Abstract
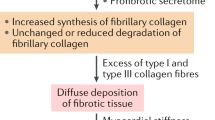
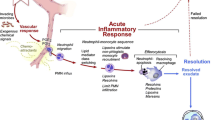
Cardiac fibrosis is associated with non-ischemic dilated cardiomyopathy, increasing its morbidity and mortality. Cardiac fibroblast is the keystone of fibrogenesis, being activated by numerous cellular and humoral factors. Macrophages, CD4+ and CD8+ T cells, mast cells, and endothelial cells stimulate fibrogenesis directly by activating cardiac fibroblasts and indirectly by synthetizing various profibrotic molecules. The synthesis of type 1 and type 3 collagen, fibronectin, and α-smooth muscle actin is rendered by various mechanisms like transforming growth factor-beta/small mothers against decapentaplegic pathway, renin angiotensin system, and estrogens, which in turn alter the extracellular matrix. Investigating the underlying mechanisms will allow the development of diagnostic and prognostic tools and discover novel specific therapies. Serum biomarkers aid in the diagnosis and tracking of cardiac fibrosis progression. The diagnostic gold standard is cardiac magnetic resonance with gadolinium administration that allows quantification of cardiac fibrosis either by late gadolinium enhancement assessment or by T1 mapping. Therefore, the goal is to stop and even reverse cardiac fibrosis by developing specific therapies that directly target fibrogenesis, in addition to the drugs used to treat heart failure. Cardiac resynchronization therapy had shown to revert myocardial remodeling and to reduce cardiac fibrosis. The purpose of this review is to provide an overview of currently available data.



Similar content being viewed by others
References
McKenna WJ, Maron BJ, Thiene G (2017) Classification, epidemiology, and global burden of cardiomyopathies. Circ Res 121:722–730. https://doi.org/10.1161/CIRCRESAHA.117.309711
Pinto YM, Elliott PM, Arbustini E, Adler Y, Anastasakis A, Böhm M, Duboc D, Gimeno J, de Groote P, Imazio M, Heymans S, Klingel K, Komajda M, Limongelli G, Linhart A, Mogensen J, Moon J, Pieper PG, Seferovic PM, Schueler S, Zamorano JL, Caforio ALP, Charron P (2016) Proposal for a revised definition of dilated cardiomyopathy, hypokinetic non-dilated cardiomyopathy, and its implications for clinical practice: a position statement of the ESC working group on myocardial and pericardial diseases. Eur Heart J 37:1850–1858. https://doi.org/10.1093/eurheartj/ehv727
Cecchi F, Tomberli B, Olivotto I (2012) Clinical and molecular classification of cardiomyopathies. Glob Cardiol Sci Pract 2012:4. https://doi.org/10.5339/gcsp.2012.4
Morita H, Seidman J, Seidman CE (2005) Genetic causes of human heart failure. J Clin Invest 115:518–526. https://doi.org/10.1172/JCI200524351
Venero JV, Doyle M, Shah M, Rathi VK, Yamrozik JA, Williams RB, Vido DA, Rayarao G, Benza R, Murali S, Glass J, Olson P, Sokos G, Biederman RWW (2015) Mid wall fibrosis on CMR with late gadolinium enhancement may predict prognosis for LVAD and transplantation risk in patients with newly diagnosed dilated cardiomyopathy-preliminary observations from a high-volume transplant centre. ESC Hear Fail 2:150–159. https://doi.org/10.1002/ehf2.12041
Kong P, Christia P, Frangogiannis NG (2014) The pathogenesis of cardiac fibrosis. Cell Mol Life Sci 71:549–574. https://doi.org/10.1007/s00018-013-1349-6
Ivey MJ, Tallquist MD (2016) Defining the cardiac fibroblast. Circ J 80:2269–2276. https://doi.org/10.1253/circj.CJ-16-1003
Legere SA, Haidl ID, Légaré J-F, Marshall JS (2019) Mast cells in cardiac fibrosis: new insights suggest opportunities for intervention. Front Immunol 10. https://doi.org/10.3389/fimmu.2019.00580
Nevers T, Salvador AM, Velazquez F, Ngwenyama N, Carrillo-Salinas FJ, Aronovitz M, Blanton RM, Alcaide P (2017) Th1 effector T cells selectively orchestrate cardiac fibrosis in nonischemic heart failure. J Exp Med 214:3311–3329. https://doi.org/10.1084/jem.20161791
Khalil H, Kanisicak O, Prasad V, Correll RN, Fu X, Schips T, Vagnozzi RJ, Liu R, Huynh T, Lee S-J, Karch J, Molkentin JD (2017) Fibroblast-specific TGF-β–Smad2/3 signaling underlies cardiac fibrosis. J Clin Invest 127:3770–3783. https://doi.org/10.1172/JCI94753
Ma Z-G, Yuan Y-P, Wu H-M, Zhang X, Tang Q-Z (2018) Cardiac fibrosis: new insights into the pathogenesis. Int J Biol Sci 14:1645–1657. https://doi.org/10.7150/ijbs.28103
Jia G, Aroor AR, Hill MA, Sowers JR (2018) Role of renin-angiotensin-aldosterone system activation in promoting cardiovascular fibrosis and stiffness. Hypertension 72:537–548. https://doi.org/10.1161/HYPERTENSIONAHA.118.11065
Ma F, Li Y, Jia L, Han Y, Cheng J, Li H, Qi Y, Du J (2012) Macrophage-stimulated cardiac fibroblast production of IL-6 is essential for TGF β/Smad activation and cardiac fibrosis induced by angiotensin II. PLoS One 7:e35144. https://doi.org/10.1371/journal.pone.0035144
Medzikovic L, Aryan L, Eghbali M (2019) Connecting sex differences, estrogen signaling, and microRNAs in cardiac fibrosis. J Mol Med. https://doi.org/10.1007/s00109-019-01833-6
Richards AM (2017) Circulating biomarkers of cardiac fibrosis. Circ Heart Fail 10. https://doi.org/10.1161/CIRCHEARTFAILURE.117.003936
Michalska-Kasiczak M, Bielecka-Dabrowa A, von Haehling S, Anker SD, Rysz J, Banach M (2018) Biomarkers, myocardial fibrosis and co-morbidities in heart failure with preserved ejection fraction: an overview. Arch Med Sci 14:890–909. https://doi.org/10.5114/aoms.2018.76279
