Abstract
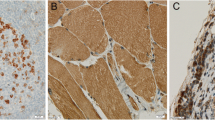
New immunomodulatory agents showed promising activity in brain metastases (BM). However, little is known about the inflammatory response in BM. New insights are needed to further guide the development of treatment strategies. We investigated 17 human autoptic tissue specimens of BM from breast cancer (n = 3), non-small cell lung cancer (NSCLC; n = 5), small cell lung cancer (n = 3) and melanoma (n = 6). Immunohistochemical staining for a comprehensive panel of 21 inflammation-associated markers was performed. Results were quantified by manual counting of the various cell populations in three areas of 0.5 mm2 (intratumoral, peritumoral, control region). Profound microglia activation with marked peritumoral accumulation and some intratumoral infiltration of HLA-DR-positive microglia/macrophages was found. A high proportion of these cells showed strong immunoreactivity for phagocytosis associated markers and MHC class 1, while a smaller subgroup of cells expressed molecules involved in radical production. Only few B- and T-lymphocytes were observed in and around BM. The number of CD8-positive T-cells was not correlated to MHC class 1 expression on tumor cells and only a fraction of T-cells showed Granzym B expression. Melanoma BM had significantly less accumulation of peritumoral microglia than NSCLC BM. The inflammatory pattern was independent from treatment of patients with glucocorticoids or radiation. The inflammatory reaction to BM is mainly characterized by activation of microglia/macrophages and shows pronounced upregulation of markers involved in phagocytosis, but seem to be insufficient in activating adaptive immunity. Treatment strategies aimed at activating specific immunity may potentiate immune attack on tumor cells.



Similar content being viewed by others
Abbreviations
- BM:
-
Brain metastases
- BRAF:
-
v-RAF murine sarcoma viral oncogene homolog B1
- CTL4:
-
Anti cytotoxic T lymphocyte associated antigen 4
- NSCLC:
-
Non-small cell lung cancer
- EGFR:
-
Epithelial growth factor receptor
- HER2:
-
In human epidermal growth factor receptor 2
- CNS:
-
Central nervous system
- SCLC:
-
Small cell lung cancer
- MHC II:
-
Major histocompatibility antigen class II
- MHC I:
-
Major histocompatibility antigen class I
- WBRT:
-
Whole-brain radiation therapy
- NO:
-
Nitric oxide
- APM:
-
Antigen-processing machingery
- TGF-beta:
-
Transforming growth factor beta
- GFAP:
-
Glial fibrillary acidic protein
- IBA-1:
-
Ionized calcium binding adaptor molecule 1
- AIF-1:
-
Allograft inflammatory factor 1
- SIGLEC-11:
-
Sialic acid-binding Ig-like lectin 11
- HMGB1:
-
High-mobility group box 1
- iNOS:
-
Inducible nitric oxide synthase
- NCF-1:
-
Neutrophil cytosolic factor 1
References
Kamar FG, Posner JB (2010) Brain metastases. Semin Neurol 30(3):217–235
Tsao M, Xu W, Sahgal A (2012) A meta-analysis evaluating stereotactic radiosurgery, whole-brain radiotherapy, or both for patients presenting with a limited number of brain metastases. Cancer 118(9):2486–2493
Kienast Y, Winkler F (2010) Therapy and prophylaxis of brain metastases. Expert Rev Anticancer Ther 10(11):1763–1777
Gaspar L, Scott C, Rotman M, Asbell S, Phillips T, Wasserman T, McKenna WG, Byhardt R (1997) Recursive partitioning analysis (RPA) of prognostic factors in three Radiation Therapy Oncology Group (RTOG) brain metastases trials. Int J Radiat Oncol Biol Phys 37(4):745–751
Sperduto PW, Kased N, Roberge D, Xu Z, Shanley R, Luo X, Sneed PK, Chao ST, Weil RJ, Suh J, Bhatt A, Jensen AW, Brown PD, Shih HA, Kirkpatrick J, Gaspar LE, Fiveash JB, Chiang V, Knisely JP, Sperduto CM, Lin N, Mehta M (2012) Summary report on the graded prognostic assessment: an accurate and facile diagnosis-specific tool to estimate survival for patients with brain metastases. J Clin Oncol 30(4):419–425
