Abstract
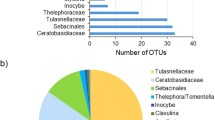
Some orchid species are present as epiphytes and lithophytes in the same habitat, but little is known about the differences of their mycorrhizal fungal communities. We used Coelogyne viscosa, which occurs both as an epiphyte and a lithophyte, as a study system to investigate orchid mycorrhizal fungal communities in lithophytes and epiphytes in Xishuangbanna National Nature Reserve (Yunnan Province, China). Twenty-three fungal operational taxonomic units (OTUs) from 18 sampling sites were identified. Results indicated that mycorrhizal fungal community composition was different between epi- and lithophytes. When we analyzed the Tulasnellaceae and Sebacinales communities separately, we found that the Sebacinales fungal communities were significantly different in the two growth habitats, but the Tulasnellaceae fungal communities were not. Our results provide evidence for distinct orchid mycorrhiza fungal communities depending on the growth habitat of the orchid. Consistent with some recent investigations of mycorrhizal fungus community composition, this study suggests that for one orchid, growth habitat affects mycorrhizal symbioses.



Similar content being viewed by others
References
Anderson MJ, Gorley RN, Clarke KR (2008) PERMANOVA+ for PRIMER: guide to software and statistical methods. PRIMER-E, Plymouth
Barrett CF, Freudenstein JV, Taylor DL, Kōljalg U (2010) Rangewide analysis of fungal associations in the fully mycoheterotrophic Corallorhiza striata complex (Orchidaceae) reveals extreme specificity on ectomycorrhizal Tomentella (Thelephoraceae) across North America. Am J Bot 97:628–643. doi:10.3732/ajb.0900230
Bray JR, Curtis JT (1957) An ordination of the upland forest communities of southern Wisconsin. Ecol Monogr 27:325–349. doi:10.2307/1942268
Brundrett MC (2007) Scientific approaches to Australian temperate terrestrial orchid conservation. Aust J Bot 55:293–307. doi:10.1071/BT06131
Brundrett MC, Scade A, Batty AL, Dixon KW, Sivasithamparam K (2003) Development of in situ and ex situ seed baiting techniques to detect mycorrhizal fungi from terrestrial orchid habitats. Mycol Res 10:1210–1220. doi:10.1017/S09537562030008463
Cameron DD, Leake JR, Read DJ (2006) Mutualistic mycorrhiza in orchids: evidence from plant-fungus carbon and nitrogen transfers in the green-leaved terrestrial orchid Goodyera repens. New Phytol 171:405–416. doi:10.1111/j.1469-8137.2006.01767.x
Chung MY, Park CW, Chung MG (2007) Extremely low levels of allozyme variation in southern Korean populations of the two rare and endangered lithophytic or epiphytic Bulbopyllum drymoglossum and Sarcanthus scolopendrifolius (Orchidaceae): implications for conservation. Biodivers Conserv 16:775–786. doi:10.1007/s10531-005-6200-4
Darriba D, Taboada GL, Doallo R, Posada D (2012) jModelTest 2: more models, new heuristics and parallel computing. Nat Methods 9:772. doi:10.1038/nmeth.2109
Dearnaley JDW (2007) Further advances in orchid mycorrhizal research. Mycorrhiza 17:475–486. doi:10.1007/s00572-007-0138-1
Dearnaley JDW, Martos F, Selosse MA (2012) Orchid mycorrhizas: molecular ecology, physiology, evolution and conservation aspects. Fungal Assoc 9:207–230. doi:10.1007/978-3-642-30826-0_12
Gómez NR, Tremblay R, Ackerman EM (2006) Distribution of life cycle stages in a lithophytic and epiphytic orchid. Folia Geobot 41:107–120. doi:10.1007/BF02805265
Gravendeel B, Smithson A, Slik FJW, Schuiteman A (2004) Epiphytism and pollinator specialization: drivers for orchid diversity? Philos Trans R Soc B 359:1523–1535. doi:10.1098/rstb.2004.1529
Jacquemyn H, Honnay O, Cammue BPA, Brys R, Lievens B (2010) Low specificity and nested subset structure characterize mycorrhizal associations in five closely related species of the genus Orchis. Mol Ecol 19:4086–4095. doi:10.1111/j.1365-294X.2010.04785.x
Jacquemyn H, Merckx V, Brys R et al (2011) Analysis of network architecture reveals phylogenetic constraints on mycorrhizal specificity in the genus Orchis (Orchidaceae). New Phytol 192:518–528. doi:10.1111/j.1469-8137.2011.03796.x
Jacquemyn H, Brys R, Honnay O, Roldán-Ruiz I, Lievens B, Wiegand T (2012a) Nonrandom spatial structuring of orchids in a hybrid zone of three Orchis species. New Phytol 193:454–464. doi:10.1111/j.1469-8137.2011.03913.x