Becker MAJ, Cornel JH, van de Ven PM, van Rossum AC, Allaart CP, Germans T (2018) The prognostic value of late gadolinium-enhanced cardiac magnetic resonance imaging in nonischemic dilated cardiomyopathy. JACC Cardiovasc Imaging 11:1274–1284. https://doi.org/10.1016/j.jcmg.2018.03.006
Valbuena-López S, Hinojar R, Puntmann VO (2016) Cardiovascular magnetic resonance in cardiology practice: a concise guide to image acquisition and clinical interpretation. Rev Española Cardiol English Ed 69:202–210. https://doi.org/10.1016/j.rec.2015.11.011
Croisille P, Revel D, Saeed M (2006) Contrast agents and cardiac MR imaging of myocardial ischemia: from bench to bedside. Eur Radiol 16:1951–1963. https://doi.org/10.1007/s00330-006-0244-z
Hinderer S, Schenke-Layland K (2019) Cardiac fibrosis – a short review of causes and therapeutic strategies. Adv Drug Deliv Rev. https://doi.org/10.1016/j.addr.2019.05.011
Fan Z, Guan J (2016) Antifibrotic therapies to control cardiac fibrosis. Biomater Res 20:13. https://doi.org/10.1186/s40824-016-0060-8
Montgomery RL, Hullinger TG, Semus HM, Dickinson BA, Seto AG, Lynch JM, Stack C, Latimer PA, Olson EN, van Rooij E (2011) Therapeutic inhibition of miR-208a improves cardiac function and survival during heart failure. Circulation 124:1537–1547. https://doi.org/10.1161/CIRCULATIONAHA.111.030932
Jaffe LM, Morin DP (2014) Cardiac resynchronization therapy: history, present status, and future directions. Ochsner J 14:596–607
Zusterzeel R, Curtis JP, Caños DA, Sanders WE, Selzman KA, Piña IL, Spatz ES, Bao H, Ponirakis A, Varosy PD, Masoudi FA, Strauss DG (2014) Sex-specific mortality risk by QRS morphology and duration in patients receiving CRT. J Am Coll Cardiol 64:887–894. https://doi.org/10.1016/j.jacc.2014.06.1162
Landry NM, Cohen S, Dixon IMC (2018) Periostin in cardiovascular disease and development: a tale of two distinct roles. Basic Res Cardiol 113:1. https://doi.org/10.1007/s00395-017-0659-5
Travers JG, Kamal FA, Robbins J, Yutzey KE, Blaxall BC (2016) Cardiac fibrosis. Circ Res 118:1021–1040. https://doi.org/10.1161/CIRCRESAHA.115.306565
Shinde AV, Humeres C, Frangogiannis NG (2017) The role of α-smooth muscle actin in fibroblast-mediated matrix contraction and remodeling. Biochim Biophys Acta Mol basis Dis 1863:298–309. https://doi.org/10.1016/j.bbadis.2016.11.006
DeLeon-Pennell KY (2016) May the fibrosis be with you: is discoidin domain receptor 2 the receptor we have been looking for? J Mol Cell Cardiol 91:201–203. https://doi.org/10.1016/j.yjmcc.2016.01.006
Kong P, Christia P, Saxena A, Su Y, Frangogiannis NG (2013) Lack of specificity of fibroblast-specific protein 1 in cardiac remodeling and fibrosis. Am J Physiol Circ Physiol 305:H1363–H1372. https://doi.org/10.1152/ajpheart.00395.2013
Wang L, Yue Y, Yang X, Fan T, Mei B, Hou J, Liang M, Chen G, Wu Z (2017) Platelet derived growth factor alpha (PDGFRα) induces the activation of cardiac fibroblasts by activating c-kit. Med Sci Monit 23:3808–3816. https://doi.org/10.12659/MSM.906038
Chu P-Y, Mariani J, Finch S, McMullen JR, Sadoshima J, Marshall T, Kaye DM (2010) Bone marrow-derived cells contribute to fibrosis in the chronically failing heart. Am J Pathol 176:1735–1742. https://doi.org/10.2353/ajpath.2010.090574
Zhao X-H, Laschinger C, Arora P, Szaszi K, Kapus A, McCulloch CA (2007) Force activates smooth muscle -actin promoter activity through the Rho signaling pathway. J Cell Sci 120:1801–1809. https://doi.org/10.1242/jcs.001586
Bansal SS, Ismahil MA, Goel M, Patel B, Hamid T, Rokosh G, Prabhu SD (2017) Activated T lymphocytes are essential drivers of pathological remodeling in ischemic heart failure. Circ Hear Fail:10. https://doi.org/10.1161/CIRCHEARTFAILURE.116.003688
Abdullah CS, Li Z, Wang X, Jin Z-Q (2016) Depletion of T lymphocytes ameliorates cardiac fibrosis in streptozotocin-induced diabetic cardiomyopathy. Int Immunopharmacol 39:251–264. https://doi.org/10.1016/j.intimp.2016.07.027
Koitabashi N, Danner T, Zaiman AL, Pinto YM, Rowell J, Mankowski J, Zhang D, Nakamura T, Takimoto E, Kass DA (2011) Pivotal role of cardiomyocyte TGF-β signaling in the murine pathological response to sustained pressure overload. J Clin Invest 121:2301–2312. https://doi.org/10.1172/JCI44824
Yue Y, Meng K, Pu Y, Zhang X (2017) Transforming growth factor beta (TGF-β) mediates cardiac fibrosis and induces diabetic cardiomyopathy. Diabetes Res Clin Pract 133:124–130. https://doi.org/10.1016/j.diabres.2017.08.018
Mishra R, Cool BL, Laderoute KR, Foretz M, Viollet B, Simonson MS (2008) AMP-activated protein kinase inhibits transforming growth factor-β-induced Smad3-dependent transcription and Myofibroblast Transdifferentiation. J Biol Chem 283:10461–10469. https://doi.org/10.1074/jbc.M800902200
Wei C, Kim I-K, Kumar S, Jayasinghe S, Hong N, Castoldi G, Catalucci D, Jones WK, Gupta S (2013) NF-κB mediated miR-26a regulation in cardiac fibrosis. J Cell Physiol 228:1433–1442. https://doi.org/10.1002/jcp.24296
Duerrschmid C, Trial J, Wang Y, Entman ML, Haudek SB (2015) Tumor necrosis factor. Circ Heart Fail 8:352–361. https://doi.org/10.1161/CIRCHEARTFAILURE.114.001893
Szekely Y, Arbel Y (2018) A review of interleukin-1 in heart disease: where do we stand today? Cardiol Ther 7:25–44. https://doi.org/10.1007/s40119-018-0104-3
Lluri G, Deb A (2019) WNT signaling and cardiac fibrosis. Pp 319–334
Xiang F-L, Fang M, Yutzey KE (2017) Loss of β-catenin in resident cardiac fibroblasts attenuates fibrosis induced by pressure overload in mice. Nat Commun 8:712. https://doi.org/10.1038/s41467-017-00840-w
Menazza S, Murphy E (2016) The expanding complexity of estrogen receptor signaling in the cardiovascular system. Circ Res 118:994–1007. https://doi.org/10.1161/CIRCRESAHA.115.305376