Mehta MP, Paleologos NA, Mikkelsen T, Robinson PD, Ammirati M, Andrews DW, Asher AL, Burri SH, Cobbs CS, Gaspar LE, Kondziolka D, Linskey ME, Loeffler JS, McDermott M, Olson JJ, Patchell RA, Ryken TC, Kalkanis SN (2010) The role of chemotherapy in the management of newly diagnosed brain metastases: a systematic review and evidence-based clinical practice guideline. J Neurooncol 96(1):71–83
Soffietti R, Cornu P, Delattre JY, Grant R, Graus F, Grisold W, Heimans J, Hildebrand J, Hoskin P, Kalljo M, Krauseneck P, Marosi C, Siegal T, Vecht C (2006) EFNS Guidelines on diagnosis and treatment of brain metastases: report of an EFNS Task Force. Eur J Neurol 13(7):674–681
Preusser M, Capper D, Ilhan-Mutlu A, Berghoff AS, Birner P, Bartsch R, Marosi C, Zielinski C, Mehta MP, Winkler F, Wick W, von Deimling A (2012) Brain metastases: pathobiology and emerging targeted therapies. Acta Neuropathol 123(2):205–222
Margolin K, Ernstoff MS, Hamid O, Lawrence D, McDermott D, Puzanov I, Wolchok JD, Clark JI, Sznol M, Logan TF, Richards J, Michener T, Balogh A, Heller KN, Hodi FS (2012) Ipilimumab in patients with melanoma and brain metastases: an open-label, phase 2 trial. Lancet Oncol 13(5):459–465
Sriram S (2011) Role of glial cells in innate immunity and their role in CNS demyelination. J Neuroimmunol 239(1–2):13–20
Graeber MB, Scheithauer BW, Kreutzberg GW (2002) Microglia in brain tumors. Glia 40(2):252–259
Graeber MB, Streit WJ (2010) Microglia: biology and pathology. Acta Neuropathol 119(1):89–105
Amor S, Puentes F, Baker D, van der Valk P (2010) Inflammation in neurodegenerative diseases. Immunology 129(2):154–169
He BP, Wang JJ, Zhang X, Wu Y, Wang M, Bay BH, Chang AY (2006) Differential reactions of microglia to brain metastasis of lung cancer. Mol Med 12(7–8):161–170
Zhang M, Olsson Y (1995) Reactions of astrocytes and microglial cells around hematogenous metastases of the human brain. Expression of endothelin-like immunoreactivity in reactive astrocytes and activation of microglial cells. J Neurol Sci 134(1–2):26–32
Mittelbronn M, Platten M, Zeiner P, Dombrowski Y, Frank B, Zachskorn C, Harter PN, Weller M, Wischhusen J (2011) Macrophage migration inhibitory factor (MIF) expression in human malignant gliomas contributes to immune escape and tumour progression. Acta Neuropathol 122(3):353–365
Mittelbronn M, Simon P, Loffler C, Capper D, Bunz B, Harter P, Schlaszus H, Schleich A, Tabatabai G, Goeppert B, Meyermann R, Weller M, Wischhusen J (2007) Elevated HLA-E levels in human glioblastomas but not in grade I to III astrocytomas correlate with infiltrating CD8+ cells. J Neuroimmunol 189(1–2):50–58
Roth P, Eisele G, Weller M (2012) Immunology of brain tumors. Handb Clin Neurol 104:45–51
Dunn GP, Fecci PE, Curry WT (2012) Cancer immunoediting in malignant glioma. Neurosurgery. doi:10.1227/NEU.0b013e31824f840d
Neves S, Mazal PR, Wanschitz J, Rudnay AC, Drlicek M, Czech T, Wustinger C, Budka H (2001) Pseudogliomatous growth pattern of anaplastic small cell carcinomas metastatic to the brain. Clin Neuropathol 20(1):38–42
De Vleeschouwer S, Fieuws S, Rutkowski S, Van Calenbergh F, Van Loon J, Goffin J, Sciot R, Wilms G, Demaerel P, Warmuth-Metz M, Soerensen N, Wolff JE, Wagner S, Kaempgen E, Van Gool SW (2008) Postoperative adjuvant dendritic cell-based immunotherapy in patients with relapsed glioblastoma multiforme. Clin Cancer Res 14(10):3098–3104
Weber JS, Amin A, Minor D, Siegel J, Berman D, O’Day SJ (2011) Safety and clinical activity of ipilimumab in melanoma patients with brain metastases: retrospective analysis of data from a phase 2 trial. Melanoma Res 21(6):530–534
