Abstract
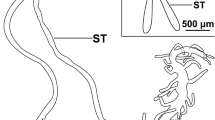
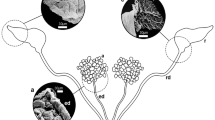
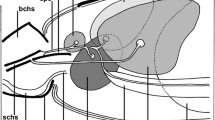
The present study on the salivary glands of semi-engorged Amblyomma cajennense females has identified the various cell types present in this tissue and allowed its morphohistochemical characterization. Marking techniques were applied to detect polysaccharides (PAS), proteins (bromophenol blue), lipids (Nile blue) and calcium (von Kossa), as well as those of scanning electron microscopy (SEM) and transmission electron microscopy (TEM). The results obtained by TEM showed and confirmed that these individuals’ glands are also formed by round acini that are connected to the common excretory duct through acinar and intermediate ducts. Histological data as well as ultrastructural ones showed that the glands are formed by types I, II and III acini. In this study with salivary glands polysaccharides, proteins, lipids and calcium were observed in the cytoplasm and/or cell secretion granules—both free or forming complexes, as the intensity of the marking varied according to the cell as well as the type of acini analyzed, showing the structural and functional complexity of the tick salivary glands, characteristics that give the multifunctional character to this organ.





Similar content being viewed by others
References
Alberts B, Johnson A, Lewis J, Raff M, Roberts K, Walter P (2004) Biologia Molecular da Célula. Editora ArtMed, Porto Alegre
Balashov YS (1972) Bloodsucking ticks (Ixodidae)—vectors of disease of man and animals, vol 8. Miscellaneous Publications of the Entomological Society of America, Washington
Balashov YS (1983) The female reproductive system. In: Balashov YS (ed) An atlas of ixodid tick ultrastructure. Entomological Society of America, Russian, pp 98–128
Barker DM, Ownby CL, Krolak JM, Claypool PL, Sauer JR (1984) The effects of attachment, feeding and mating on the morphology of the type I alveolus of salivary glands of the lone star tick, Amblyomma americanum. Int J Parasitol 70:99–113
Binnington KC (1978) Sequential changes in salivary gland structure during attachment and feeding of the cattle tick, Boophilus microplus. Int J Parasitol 8:97–115
Bowan AS, Coons LB, Needham GR, Sauer JR (1997) Tick saliva: recent advances and implications for vector competence. Med Vet Entomol 11:277–285
Caperucci DC, Camargo-Mathias MI (2006) Lipids in oocytes of Neoponera villosa ants (Hymenoptera: Ponerinae). Sociobiology 47:531–542
Denardi SE, Bechara GH, Oliveira PR, Nunes ET, Saito KC, Camargo Mathias CM (2004) Morphological characterization of the ovary and vitellogenesis dynamics in the tick Amblyomma cajennense (Acari: Ixodidae). Vet Parasitol 125:379–395
Denardi SE, Camargo-Mathias MI, Bechara GH (2006) Amblyomma cajennense (Acari: Ixodidade): salivary gland cells of partially engorged females ticks and the production of lipid by their mitochondria. Exp Parasitol 113:30–35
Fawcett DW, Binnington K, Voigt WP (1986) The cell biology of the ixodid tick salivary gland. In: Sauer JR, Hair A (eds) Morphology, physiology, and behavioral biology of ticks. Ellis Horwood, Chichester, pp 22–45
Gill HS, Walker AR (1987) The salivary glands of Hyalomma anatolicum anatolicum: structural changes during attachment and feeding. Int J Parasitol 17(8):1381–1392
Junqueira LCU, Junqueira LMMS (1983) Técnicas básicas de citologia e histologia. Editora Santos, São Paulo
Lavoipierre MMJ, Riek RF (1955) Observations on the feeding habits of argasid ticks and on the effect of their bites on laboratory animals, together with a note on the production of coxal fluid by several of the species studied. Ann Trop Med Parasit 49:96–113
Lison LL (1960) Histochimie et Cytochimie Animales. Gauthier-Villars, Éditeur
Marzouk AS, Darwish ZEA (1994) Changes in the salivary glands of female Hyalomma (Hyalomma) dromedarii during and after feeding. J Egypt Soc Parasitol 24(1):39–57
McManus JFA (1946) Histological demonstration of mucin after periodic acid. Nature 158:202
McMullen HL, Sauer JR, Burton RL (1976) Possible role in uptake of water vapour by ixodid tick salivary glands. J Insect Physiol 22:1281–1285