Kang S, Liu Y, Sun D, Zhou C, Liu A, Xu C, Hao Y, Li D, Yan C, Sun H (2012) Chronic activation of the G protein-coupled receptor 30 with agonist G-1 attenuates heart failure. PLoS One 7:e48185. https://doi.org/10.1371/journal.pone.0048185
Wang H, Jessup JA, Lin MS, Chagas C, Lindsey SH, Groban L (2012) Activation of GPR30 attenuates diastolic dysfunction and left ventricle remodelling in oophorectomized mRen2.Lewis rats. Cardiovasc Res 94:96–104. https://doi.org/10.1093/cvr/cvs090
Pedram A, Razandi M, Narayanan R, Levin ER (2016) Estrogen receptor beta signals to inhibition of cardiac fibrosis. Mol Cell Endocrinol 434:57–68. https://doi.org/10.1016/j.mce.2016.06.018
Dworatzek E, Mahmoodzadeh S, Schriever C, Kusumoto K, Kramer L, Santos G, Fliegner D, Leung Y-K, Ho S-M, Zimmermann W-H, Lutz S, Regitz-Zagrosek V (2019) Sex-specific regulation of collagen I and III expression by 17β-estradiol in cardiac fibroblasts: role of estrogen receptors. Cardiovasc Res 115:315–327. https://doi.org/10.1093/cvr/cvy185
Wang H, Zhao Z, Lin M, Groban L (2015) Activation of GPR30 inhibits cardiac fibroblast proliferation. Mol Cell Biochem 405:135–148. https://doi.org/10.1007/s11010-015-2405-3
Verdonschot JAJ, Hazebroek MR, Derks KWJ, Barandiarán Aizpurua A, Merken JJ, Wang P, Bierau J, van den Wijngaard A, Schalla SM, Abdul Hamid MA, van Bilsen M, van Empel VPM, Knackstedt C, Brunner-La Rocca H-P, Brunner HG, Krapels IPC, Heymans SRB (2018) Titin cardiomyopathy leads to altered mitochondrial energetics, increased fibrosis and long-term life-threatening arrhythmias. Eur Heart J 39:864–873. https://doi.org/10.1093/eurheartj/ehx808
Chatzifrangkeskou M, Le Dour C, Wu W, Morrow JP, Joseph LC, Beuvin M, Sera F, Homma S, Vignier N, Mougenot N, Bonne G, Lipson KE, Worman HJ, Muchir A (2016) ERK1/2 directly acts on CTGF/CCN2 expression to mediate myocardial fibrosis in cardiomyopathy caused by mutations in the lamin A/C gene. Hum Mol Genet 25:2220–2233. https://doi.org/10.1093/hmg/ddw090
Li W, Yin L, Shen C, Hu K, Ge J, Sun A (2018) SCN5A variants: association with cardiac disorders. Front Physiol 9. https://doi.org/10.3389/fphys.2018.01372
Levick SP, Soto-Pantoja DR, Bi J, Hundley WG, Widiapradja A, Manteufel EJ, Bradshaw TW, Meléndez GC (2018) Doxorubicin-induced myocardial fibrosis involves the neurokinin-1 receptor and direct effects on cardiac fibroblasts. Hear Lung Circ. https://doi.org/10.1016/j.hlc.2018.08.003
Robinson P, Kasembeli M, Bharadwaj U, Engineer N, Eckols KT, Tweardy DJ (2016) Substance P receptor signaling mediates doxorubicin-induced cardiomyocyte apoptosis and triple-negative breast cancer chemoresistance. Biomed Res Int 2016:1–9. https://doi.org/10.1155/2016/1959270
El-Agamy DS, El-Harbi KM, Khoshhal S, Ahmed N, Elkablawy MA, Shaaban AA, Abo-Haded HM (2018) Pristimerin protects against doxorubicin-induced cardiotoxicity and fibrosis through modulation of Nrf2 and MAPK/NF-kB signaling pathways. Cancer Manag Res Volume 11:47–61. https://doi.org/10.2147/CMAR.S186696
Chu W, Li C, Qu X, Zhao D, Wang X, Yu X, Cai F, Liang H, Zhang Y, Zhao X, Li B, Qiao G, Dong D, Lu Y, Du Z, Yang B (2012) Arsenic-induced interstitial myocardial fibrosis reveals a new insight into drug-induced long QT syndrome. Cardiovasc Res 96:90–98. https://doi.org/10.1093/cvr/cvs230
Zhang Y, Wu X, Li Y, Zhang H, Li Z, Zhang Y, Zhang L, Ju J, Liu X, Chen X, Glybochko PV, Nikolenko V, Kopylov P, Xu C, Yang B (2016) Endothelial to mesenchymal transition contributes to arsenic-trioxide-induced cardiac fibrosis. Sci Rep 6:33787. https://doi.org/10.1038/srep33787
Heinzerling L, Ott PA, Hodi FS, Husain AN, Tajmir-Riahi A, Tawbi H, Pauschinger M, Gajewski TF, Lipson EJ, Luke JJ (2016) Cardiotoxicity associated with CTLA4 and PD1 blocking immunotherapy. J Immunother Cancer 4:50. https://doi.org/10.1186/s40425-016-0152-y
Geng Y, Liu X, Liang J, Habiel DM, Vrishika K, Coelho AL, Deng N, Xie T, Wang Y, Liu N, Huang G, Kurkciyan A, Liu Z, Tang J, Hogaboam CM, Jiang D, Noble PW (2019) PD-L1 on invasive fibroblasts drives fibrosis in a humanized model of idiopathic pulmonary fibrosis. JCI Insight. https://doi.org/10.1172/jci.insight.125326
Delgobo M, Frantz S (2018) Heart failure in cancer: role of checkpoint inhibitors. J Thorac Dis 10:S4323–S4334. https://doi.org/10.21037/jtd.2018.10.07
Maisch B (2016) Alcoholic cardiomyopathy. Herz 41:484–493. https://doi.org/10.1007/s00059-016-4469-6
Fernández-Solà J, Planavila Porta A (2016) New treatment strategies for alcohol-induced heart damage. Int J Mol Sci 17:1651. https://doi.org/10.3390/ijms17101651
Havakuk O, Rezkalla SH, Kloner RA (2017) The cardiovascular effects of cocaine. J Am Coll Cardiol 70:101–113. https://doi.org/10.1016/j.jacc.2017.05.014
Paratz ED, Cunningham NJ, MacIsaac AI (2016) The cardiac complications of methamphetamines. Hear Lung Circ 25:325–332. https://doi.org/10.1016/j.hlc.2015.10.019
Tschöpe C, Müller I, Xia Y, Savvatis K, Pappritz K, Pinkert S, Lassner D, Heimesaat MM, Spillmann F, Miteva K, Bereswill S, Schultheiss H-P, Fechner H, Pieske B, Kühl U, Van Linthout S (2017) NOD2 (nucleotide-binding oligomerization domain 2) is a major pathogenic mediator of coxsackievirus B3-induced myocarditis. Circ hear fail 10. https://doi.org/10.1161/CIRCHEARTFAILURE.117.003870
Cao Y, Xu W, Xiong S (2013) Adoptive transfer of regulatory T cells protects against coxsackievirus B3-induced cardiac fibrosis. PLoS One 8:e74955. https://doi.org/10.1371/journal.pone.0074955