Jacquemyn H, Brys R, Lievens B, Wiegand T (2012b) Spatial variation in below-ground seed germination and divergent mycorrhizal associations correlate with spatial segregation of three co-occurring orchid species. J Ecol 100:1328–1337. doi:10.1111/j.1365-2745.2012.01998.x
Jacquemyn H, Deja A, De Hert K, Bailarote BC, Lievens B (2012c) Variation in mycorrhizal associations with Tulasnelloid fungi among populations of five Dactylorhiza species. Plos One 7:e42212. doi:10.1371/journal.pone.0042212
Jacquemyn H, Brys R, Merckx VSFT, Waud M, Lievens B, Wiegand T (2014) Coexisting orchid species have distinct mycorrhizal communities and display strong spatial segregation. New Phytol 202:616–627. doi:10.1111/nph.12640
Jin XH (2005) Generic delimitation and a new infrageneric system in the genus Holcoglossum (Orchidaceae: Aeridinae). Bot J Linn Soc 149:465–468. doi:10.1111/j.1095-8339.2005.00463.x
Joppa LN, Roberts DL, Pimm SL (2011) How many species of flowering plants are there? Proc Roy Soc B 278:554–559. doi:10.1098/rspb.2010.1004
Kristiansen KA, Freudenstein JV, Rasmussen FN, Rasmussen HN (2004) Molecular identification of mycorrhizal fungi in Neuwiedia veratrifolia (Orchidaceae). Mol Phylogenet Evol 33:251–258. doi:10.1016/j.ympev.2004.05.015
Larkin MA, Blackshields G, Brown NP et al (2007) Clustal W and Clustal X version 2.0. Bioinformatics 23:2947–2948. doi:10.1093/bioinformatics/btm404
Losos JB (2008) Phylogenetic niche conservatism, phylogenetic signal and the relationship between phylogenetic relatedness and ecological similarity among species. Ecol Lett 11:995–1007. doi:10.1111/j.1461-0248.01229.x
Lozupone C, Knight R (2005) UniFrac: a new phylogenetic method for comparing microbial communities. Appl Environ Microb 71:8228–8235. doi:10.1128/AEM.71.12.8228-8235.2005
Lozupone C, Hamady M, Knight R (2006) UniFrac: an online tool for comparing microbial community diversity in a phylogenetic context. BMC Bioinformat 7:371. doi:10.1186/1471-2105-7-371
Magurran AE (1988) Ecological diversity and its measurement. Princeton University Press, Princeton
Maherali H, Klironomos JN (2007) Influence of phylogeny on fungal community assembly and ecosystem functioning. Science 316:1746–1748. doi:10.1126/science.1143082
Martos F, Munoz F, Pailler T, Kottke I, Gonneau C, Selosse MA (2012) The role of epiphytism in architecture and evolutionary constraint within mycorrhizal networks of tropical orchids. Mol Ecol 21:5098–5109. doi:10.1111/j.1365-294X.2012.05692.x
McCormick MK, Whigham DF, O’Neill J (2004) Mycorrhizal diversity in photosynthetic terrestrial orchids. New Phytol 163:425–438. doi:10.1111/j.1469-8137.2004.01114.x
McCormick MK, Whigham DF, Sloan D, O’Malley K, Hodkinson B (2006) Orchid-fungus fidelity: a marriage meant to last? Ecology 87:903–911. doi:10.1890/0012-9658
McCormick MK, Whigham DF, O’Neill JP et al (2009) Abundance and distribution of Corallorhiza odontorhiza reflect variations in climate and ectomycorrhizae. Ecol Monogr 79:619–635. doi:10.1890/08-0729.1
McCormick MK, Taylor DL, Uhaszova KJ, Burnett RK, Whigham DF, O’Neill JP (2012) Limitations on orchid recruitment: not a simple picture. Mol Ecol 21:1511–1523. doi:10.1111/j.1365-294X.2012.05468.x
McCune B, Mefford MJ (2011) PC-ORD. Multivariate analysis of ecological data. Version 6. MjM Software Design, Gleneden Beach
Otero JT, Ackerman J, Bayman P (2002) Diversity and host specificity of endophytic Rhizoctonia-like fungi from tropical orchids. Am J Bot 89:1852–1858. doi:10.3732/ajb.89.11.1852
Otero JT, Bayman P, Ackerman JD (2005) Variation in mycorrhizal performance in an epiphytic orchid: the potential for natural selection. Evol Ecol 19:29–43. doi:10.1007/s10682-004-5441-0
Pandey M, Sharma J, Taylor DL, Yadon VL (2013) A narrowly endemic photosynthetic orchid is non-specific in its mycorrhizal associations. Mol Ecol 22:2341–2354. doi:10.1111/mec.12249
Peay KG, Kennedy PG, Davies SJ, Tan S, Bruns TD (2010) Potential link between plant and fungal distributions in a dipterocarp rainforest: community and phylogenetic structure of tropical ectomycorrhizal fungi across a plant and soil ecotone. New Phytol 185:529–542. doi:10.1111/j.1469-8137.2009.03075.x
Rasmussen HN, Rasmussen FN (2009) Orchid mycorrhiza: implications of a mycophagous life style. Oikos 118:334–345. doi:10.1111/j.1600-0706.2008.17116.x