Finocchiaro G, Pellegatta S (2011) Immunotherapy for glioma: getting closer to the clinical arena? Curr Opin Neurol 24(6):641–647
Dringen R (2005) Oxidative and antioxidative potential of brain microglial cells. Antioxid Redox Signal 7(9–10):1223–1233
Daginakatte GC, Gutmann DH (2007) Neurofibromatosis-1 (Nf1) heterozygous brain microglia elaborate paracrine factors that promote Nf1-deficient astrocyte and glioma growth. Hum Mol Genet 16(9):1098–1112
Brantley EC, Guo L, Zhang C, Lin Q, Yokoi K, Langley RR, Kruzel E, Maya M, Kim SW, Kim SJ, Fan D, Fidler IJ (2010) Nitric oxide-mediated tumoricidal activity of murine microglial cells. Transl Oncol 3(6):380–388
Dal Bianco A, Bradl M, Frischer J, Kutzelnigg A, Jellinger K, Lassmann H (2008) Multiple sclerosis and Alzheimer’s disease. Ann Neurol 63(2):174–183
Haider L, Fischer MT, Frischer JM, Bauer J, Hoftberger R, Botond G, Esterbauer H, Binder CJ, Witztum JL, Lassmann H (2011) Oxidative damage in multiple sclerosis lesions. Brain 134(Pt 7):1914–1924
Kamata T (2009) Roles of Nox1 and other Nox isoforms in cancer development. Cancer Sci 100(8):1382–1388
Murata J, Corradin SB, Janzer RC, Juillerat-Jeanneret L (1994) Tumor cells suppress cytokine-induced nitric-oxide (NO) production in cerebral endothelial cells. Int J Cancer 59(5):699–705
Noda M, Seike T, Fujita K, Yamakawa Y, Kido M, Iguchi H (2009) The role of immune cells in brain metastasis of lung cancer cells and neuron-tumor cell interaction. Ross Fiziol Zh Im I M Sechenova 95(12):1386–1396
Strik HM, Stoll M, Meyermann R (2004) Immune cell infiltration of intrinsic and metastatic intracranial tumours. Anticancer Res 24(1):37–42
Huang Y, Yin H, Han J, Huang B, Xu J, Zheng F, Tan Z, Fang M, Rui L, Chen D, Wang S, Zheng X, Wang CY, Gong F (2007) Extracellular hmgb1 functions as an innate immune-mediator implicated in murine cardiac allograft acute rejection. Am J Transplant 7(4):799–808
Galon J, Costes A, Sanchez-Cabo F, Kirilovsky A, Mlecnik B, Lagorce-Pages C, Tosolini M, Camus M, Berger A, Wind P, Zinzindohoue F, Bruneval P, Cugnenc PH, Trajanoski Z, Fridman WH, Pages F (2006) Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science 313(5795):1960–1964
de Jong RA, Leffers N, Boezen HM, ten Hoor KA, van der Zee AG, Hollema H, Nijman HW (2009) Presence of tumor-infiltrating lymphocytes is an independent prognostic factor in type I and II endometrial cancer. Gynecol Oncol 114(1):105–110
Leffers N, Gooden MJ, de Jong RA, Hoogeboom BN, ten Hoor KA, Hollema H, Boezen HM, van der Zee AG, Daemen T, Nijman HW (2009) Prognostic significance of tumor-infiltrating T-lymphocytes in primary and metastatic lesions of advanced stage ovarian cancer. Cancer Immunol Immunother 58(3):449–459
Robert C, Thomas L, Bondarenko I, O’Day S, M DJ, Garbe C, Lebbe C, Baurain JF, Testori A, Grob JJ, Davidson N, Richards J, Maio M, Hauschild A, Miller WH, Jr., Gascon P, Lotem M, Harmankaya K, Ibrahim R, Francis S, Chen TT, Humphrey R, Hoos A, Wolchok JD (2011) Ipilimumab plus dacarbazine for previously untreated metastatic melanoma. N Engl J Med 364(26): 2517–2526
Loeffler C, Dietz K, Schleich A, Schlaszus H, Stoll M, Meyermann R, Mittelbronn M (2011) Immune surveillance of the normal human CNS takes place in dependence of the locoregional blood-brain barrier configuration and is mainly performed by CD3(+)/CD8(+) lymphocytes. Neuropathology 31(3):230–238
Bradl M, Hohlfeld R (2003) Molecular pathogenesis of neuroinflammation. J Neurol Neurosurg Psychiatry 74(10):1364–1370