Chen P, Xie Y, Shen E, Li GG, Yu Y, Zhang CB, Yang Y, Zou Y, Ge J, Chen R, Chen H (2011) Astragaloside IV attenuates myocardial fibrosis by inhibiting TGF-β1 signaling in coxsackievirus B3-induced cardiomyopathy. Eur J Pharmacol 658:168–174. https://doi.org/10.1016/j.ejphar.2011.02.040
Hsue PY, Tawakol A (2016) Inflammation and fibrosis in HIV. Circ Cardiovasc Imaging 9. https://doi.org/10.1161/CIRCIMAGING.116.004427
Laurence J, Elhadad S, Robison T, Terry H, Varshney R, Woolington S, Ghafoory S, Choi ME, Ahamed J (2017) HIV protease inhibitor-induced cardiac dysfunction and fibrosis is mediated by platelet-derived TGF-β1 and can be suppressed by exogenous carbon monoxide. PLoS One 12:e0187185. https://doi.org/10.1371/journal.pone.0187185
Fowlkes V, Clark J, Fix C, Law BA, Morales MO, Qiao X, Ako-Asare K, Goldsmith JG, Carver W, Murray DB, Goldsmith EC (2013) Type II diabetes promotes a myofibroblast phenotype in cardiac fibroblasts. Life Sci 92:669–676. https://doi.org/10.1016/j.lfs.2013.01.003
Russo I, Frangogiannis NG (2016) Diabetes-associated cardiac fibrosis: cellular effectors, molecular mechanisms and therapeutic opportunities. J Mol Cell Cardiol 90:84–93. https://doi.org/10.1016/j.yjmcc.2015.12.011
Yuan H, Fan Y, Wang Y, Gao T, Shao Y, Zhao B, Li H, Xu C, Wei C (2019) Calcium-sensing receptor promotes high glucose-induced myocardial fibrosis via upregulation of the TGF-β1/Smads pathway in cardiac fibroblasts. Mol Med Rep. https://doi.org/10.3892/mmr.2019.10330
Mitrut R, Stepan AE, Pirici D Histopathological aspects of the myocardium in dilated cardiomyopathy. Curr Heal Sci J 44:243–249. https://doi.org/10.12865/CHSJ.44.03.07
Cunningham KS (2006) An approach to endomyocardial biopsy interpretation. J Clin Pathol 59:121–129. https://doi.org/10.1136/jcp.2005.026443
Liu T, Song D, Dong J, Zhu P, Liu J, Liu W, Ma X, Zhao L, Ling S (2017) Current understanding of the pathophysiology of myocardial fibrosis and its quantitative assessment in heart failure. Front Physiol 8. https://doi.org/10.3389/fphys.2017.00238
López B, González A, Ravassa S, Beaumont J, Moreno MU, San José G, Querejeta R, Díez J (2015) Circulating biomarkers of myocardial fibrosis. J Am Coll Cardiol 65:2449–2456. https://doi.org/10.1016/j.jacc.2015.04.026
Aoki T, Fukumoto Y, Sugimura K, Oikawa M, Satoh K, Nakano M, Nakayama M, Shimokawa H (2011) Prognostic impact of myocardial interstitial fibrosis in non-ischemic heart failure. Circ J 75:2605–2613. https://doi.org/10.1253/circj.CJ-11-0568
Strimbu K, Tavel JA (2010) What are biomarkers? Curr Opin HIV AIDS 5:463–466. https://doi.org/10.1097/COH.0b013e32833ed177
Seo W-Y, Kim J-H, Baek D-S, Kim S-J, Kang S, Yang WS, Song J-A, Lee M-S, Kim S, Kim Y-S (2017) Production of recombinant human procollagen type I C-terminal propeptide and establishment of a sandwich ELISA for quantification. Sci Rep 7:15946. https://doi.org/10.1038/s41598-017-16290-9
López B, Querejeta R, González A, Larman M, Díez J (2012) Collagen cross-linking but not collagen amount associates with elevated filling pressures in hypertensive patients with stage C heart failure. Hypertension 60:677–683. https://doi.org/10.1161/HYPERTENSIONAHA.112.196113
Izawa H, Murohara T, Nagata K, Isobe S, Asano H, Amano T, Ichihara S, Kato T, Ohshima S, Murase Y, Iino S, Obata K, Noda A, Okumura K, Yokota M (2005) Mineralocorticoid receptor antagonism ameliorates left ventricular diastolic dysfunction and myocardial fibrosis in mildly symptomatic patients with idiopathic dilated cardiomyopathy. Circulation 112:2940–2945. https://doi.org/10.1161/CIRCULATIONAHA.105.571653
Kaufman BD, Videon N, Zhang X, Harris MA, Shaddy RE, Goldmuntz E (2015) Procollagen type III amino-terminal propeptide: a serum biomarker of left ventricular remodelling in paediatric dilated cardiomyopathy. Cardiol Young 25:228–236. https://doi.org/10.1017/S1047951113001820
Sciacchitano S, Lavra L, Morgante A, Ulivieri A, Magi F, De Francesco G, Bellotti C, Salehi L, Ricci A (2018) Galectin-3: one molecule for an alphabet of diseases, from A to Z. Int J Mol Sci 19:379. https://doi.org/10.3390/ijms19020379
Calvier L, Martinez-Martinez E, Miana M, Cachofeiro V, Rousseau E, Sádaba JR, Zannad F, Rossignol P, López-Andrés N (2015) The impact of Galectin-3 inhibition on aldosterone-induced cardiac and renal injuries. JACC Hear Fail 3:59–67. https://doi.org/10.1016/j.jchf.2014.08.002
MacKinnon AC, Gibbons MA, Farnworth SL, Leffler H, Nilsson UJ, Delaine T, Simpson AJ, Forbes SJ, Hirani N, Gauldie J, Sethi T (2012) Regulation of transforming growth factor-β1–driven lung fibrosis by galectin-3. Am J Respir Crit Care Med 185:537–546. https://doi.org/10.1164/rccm.201106-0965OC
Wu C-K, Su M-Y, Lee J-K, Chiang F-T, Hwang J-J, Lin J-L, Chen J-J, Liu F-T, Tsai C-T (2015) Galectin-3 level and the severity of cardiac diastolic dysfunction using cellular and animal models and clinical indices. Sci Rep 5:17007. https://doi.org/10.1038/srep17007
Song X, Qian X, Shen M, Jiang R, Wagner MB, Ding G, Chen G, Shen B (2015) Protein kinase C promotes cardiac fibrosis and heart failure by modulating galectin-3 expression. Biochim Biophys Acta, Mol Cell Res 1853:513–521. https://doi.org/10.1016/j.bbamcr.2014.12.001
Suthahar N, Meijers WC, Silljé HHW, Ho JE, Liu F-T, de Boer RA (2018) Galectin-3 activation and inhibition in heart failure and cardiovascular disease: an update. Theranostics 8:593–609. https://doi.org/10.7150/thno.22196