Schödelbauerová I, Tremblay RL, Kindlmann P (2010) Predication vs. reality: can a PVA model predict population persistence 13 years later? Biodivers Conserv 19:637–650. doi:10.1007/s10531-009-9724-1
Shefferson RP, Weiß M, Kull T, Taylor DL (2005) High specificity generally characterizes mycorrhizal association in rare lady’s slipper orchids, genus Cypripedium. Mol Ecol 14:613–626. doi:10.1111/j.1365-294X.2005.02424.x
Shefferson RP, Taylor DL, Weiß M et al (2007) The evolutionary history of mycorrhizal specificity among lady’s slipper orchids. Evolution 61:1380–1390. doi:10.1111/j.1558-5646.2007.00112.x
Shefferson RP, Kull T, Tali K (2008) Mycorrhizal interactions of orchids colonizing Estonian mine tailings hills. Am J Bot 95:156–164. doi:10.3732/ajb.95.2.156
Shefferson RP, Cowden CC, McCormick MK, Yukawa T, Ogura-Tsujita Y, Hashimoto T (2010) Evolution of host breadth in broad interactions: mycorrhizal specificity in East Asian and North American rattlesnake plantains (Goodyera spp.) and their fungal hosts. Mol Ecol 19:3008–3017. doi:10.1111/j.1365-294X.2010.04693.x
Swarts ND, Sinclair EA, Francis A, Dixon KW (2010) Ecological specialization in mycorrhizal symbiosis leads to rarity in an endangered orchid. Mol Ecol 19:3226–3242. doi:10.1111/j.1365-294X.2010.04736.x
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28:2731–2739. doi:10.1093/molbev/msr121
Taylor DL, McCormick MK (2008) Internal transcribed spacer primers and sequences for improved characterization of basidiomycetous orchid mycorrhizas. New Phytol 177:1020–1033. doi:10.1111/j.1469-8137.2007.02320.x
Taylor DL, Bruns TD, Leake JR, Read DJ (2002) Mycorrhizal specificity and function in myco-heterotrophic plants. In: Sanders I, van der Hijden M (eds) Mycorrhizal ecology, vol 157. Springer, Berlin, pp 375–413
Waterman RJ, Bidartondo MI, Stofberg J et al (2011) The effects of above- and belowground mutualisms on orchid speciation and coexistence. Am Nat 117:E54–E68. doi:10.1086/657955
White TJ, Bruns TD, Lee SB, Taylor JW (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols: a guide to methods and applications. Academic, San Diego
Yuan L, Yang ZL, Li SY, Hu H, Huang JL (2010) Mycorrhizal specificity, preference, and plasticity of six slipper orchids from South Western China. Mycorrhiza 20:559–568. doi:10.1007/s00572-010-0307-5
Acknowledgments
This research was financially supported by the National Natural Sciences Foundation of China (No. 81274197). We thank Randy Molina and the two anonymous referees for the useful comments on this manuscript.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendix S1
Spatial distribution of nine sampling sites for lithophytic and epiphytic Coelogyne viscosa, respectively, in a 70 × 60 m plot sampled in Xishuangbanna, Yunnan, China. (DOC 25 kb)
Appendix S2
Maximum-likelihood trees constructed with operational taxonomic unit (OTU) sequences from three fungal taxa, that is, (a) Tulasnellaceae (b) Sebacinales and (c) Ceratobasidiaceae observed in C. viscosa roots collected from lithophytic and epiphytic habitats. Tulasnellaceae trees (a) were rooted Tulasnella violea (GenBank accession number DQ520097). Sebacinaceae (b) and Ceratobasidiaceae (c) were rooted with Sebacina allantoidea (GenBank accession number AF490396) and Botryobasidium subcoronatum (GenBank accession number DQ200924), respectively. Bootstrap values <50 are not shown above nodes. Scale bar in the figure represents estimated number of DNA substitutions per site. Sequences downloaded from GenBank are shown with accession numbers. OTU sequences from this study are shown in Blue. E, epiphytic; L, lithophytic; E + L means this OTU was found in both epiphytic and lithophytic orchids. (DOC 32 kb)
Appendix S3
Frequency distribution of identified operational taxonomic units (OTUs) in epiphytes and lithophytes of Coelogyne viscosa. (a) All plants; (b) Lithophytes only; (c) Epiphytes only. (DOC 24 kb)
Rights and permissions
About this article
Cite this article
Xing, X., Gai, X., Liu, Q. et al. Mycorrhizal fungal diversity and community composition in a lithophytic and epiphytic orchid. Mycorrhiza 25, 289–296 (2015). https://doi.org/10.1007/s00572-014-0612-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00572-014-0612-5