Prins RM, Soto H, Konkankit V, Odesa SK, Eskin A, Yong WH, Nelson SF, Liau LM (2011) Gene expression profile correlates with T-cell infiltration and relative survival in glioblastoma patients vaccinated with dendritic cell immunotherapy. Clin Cancer Res 17(6):1603–1615
Roth P, Mittelbronn M, Wick W, Meyermann R, Tatagiba M, Weller M (2007) Malignant glioma cells counteract antitumor immune responses through expression of lectin-like transcript-1. Cancer Res 67(8):3540–3544
Mehling M, Simon P, Mittelbronn M, Meyermann R, Ferrone S, Weller M, Wiendl H (2007) WHO grade associated downregulation of MHC class I antigen-processing machinery components in human astrocytomas: does it reflect a potential immune escape mechanism? Acta Neuropathol 114(2):111–119
Schneider T, Becker A, Ringe K, Reinhold A, Firsching R, Sabel BA (2008) Brain tumor therapy by combined vaccination and antisense oligonucleotide delivery with nanoparticles. J Neuroimmunol 195(1–2):21–27
Pekny M, Nilsson M (2005) Astrocyte activation and reactive gliosis. Glia 50(4):427–434
Unanue ER (2002) Perspective on antigen processing and presentation. Immunol Rev 185:86–102
Stam NJ, Vroom TM, Peters PJ, Pastoors EB, Ploegh HL (1990) HLA-A- and HLA-B-specific monoclonal antibodies reactive with free heavy chains in western blots, in formalin-fixed, paraffin-embedded tissue sections and in cryo-immuno-electron microscopy. Int Immunol 2(2):113–125
Mattiace LA, Davies P, Dickson DW (1990) Detection of HLA-DR on microglia in the human brain is a function of both clinical and technical factors. Am J Pathol 136(5):1101–1114
Martinez-Pomares L, Platt N, McKnight AJ, da Silva RP, Gordon S (1996) Macrophage membrane molecules: markers of tissue differentiation and heterogeneity. Immunobiology 195(4–5):407–416
Ito D, Imai Y, Ohsawa K, Nakajima K, Fukuuchi Y, Kohsaka S (1998) Microglia-specific localisation of a novel calcium binding protein, Iba1. Brain Res Mol Brain Res 57(1):1–9
Ahmed Z, Shaw G, Sharma VP, Yang C, McGowan E, Dickson DW (2007) Actin-binding proteins coronin-1a and IBA-1 are effective microglial markers for immunohistochemistry. J Histochem Cytochem 55(7):687–700
Avril T, Attrill H, Zhang J, Raper A, Crocker PR (2006) Negative regulation of leucocyte functions by CD33-related siglecs. Biochem Soc Trans 34(Pt 6):1024–1027
Bien CG, Bauer J, Deckwerth TL, Wiendl H, Deckert M, Wiestler OD, Schramm J, Elger CE, Lassmann H (2002) Destruction of neurons by cytotoxic T-cells: a new pathogenic mechanism in Rasmussen’s encephalitis. Ann Neurol 51(3):311–318
Payne J, Maher F, Simpson I, Mattice L, Davies P (1997) Glucose transporter Glut 5 expression in microglial cells. Glia 21(3):327–331
Smith KJ, Lassmann H (2002) The role of nitric oxide in multiple sclerosis. Lancet Neurol 1(4):232–241
Bruce-Keller AJ, Gupta S, Parrino TE, Knight AG, Ebenezer PJ, Weidner AM, LeVine H 3rd, Keller JN, Markesbery WR (2010) NOX activity is increased in mild cognitive impairment. Antioxid Redox Signal 12(12):1371–1382
Janeway CA (2002) Immunology, 5 edn. Garland Publishing, Heidelberg
Rousalova I, Krepela E (2010) Granzyme B-induced apoptosis in cancer cells and its regulation (review). Int J Oncol 37(6):1361–1378
Acknowledgments
We thank Irene Leisser and Marianne Leisser for technical assistance with preparation of tissue specimens. This study was performed within the PhD thesis project of Anna Sophie Berghoff in the PhD program “Clinical Neuroscience (CLINS)” at the Medical University Vienna.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Berghoff, A.S., Lassmann, H., Preusser, M. et al. Characterization of the inflammatory response to solid cancer metastases in the human brain. Clin Exp Metastasis 30, 69–81 (2013). https://doi.org/10.1007/s10585-012-9510-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-012-9510-4