Liu Y-H, D’Ambrosio M, Liao T, Peng H, Rhaleb N-E, Sharma U, André S, Gabius H-J, Carretero OA (2009) N -acetyl-seryl-aspartyl-lysyl-proline prevents cardiac remodeling and dysfunction induced by galectin-3, a mammalian adhesion/growth-regulatory lectin. Am J Physiol Circ Physiol 296:H404–H412. https://doi.org/10.1152/ajpheart.00747.2008
Martínez-Martínez E, Brugnolaro C, Ibarrola J, Ravassa S, Buonafine M, López B, Fernández-Celis A, Querejeta R, Santamaria E, Fernández-Irigoyen J, Rábago G, Moreno MU, Jaisser F, Díez J, González A, López-Andrés N (2019) CT-1 (cardiotrophin-1)-Gal-3 (galectin-3) axis in cardiac fibrosis and inflammation. Hypertension 73:602–611. https://doi.org/10.1161/HYPERTENSIONAHA.118.11874
Agoston-Coldea L, Bheecarry K, Petra C, Strambu L, Ober C, Revnic R, Lupu S, Mocan T, Fodor D (2018) The value of global longitudinal strain and galectin-3 for predicting cardiovascular events in patients with severe aortic stenosis. Med Ultrason 20:205. https://doi.org/10.11152/mu-1456
Ho JE, Liu C, Lyass A, Courchesne P, Pencina MJ, Vasan RS, Larson MG, Levy D (2012) Galectin-3, a marker of cardiac fibrosis, predicts incident heart failure in the community. J Am Coll Cardiol 60:1249–1256. https://doi.org/10.1016/j.jacc.2012.04.053
de Boer RA, Lok DJA, Jaarsma T, van der Meer P, Voors AA, Hillege HL, van Veldhuisen DJ (2011) Predictive value of plasma galectin-3 levels in heart failure with reduced and preserved ejection fraction. Ann Med 43:60–68. https://doi.org/10.3109/07853890.2010.538080
Vergaro G, Del Franco A, Giannoni A, Prontera C, Ripoli A, Barison A, Masci PG, Aquaro GD, Cohen Solal A, Padeletti L, Passino C, Emdin M (2015) Galectin-3 and myocardial fibrosis in nonischemic dilated cardiomyopathy. Int J Cardiol 184:96–100. https://doi.org/10.1016/j.ijcard.2015.02.008
Villacorta H, Maisel AS (2015) Soluble ST2 testing: a promising biomarker in the management of heart failure. Arq Bras Cardiol. https://doi.org/10.5935/abc.20150151
Lupu S, Agoston-Coldea L (2015) Soluble ST2 in ventricular dysfunction. Pp 139–159
Bayes-Genis A, de Antonio M, Vila J, Peñafiel J, Galán A, Barallat J, Zamora E, Urrutia A, Lupón J (2014) Head-to-head comparison of 2 myocardial fibrosis biomarkers for long-term heart failure risk stratification. J Am Coll Cardiol 63:158–166. https://doi.org/10.1016/j.jacc.2013.07.087
Santhanakrishnan R, Chong JPC, Ng TP, Ling LH, Sim D, Leong KT, Yeo PS, Ong HY, Jaufeerally F, Wong R, Chai P, Low AF, Richards AM, Lam CSP (2012) Growth differentiation factor 15, ST2, high-sensitivity troponin T, and N-terminal pro brain natriuretic peptide in heart failure with preserved vs. reduced ejection fraction. Eur J Heart Fail 14:1338–1347. https://doi.org/10.1093/eurjhf/hfs130
Wang Y-C, Yu C-C, Chiu F-C, Tsai C-T, Lai L-P, Hwang J-J, Lin J-L (2013) Soluble ST2 as a biomarker for detecting stable heart failure with a Normal ejection fraction in hypertensive patients. J Card Fail 19:163–168. https://doi.org/10.1016/j.cardfail.2013.01.010
Agoston-Coldea L, Lupu S, Hicea S, Paradis A, Mocan T (2014) Serum levels of the soluble IL-1 receptor family member ST2 and right ventricular dysfunction. Biomark Med 8:95–106. https://doi.org/10.2217/bmm.13.116
Wojciechowska C, Romuk E, Nowalany-Kozielska E, Jacheć W (2017) Serum galectin-3 and ST2 as predictors of unfavorable outcome in stable dilated cardiomyopathy patients. Hell J Cardiol 58:350–359. https://doi.org/10.1016/j.hjc.2017.03.006
You H, Jiang W, Jiao M, Wang X, Jia L, You S, Li Y, Wen H, Jiang H, Yuan H, Huang J, Qiao B, Yang Y, Jin M, Wang Y, Du J (2019) Association of soluble ST2 serum levels with outcomes in pediatric dilated cardiomyopathy. Can J Cardiol 35:727–735. https://doi.org/10.1016/j.cjca.2019.02.016
Desmedt S, Desmedt V, De Vos L, Delanghe JR, Speeckaert R, Speeckaert MM (2019) Growth differentiation factor 15: a novel biomarker with high clinical potential. Crit Rev Clin Lab Sci 56:333–350. https://doi.org/10.1080/10408363.2019.1615034
Wang F, Guo Y, Yu H, Zheng L, Mi L, Gao W (2010) Growth differentiation factor 15 in different stages of heart failure: potential screening implications. Biomarkers 15:671–676. https://doi.org/10.3109/1354750X.2010.510580
Nair N, Gongora E (2018) Correlations of GDF-15 with sST2, MMPs, and worsening functional capacity in idiopathic dilated cardiomyopathy. J Circ Biomarkers 7:184945441775173. https://doi.org/10.1177/1849454417751735
Wang F-F, Chen B-X, Yu H-Y, Mi L, Li Z-J, Gao W (2016) Correlation between growth differentiation factor-15 and collagen metabolism indicators in patients with myocardial infarction and heart failure. J Geriatr Cardiol 13:88–93. https://doi.org/10.11909/j.issn.1671-5411.2016.01.002
Cui J, Zhou B, Ross SA, Zempleni J (2017) Nutrition, microRNAs, and human health. Adv Nutr An Int Rev J 8:105–112. https://doi.org/10.3945/an.116.013839
Huang W (2017) MicroRNAs: biomarkers, diagnostics, and therapeutics. Pp 57–67
Cao W, Shi P, Ge J-J (2017) miR-21 enhances cardiac fibrotic remodeling and fibroblast proliferation via CADM1/STAT3 pathway. BMC Cardiovasc Disord 17:88. https://doi.org/10.1186/s12872-017-0520-7
Yuan J, Chen H, Ge D, Xu Y, Xu H, Yang Y, Gu M, Zhou Y, Zhu J, Ge T, Chen Q, Gao Y, Wang Y, Li X, Zhao Y (2017) Mir-21 promotes cardiac fibrosis after myocardial infarction via targeting Smad7. Cell Physiol Biochem 42:2207–2219. https://doi.org/10.1159/000479995
Brønnum H, Andersen DC, Schneider M, Sandberg MB, Eskildsen T, Nielsen SB, Kalluri R, Sheikh SP (2013) miR-21 promotes fibrogenic epithelial-to-mesenchymal transition of epicardial mesothelial cells involving programmed cell death 4 and sprouty-1. PLoS one 8:e56280. https://doi.org/10.1371/journal.pone.0056280