Nunes ET, Bechara GH, Saito KC, Denardi SE, Camargo-Mathias MI (2005) Morphological, histological, and ultrastructural characterization of degenerating salivary glands in females of the cattle-tick Rhipicephalus (Boophilus) microplus (Canestrini, 1887) (Acari: Ixodidae). Micron 36:437–447
Nunes ET, Camargo Mathias MI, Bechara GH (2006) Rhipicephalus (Boophilus) microplus (Canestrini, 1887) (Acari: Ixodidae): Acid phosphatase and ATPase activity localization in salivary glands of females during the feeding period. Exp Parasitol 114:109–117
Nunes PH, Bechara GH, Camargo Mathias MI (2008) Morphological changes in the salivary glands of Amblyomma cajennense females (Acari: Ixodidae) in different feeding stages on rabbits at first infestation. Exp Appl Acarol 45:199–209
Nuttal GHF, Strickland C (1908) On the presence of an anti-coagulin in the salivary glands and the intestines of Argas persicus. Parasitology 1:300–310
Paesen GC, Adams PL, Harlos K, Nuttall PA, Stuart DI (1999) Tick histamine-binding proteins: isolation, cloning and three-dimensional structure. Mol Cell 3(5):661–671
Pawlowsky EN, Chodukin NJ (1929) Ueber die Antikoaguline und andere wirksame Bestandteile der Zecke Ornithodorus papillipes. Bir Z Parasitenk 2:90–96
Pearse AGE (1985) Histochemistry. Theoretical and applied, 4th edn. J&A Churchill, London
Pereira CPM, Oliveira PR, Furquim KCS, Bechara GH, Camargo-Mathias MI (2009) Effects of fipronil (active ingredient of Frontline1) on salivary gland cells of Rhipicephalus sanguineus females (Latreille, 1806) (Acari: Ixodidae). Vet Parasitol 166:124–130
Ranade V (1933) On the cytoplasmic inclusions in the oogenesis of Periplaneta americana. Linn Allahad University Sci Bull 9:85–121
Ribeiro JMC, Mather TN (1998) Ixodes scapularis: salivary kininase activity is a metallo-dipeptidyl carboxypeptidase. Exp Parasitol 89:213–221
Ribeiro JMC, Makoul GT, Levine J, Robinson DR, Spielman A (1985) Antihemostatic, anti-inflammatory and immunosuppressive properties of the saliva of a tick, Ixodes dammini. J Exp Med 161:332–344
Rudolf D, Knulle W (1974) Site of the mechanism of water vapour uptake from the atmosphere in ixodid ticks. Nature 249:84–85
Sauer JR (1977) Acarine salivary glands—physiological relationships. J Med Entomol 14:1–9
Schumaker TT, Serra-Freire NM (1991) Histologia das glândulas salivares de adultos de Argas (Persicargas) miniatus (Koch, 1844) (Ixodoidea: Argasidae) em jejum, em alimentação e alimentados. Rev Bras Entomol 35:49–72
Serra-Freire NM, Olivieri JA (1993) Structure of the salivary glands of the unfed male tick Amblyomma cajennense (Fabricius) (Acari: Ixodidae). Mem Inst Oswaldo Cruz 88(2):207–213
Sonenshine DE (1991) Biology of ticks. Oxford University Press, New York
Till WM (1961) A contribution to the anatomy and histology of the brown ear tick Rhipicephalus appendiculatus Neumann, vol 6. Memoirs of the Entomological Society of Southern Africa, Pretoria
Vidal BC, Mello MLS (1987) Biologia celular. Livraria Atheneu, Rio de Janeiro
Walker AR, Fletcher JD, Gill H (1985) Structural and histochemical changes in the salivary glands of Rhipicephalus appendiculatus during feeding. Int J Parasitol 15(1):81–100
Wikel SK (1996) Host immunity to ticks. Ann Rev Entomol 41:1–22
Wikel SK (1999) Tick modulation of host immunity: an important factor in pathogen transmission. Int J Parasitol 29:851–859
Acknowledgments
We are indebted to FAPESP (Fundação de Amparo à Pesquisa do Estado de São Paulo) for grants number 07/59020-0 and to CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico). Authors thank Mr. Gérson de Mello Sousa and Cristiane M. Mileo for their technical assistance. Part of this work has been facilitated through the Integrated Consortium on Ticks and Tick-borne Diseases (ICTTD-3) supported by the European Union under contract number 510561-INCO.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Denardi, S.E., Furquim, K.C.S., Bechara, G.H. et al. Salivary glands of Amblyomma cajennense (Acari: Ixodidae): a histological and an ultrastructural overview. Exp Appl Acarol 54, 177–189 (2011). https://doi.org/10.1007/s10493-010-9421-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10493-010-9421-5