2445–2449.
Lima, F. C. T., 2003. Subfamily Bryconinae (Characins, Tetras). In Reis, R. E., S. O. Kullander & C. J. Ferraris (eds), RE Check List of the Freshwater Fishes of South and Central America. EDPURCS, Porto Alegre: 174–181.
Machado, V. N., S. C. Willis, A. S. Teixeira, T. Hrbek & I. P. Farias, 2017. Population genetic structure of the Amazonian black flannelmouth characin (Characiformes, Prochilodontidae: Prochilodus nigricans Spix & Agassiz, 1829): contemporary and historical gene flow of a migratory and abundant fishery species. Environmental Biology of Fishes 100: 1–16. doi:10.1007/s10641-016-0547-0.
Martin, C. W., M. M. Valentine & J. F. Valentine, 2010. Competitive interactions between invasive nile tilapia and native fish: The potential for altered trophic exchange and modification of food webs. PLoS ONE 5: e14395.
Matsumoto, C. K. & A. W. S. Hilsdorf, 2009. Microsatellite variation and population genetic structure of a neotropical endangered Bryconinae species Brycon insignis Steindachner, 1877: Implications for its conservation and sustainable management. Neotropical Ichthyology 7: 395–402.
McClelland, E. K. & K. A. Naish, 2006. What is the fitness outcome of crossing unrelated fish populations? A meta-analysis and an evaluation of future research directions. Conservation Genetics 8: 397–416.
Mennerat, A., F. Nilsen, D. Ebert & A. Skorping, 2010. Intensive farming: Evolutionary implications for parasites and pathogens. Evolutionary Biology 37: 59–67.
Naylor, R. L., R. J. Goldburg, J. H. Primavera, N. Kautsky, M. C. M. Beveridge, J. Clay, C. Folke, J. Lubchenco, H. Mooney & M. Trowll, 2000. Effect of aquaculture on world fish supplies. Nature 405: 1017–1024.
Nei, M., 1987. Molecular Evolutionary Genetics. Columbia University Press, New York, NY, 512 pp.
Nei, M. & A. K. Roychoudhury, 1974. Sampling variances of heterozygosity and genetic distance. Genetics 76: 379–390.
Ortega, J. C. G., H. F. Júlio Jr., L. C. Gomes & A. A. Agostinho, 2014. Fish farming as the main driver of fish introductions in Neotropical reservoirs. Hydrobiologia 746: 147–158. doi:10.1007/s10750-014-2025-z.
Palstra, F. P. & D. J. Fraser, 2012. Effective/census population size ratio estimation: a compendium and appraisal. Ecology and Evolution 2: 2357–2365. doi:10.1002/ece3.329.
Pinto, L. G. Q., 2014. Potential Fish Species in Amazonia for National Aquaculture. The Fish Site: http://www.thefishsite.com/articles/1795/potential-fish-species-in-amazonia-for-national-aquaculture/.
Pritchard, J. K., M. Stephens & P. Donnelly, 2000. Inference of population structure using multilocus genotype data. Genetics 155: 945–959.
Rice, W. R., 1989. Analyzing tables of statistical tests. Evolution 43: 223–225.
Rosenberg, N. A., 2004. DISTRUCT: A program for the graphical display of population structure. Molecular Ecology Notes 4: 137–138.
Sanches, A. & P. M. Galetti Jr., 2006. Microsatellites loci isolated in the freshwater fish Brycon hilarii. Molecular Ecology Notes 6: 1045–1046.
Sanches, A. & P. M. Galetti Jr., 2007. Genetic evidence of population structuring in the Neotropical freshwater fish Brycon hilarii (Valenciennes, 1850). Brazilian Journal of Biology 67: 889–895.
Sanches, A., P. M. Galetti Jr., F. Galzerani, J. Derazo, B. Cutilak Bianchi & T. Hatanaka, 2012. Genetic population structure of two migratory freshwater fish species (Brycon orthotaenia and Prochilodus argenteus) from the São Francisco River in Brazil and its significance for conservation. Latin American Journal of Aquatic Research 40: 177–186.
Santos, M. D. C. F., M. L. Ruffino & I. P. Farias, 2007. High levels of genetic variability and panmixia of the tambaqui Colossoma macropomum (Cuvier, 1818) in the main channel of the Amazon River. Journal of Fish Biology 71A: 33–44.
Schuelke, M., 2000. An economic method for the fluorescent labeling of PCR fragments. Nature Biotechnology 18: 233–234.
Sioli, H., 1967. Studies in Amazonian waters. Atas do Simpósio sobre a Biota Amazônica 3: 9–50.
Sioli, H., 1984. The Amazon and its main affluents: hydrography, morphology of the river courses and river types. Limnology and Landscape Ecology of a Mighty Tropical River and its Basin. In Sioli, H. (ed.), The Amazon. Springer, New York: 127–165.
Smith, N. J. H., 1979. A Pesca no Rio Amazonas. INPA, Manaus.
Templeton, A. R., 1986. Coadaptation and outbreeding depression. In Soulé, M. E. (ed.), Conservation Biology: The Science of Scarsity and Diversity. Sinauer Associates, Sunderland: 105–121.
Wright, S., 1951. The genetical structure of populations. Annals of Eugenics 15: 323–354.
Van Oosterhout, C., W. F. Hutchinson, D. P. M. Wills & P. Shipley, 2004. MICRO-CHECKER: software for identifying and correcting genotyping errors in microsatellite data. Molecular Ecology Notes 4: 535–538.
Waples, R. S. & C. Do, 2008. Ldne: a program for estimating effective population size from data on linkage disequilibrium. Molecular Ecology Resources 8: 753–756. doi:10.1111/j.1755-0998.2007.02061.x.
Yousefian, M. & A. Nejati, 2008. Inbreeding depression by family matching in rainbow trout (Oncorhynchus mykiss). Journal of Fisheries and Aquatic Science 3: 384–391.
Zengeya, T. A., M. P. Robertson, A. J. Booth & C. T. Chimimba, 2013. A qualitative ecological risk assessment of the invasive Nile tilapia, Oreochromis niloticus in a sub-tropical African river system (Limpopo River, South Africa). Aquatic Conservation: Marine and Freshwater Ecosystems 23: 51–64.
Acknowledgements
This research was supported by Grants from FINEP/DARPA (Convênio No. 01.09.0472.00) to IPF and GB. This study formed a portion of ROC PIBIC undergraduate program at UFAM. IPF and TH are supported by Bolsa de Pesquisa scholarship from CNPq, and ROC was supported by a fellowship from FAPEAM.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: Christian Sturmbauer
Electronic supplementary material
Below is the link to the electronic supplementary material.
10750_2017_3278_MOESM1_ESM.eps
Supplementary Figure S1 Schematic illustration of allelic frequencies at 10 studied microsatellite loci. Circles represent distinct alleles, and their areas are directly proportional to their frequencies. Supplementary material 1 (EPS 809 kb)
Rights and permissions
About this article
Cite this article
de Oliveira, R.C., Santos, M.d.F., Bernardino, G. et al. From river to farm: an evaluation of genetic diversity in wild and aquaculture stocks of Brycon amazonicus (Spix & Agassiz, 1829), Characidae, Bryconinae. Hydrobiologia 805, 75–88 (2018). https://doi.org/10.1007/s10750-017-3278-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-017-3278-0