Li X, Liu CY, Li YS, Xu J, Li DG, Han D (2016) Deep RNA sequencing elucidates microRNA-regulated molecular pathways in ischemic cardiomyopathy and nonischemic cardiomyopathy. Genet Mol Res:15. https://doi.org/10.4238/gmr.15027465
Dai Y, Dai D, Mehta JL (2014) MicroRNA-29, a mysterious regulator in myocardial fibrosis and circulating miR-29a as a biomarker. J Am Coll Cardiol 64:2181. https://doi.org/10.1016/j.jacc.2014.03.064
Sassi Y, Avramopoulos P, Ramanujam D, Grüter L, Werfel S, Giosele S, Brunner A-D, Esfandyari D, Papadopoulou AS, De Strooper B, Hübner N, Kumarswamy R, Thum T, Yin X, Mayr M, Laggerbauer B, Engelhardt S (2017) Cardiac myocyte miR-29 promotes pathological remodeling of the heart by activating Wnt signaling. Nat Commun 8:1614. https://doi.org/10.1038/s41467-017-01737-4
Yamada Y, Takanashi M, Sudo K, Ueda S, Ohno S, Kuroda M (2017) Novel form of miR-29b suppresses bleomycin-induced pulmonary fibrosis. PLoS One 12:e0171957. https://doi.org/10.1371/journal.pone.0171957
Drummond CA, Fan X, Haller ST, Kennedy DJ, Liu J, Tian J (2018) Na/K-ATPase signaling mediates miR-29b-3p regulation and cardiac fibrosis formation in mice with chronic kidney disease. PLoS One 13:e0197688. https://doi.org/10.1371/journal.pone.0197688
Liang J, Zou X, Fang X, Xu J, Xiao Z, Zhu J, Li H, Yang J, Zeng N, Yuan S, Pan R, Fu Y, Zhang M, Luo J, Wang S, Shan Z (2019) The Smad3-miR-29b/miR-29c axis mediates the protective effect of macrophage migration inhibitory factor against cardiac fibrosis. Biochim Biophys Acta Mol basis Dis 1865:2441–2450. https://doi.org/10.1016/j.bbadis.2019.06.004
Jiang X, Tsitsiou E, Herrick SE, Lindsay MA (2010) MicroRNAs and the regulation of fibrosis. FEBS J 277:2015–2021. https://doi.org/10.1111/j.1742-4658.2010.07632.x
Chen L, Ji Q, Zhu H, Ren Y, Fan Z, Tian N (2018) miR-30a attenuates cardiac fibrosis in rats with myocardial infarction by inhibiting CTGF. Exp Ther Med. https://doi.org/10.3892/etm.2018.5952
Angelini A, Li Z, Mericskay M, Decaux J-F (2015) Regulation of connective tissue growth factor and cardiac fibrosis by an SRF/MicroRNA-133a axis. PLoS One 10:e0139858. https://doi.org/10.1371/journal.pone.0139858
Li N, Zhou H, Tang Q (2018) miR-133: a suppressor of cardiac remodeling? Front Pharmacol 9. https://doi.org/10.3389/fphar.2018.00903
Wang DS, Zhang HQ, Zhang B, Yuan ZB, Yu ZK, Yang T, Zhang SQ, Liu Y, Jia XX (2016) miR-133 inhibits pituitary tumor cell migration and invasion via down-regulating FOXC1 expression. Genet Mol res 15. https://doi.org/10.4238/gmr.15017453
Chen S, Puthanveetil P, Feng B, Matkovich SJ, Dorn GW, Chakrabarti S (2014) Cardiac miR-133a overexpression prevents early cardiac fibrosis in diabetes. J Cell Mol Med 18:415–421. https://doi.org/10.1111/jcmm.12218
Nair N, Kumar S, Gongora E, Gupta S (2013) Circulating miRNA as novel markers for diastolic dysfunction. Mol Cell Biochem 376:33–40. https://doi.org/10.1007/s11010-012-1546-x
Wong LL, Armugam A, Sepramaniam S, Karolina DS, Lim KY, Lim JY, Chong JPC, Ng JYX, Chen Y-T, Chan MMY, Chen Z, Yeo PSD, Ng TP, Ling LH, Sim D, Leong KTG, Ong HY, Jaufeerally F, Wong R, Chai P, Low AF, Lam CSP, Jeyaseelan K, Richards AM (2015) Circulating microRNAs in heart failure with reduced and preserved left ventricular ejection fraction. Eur J Heart Fail 17:393–404. https://doi.org/10.1002/ejhf.223
Watson CJ, Gupta SK, O’Connell E, Thum S, Glezeva N, Fendrich J, Gallagher J, Ledwidge M, Grote-Levi L, McDonald K, Thum T (2015) MicroRNA signatures differentiate preserved from reduced ejection fraction heart failure. Eur J Heart Fail 17:405–415. https://doi.org/10.1002/ejhf.244
Mewton N, Liu CY, Croisille P, Bluemke D, Lima JAC (2011) Assessment of myocardial fibrosis with cardiovascular magnetic resonance. J Am Coll Cardiol 57:891–903. https://doi.org/10.1016/j.jacc.2010.11.013
Iles LM, Ellims AH, Llewellyn H, Hare JL, Kaye DM, McLean CA, Taylor AJ (2015) Histological validation of cardiac magnetic resonance analysis of regional and diffuse interstitial myocardial fibrosis. Eur Heart J Cardiovasc Imaging 16:14–22. https://doi.org/10.1093/ehjci/jeu182
Messroghli DR, Radjenovic A, Kozerke S, Higgins DM, Sivananthan MU, Ridgway JP (2004) Modified Look-Locker inversion recovery (MOLLI) for high-resolutionT1 mapping of the heart. Magn Reson Med 52:141–146. https://doi.org/10.1002/mrm.20110
McCrohon JA, Moon JCC, Prasad SK, McKenna WJ, Lorenz CH, Coats AJS, Pennell DJ (2003) Differentiation of heart failure related to dilated cardiomyopathy and coronary artery disease using gadolinium-enhanced cardiovascular magnetic resonance. Circulation 108:54–59. https://doi.org/10.1161/01.CIR.0000078641.19365.4C
Assomull RG, Prasad SK, Lyne J, Smith G, Burman ED, Khan M, Sheppard MN, Poole-Wilson PA, Pennell DJ (2006) Cardiovascular magnetic resonance, fibrosis, and prognosis in dilated cardiomyopathy. J Am Coll Cardiol 48:1977–1985. https://doi.org/10.1016/j.jacc.2006.07.049
Duan X, Li J, Zhang Q, Zeng Z, Luo Y, Jiang J, Chen Y (2015) Prognostic value of late gadolinium enhancement in dilated cardiomyopathy patients: a meta-analysis. Clin Radiol 70:999–1008. https://doi.org/10.1016/j.crad.2015.05.007
Machii M, Satoh H, Shiraki K, Saotome M, Urushida T, Katoh H, Takehara Y, Sakahara H, Ohtani H, Wakabayashi Y, Ukigai H, Tawarahara K, Hayashi H (2014) Distribution of late gadolinium enhancement in end-stage hypertrophic cardiomyopathy and dilated cardiomyopathy: differential diagnosis and prediction of cardiac outcome. Magn Reson Imaging 32:118–124. https://doi.org/10.1016/j.mri.2013.10.011
Tachi M, Amano Y, Inui K, Takeda M, Yamada F, Asai K, Kumita S (2016) Relationship of postcontrast myocardial T1 value and delayed enhancement to reduced cardiac function and serious arrhythmia in dilated cardiomyopathy with left ventricular ejection fraction less than 35%. Acta Radiol 57:430–436. https://doi.org/10.1177/0284185115580840
Patel AR, Kramer CM (2017) Role of cardiac magnetic resonance in the diagnosis and prognosis of nonischemic cardiomyopathy. JACC Cardiovasc Imaging 10:1180–1193. https://doi.org/10.1016/j.jcmg.2017.08.005
Taylor RJ, Umar F, Lin ELS, Ahmed A, Moody WE, Mazur W, Stegemann B, Townend JN, Steeds RP, Leyva F (2015) Mechanical effects of left ventricular midwall fibrosis in non-ischemic cardiomyopathy. J Cardiovasc Magn Reson 18:1. https://doi.org/10.1186/s12968-015-0221-2
Jellis CL, Kwon DH (2014) Myocardial T1 mapping: modalities and clinical applications. Cardiovasc Diagn Ther 4:126–137. https://doi.org/10.3978/j.issn.2223-3652.2013.09.03
Burt JR, Zimmerman SL, Kamel IR, Halushka M, Bluemke DA (2014) Myocardial T1 mapping: techniques and potential applications. RadioGraphics 34:377–395. https://doi.org/10.1148/rg.342125121
Flett AS, Hayward MP, Ashworth MT, Hansen MS, Taylor AM, Elliott PM, McGregor C, Moon JC (2010) Equilibrium contrast cardiovascular magnetic resonance for the measurement of diffuse myocardial fibrosis. Circulation 122:138–144. https://doi.org/10.1161/CIRCULATIONAHA.109.930636
Halliday BP, Gulati A, Ali A, Guha K, Newsome S, Arzanauskaite M, Vassiliou VS, Lota A, Izgi C, Tayal U, Khalique Z, Stirrat C, Auger D, Pareek N, Ismail TF, Rosen SD, Vazir A, Alpendurada F, Gregson J, Frenneaux MP, Cowie MR, Cleland JGF, Cook SA, Pennell DJ, Prasad SK (2017) Association between midwall late gadolinium enhancement and sudden cardiac death in patients with dilated cardiomyopathy and mild and moderate left ventricular systolic dysfunction. Circulation 135:2106–2115. https://doi.org/10.1161/CIRCULATIONAHA.116.026910
Klem I, Weinsaft JW, Bahnson TD, Hegland D, Kim HW, Hayes B, Parker MA, Judd RM, Kim RJ (2012) Assessment of myocardial scarring improves risk stratification in patients evaluated for cardiac defibrillator implantation. J Am Coll Cardiol 60:408–420. https://doi.org/10.1016/j.jacc.2012.02.070
Ota S (2019) The pattern of myocardial fibrosis detected by cardiovascular magnetic resonance imaging provides prognostic information in patients with idiopathic dilated cardiomyopathy. J Am Coll Cardiol 73:1551. https://doi.org/10.1016/S0735-1097(19)32157-6
Kono AK, Ishii K, Kumagai H, Taniguchi Y, Kajiya T, Sugimura K (2010) Late gadolinium enhancement on cardiac magnetic resonance imaging: is it associated with a higher incidence of nonsustained ventricular tachycardia in patients with idiopathic dilated cardiomyopathy? Jpn J Radiol 28:355–361. https://doi.org/10.1007/s11604-010-0433-1
Leyva F, Foley PW, Chalil S, Ratib K, Smith RE, Prinzen F, Auricchio A (2011) Cardiac resynchronization therapy guided by late gadolinium-enhancement cardiovascular magnetic resonance. J Cardiovasc Magn Reson 13:29. https://doi.org/10.1186/1532-429X-13-29
Leong DP, Chakrabarty A, Shipp N, Molaee P, Madsen PL, Joerg L, Sullivan T, Worthley SG, De Pasquale CG, Sanders P, Selvanayagam JB (2012) Effects of myocardial fibrosis and ventricular dyssynchrony on response to therapy in new-presentation idiopathic dilated cardiomyopathy: insights from cardiovascular magnetic resonance and echocardiography. Eur Heart J 33:640–648. https://doi.org/10.1093/eurheartj/ehr391
Leyva F, Taylor RJ, Foley PWX, Umar F, Mulligan LJ, Patel K, Stegemann B, Haddad T, Smith REA, Prasad SK (2012) Left ventricular midwall fibrosis as a predictor of mortality and morbidity after cardiac resynchronization therapy in patients with nonischemic cardiomyopathy. J Am Coll Cardiol 60:1659–1667. https://doi.org/10.1016/j.jacc.2012.05.054
Gulati A, Jabbour A, Ismail TF, Guha K, Khwaja J, Raza S, Morarji K, Brown TDH, Ismail NA, Dweck MR, Di Pietro E, Roughton M, Wage R, Daryani Y, O’Hanlon R, Sheppard MN, Alpendurada F, Lyon AR, Cook SA, Cowie MR, Assomull RG, Pennell DJ, Prasad SK (2013) Association of fibrosis with mortality and sudden cardiac death in patients with nonischemic dilated cardiomyopathy. JAMA 309:896. https://doi.org/10.1001/jama.2013.1363
Puntmann VO, Carr-White G, Jabbour A, Yu C-Y, Gebker R, Kelle S, Hinojar R, Doltra A, Varma N, Child N, Rogers T, Suna G, Arroyo Ucar E, Goodman B, Khan S, Dabir D, Herrmann E, Zeiher AM, Nagel E (2016) T1-mapping and outcome in nonischemic cardiomyopathy. JACC Cardiovasc Imaging 9:40–50. https://doi.org/10.1016/j.jcmg.2015.12.001
Pi S-H, Kim SM, Choi J-O, Kim EK, Chang S-A, Choe YH, Lee S-C, Jeon E-S (2018) Prognostic value of myocardial strain and late gadolinium enhancement on cardiovascular magnetic resonance imaging in patients with idiopathic dilated cardiomyopathy with moderate to severely reduced ejection fraction. J Cardiovasc Magn Reson 20:36. https://doi.org/10.1186/s12968-018-0466-7
Oh J, Hong YJ, Ha J, Chun KH, Kim H, Lee CJ, Kim YJ, Choi BW, Kang SM (2019) P3555Lower native T1, extracellular volume and T2 on cardiac magnetic resonance imaging is related to more left ventricular reverse remodeling in nonischemic dilated cardiomyopathy. Eur heart J 40. https://doi.org/10.1093/eurheartj/ehz745.0418
Yancy CW, Jessup M, Bozkurt B, Butler J, Casey DE, Drazner MH, Fonarow GC, Geraci SA, Horwich T, Januzzi JL, Johnson MR, Kasper EK, Levy WC, Masoudi FA, McBride PE, McMurray JJV, Mitchell JE, Peterson PN, Riegel B, Sam F, Stevenson LW, Tang WHW, Tsai EJ, Wilkoff BL (2013) 2013 ACCF/AHA guideline for the management of heart failure. J Am Coll Cardiol 62:e147–e239. https://doi.org/10.1016/j.jacc.2013.05.019
Ponikowski P, Voors AA, Anker SD, Bueno H, Cleland JGF, Coats AJS, Falk V, González-Juanatey JR, Harjola V-P, Jankowska EA, Jessup M, Linde C, Nihoyannopoulos P, Parissis JT, Pieske B, Riley JP, Rosano GMC, Ruilope LM, Ruschitzka F, Rutten FH, van der Meer P (2016) 2016 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J 37:2129–2200. https://doi.org/10.1093/eurheartj/ehw128
Russo V, Papa AA, Williams EA, Rago A, Palladino A, Politano L, Nigro G (2018) ACE inhibition to slow progression of myocardial fibrosis in muscular dystrophies. Trends Cardiovasc Med 28:330–337. https://doi.org/10.1016/j.tcm.2017.12.006
Leask A (2015) Getting to the heart of the matter. Circ Res 116:1269–1276. https://doi.org/10.1161/CIRCRESAHA.116.305381
Rog-Zielinska EA, Norris RA, Kohl P, Markwald R (2016) The living scar – cardiac fibroblasts and the injured heart. Trends Mol Med 22:99–114. https://doi.org/10.1016/j.molmed.2015.12.006
Tietjens J, Teerlink JR (2016) Serelaxin and acute heart failure. Heart 102:95–99. https://doi.org/10.1136/heartjnl-2014-306786
Wu X, Wang H, Wang Y, Shen H, Tan Y (2018) Serelaxin inhibits differentiation and fibrotic behaviors of cardiac fibroblasts by suppressing ALK-5/Smad2/3 signaling pathway. Exp Cell Res 362:17–27. https://doi.org/10.1016/j.yexcr.2017.10.004
Zhang N, Wei W-Y, Li L-L, Hu C, Tang Q-Z (2018) Therapeutic potential of polyphenols in cardiac fibrosis. Front Pharmacol 9. https://doi.org/10.3389/fphar.2018.00122
Xu C, Hu Y, Hou L, Ju J, Li X, Du N, Guan X, Liu Z, Zhang T, Qin W, Shen N, Bilal MU, Lu Y, Zhang Y, Shan H (2014) β-Blocker carvedilol protects cardiomyocytes against oxidative stress-induced apoptosis by up-regulating miR-133 expression. J Mol Cell Cardiol 75:111–121. https://doi.org/10.1016/j.yjmcc.2014.07.009
Ihm S-H, Chang K, Kim H-Y, Baek SH, Youn H-J, Seung K-B, Kim J-H (2010) Peroxisome proliferator-activated receptor-γ activation attenuates cardiac fibrosis in type 2 diabetic rats: the effect of rosiglitazone on myocardial expression of receptor for advanced glycation end products and of connective tissue growth factor. Basic Res Cardiol 105:399–407. https://doi.org/10.1007/s00395-009-0071-x
Shim CY, Song B-W, Cha M-J, Hwang K-C, Park S, Hong G-R, Kang S-M, Lee JE, Ha J-W, Chung N (2014) Combination of a peroxisome proliferator-activated receptor-gamma agonist and an angiotensin II receptor blocker attenuates myocardial fibrosis and dysfunction in type 2 diabetic rats. J Diabetes Investig 5:362–371. https://doi.org/10.1111/jdi.12153
Wang L-X, Yang X, Yue Y, Fan T, Hou J, Chen G-X, Liang M-Y, Wu Z-K (2017) Imatinib attenuates cardiac fibrosis by inhibiting platelet-derived growth factor receptors activation in isoproterenol induced model. PLoS One 12:e0178619. https://doi.org/10.1371/journal.pone.0178619
Chaudhry PA, Mishima T, Sharov VG, Hawkins J, Alferness C, Paone G, Sabbah HN (2000) Passive epicardial containment prevents ventricular remodeling in heart failure. Ann Thorac Surg 70:1275–1280. https://doi.org/10.1016/S0003-4975(00)01755-0
Doenst T, Ahn-Veelken L, Schlensak C, Berchtold-Herz M, Sarai K, Schaefer M, van de Loo A, Beyersdorf F (2001) Left ventricular reduction for idiopathic dilated cardiomyopathy as alternative to transplant - truth or dare?*. Thorac Cardiovasc Surg 49:70–74. https://doi.org/10.1055/s-2001-11709
Calafiore A (1999) Surgical treatment of dilated cardiomyopathy with conventional techniques*1. Eur J Cardio-Thoracic Surg 16:S73–S78. https://doi.org/10.1016/S1010-7940(99)00193-1
Isomura T, Suma H, Horii T, Sato T, Kikuchi N (2000) Partial left ventriculectomy, ventriculoplasty or valvular surgery for idiopathic dilated cardiomyopathy – the role of intra-operative echocardiography. Eur J Cardio-Thoracic Surg 17:239–245. https://doi.org/10.1016/S1010-7940(00)00322-5
Suma H, Isomura T, Horii T, Nomura F (2006) Septal anterior ventricular exclusion procedure for idiopathic dilated cardiomyopathy. Ann Thorac Surg 82:1344–1348. https://doi.org/10.1016/j.athoracsur.2006.04.096
WANG J, GONG X, CHEN H, QIN S, ZHOU N, SU Y, GE J (2017) Effect of cardiac resynchronization therapy on myocardial fibrosis and relevant cytokines in a canine model with experimental heart failure. J Cardiovasc Electrophysiol 28:438–445. https://doi.org/10.1111/jce.13171
Broch K, Murbræch K, Andreassen AK, Hopp E, Aakhus S, Gullestad L (2015) Contemporary outcome in patients with idiopathic dilated cardiomyopathy. Am J Cardiol 116:952–959. https://doi.org/10.1016/j.amjcard.2015.06.022
Keeling PJ, Goldman JH, Slade AKB, Elliott PM, Caforio ALP, Poloniecki J, McKenna WJ (1995) Prognosis of idiopathic dilated cardiomyopathy. J Card Fail 1:337–345. https://doi.org/10.1016/S1071-9164(05)80002-8
Author information
Authors and Affiliations
Contributions
B.C.M., A.Z., and L.A.C. researched data for the article. B.C.M. and A.Z. wrote the article. L.A.C. discussed the content of the article, and B.C.M., A.Z., and L.A.C. reviewed and edited before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cojan-Minzat, B.O., Zlibut, A. & Agoston-Coldea, L. Non-ischemic dilated cardiomyopathy and cardiac fibrosis. Heart Fail Rev 26, 1081–1101 (2021). https://doi.org/10.1007/s10741-020-09940-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10741-020-09940-0