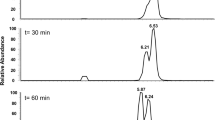
Abstract
The degradation pathways of benzoate at high concentration in Pseudomonas putida P8 were directly elucidated through mass spectrometric identification of some key catabolic enzymes. Proteins from P. putida P8 grown on benzoate or succinate were separated using two-dimensional gel electrophoresis. For cells grown on benzoate, eight distinct proteins, which were absent in the reference gel patterns from succinate-grown cells, were found. All the eight proteins were identified by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry as catabolic enzymes involved in benzoate degradation. Among them, CatB (EC5.5.1.1), PcaI (EC2.8.3.6), and PcaF (EC2.3.1.174) were the enzymes involved in the ortho-cleavage pathway; DmpC (EC1.2.1.32), DmpD (EC3.1.1.-), DmpE (EC4.2.1.80), DmpF (EC1.2.1.10), and DmpG (EC4.1.3.-) were the meta-cleavage pathway enzymes. In addition, enzyme activity assays showed that the activities of both catechol 1,2-dioxygenase (C12D; EC1.13.11.1) and catechol 2,3-dioxygenase (C23D; EC1.13.11.2) were detected in benzoate-grown P. putida cells, undoubtedly suggesting the simultaneous expression of both the ortho- and the meta-cleavage pathways in P. putida P8 during the biodegradation of benzoate at high concentration.


Similar content being viewed by others
References
Aldrich TL, Frantz B, Gill JF, Kilbane JJ, Chakrabarty AM (1987) Cloning and complete nucleotide-sequence determination of the catB gene encoding cis,cis-muconate lactonizing enzyme. Gene 52:185–195
Alexander M (1981) Biodegradation of chemicals of environmental concern. Science 211:132–138
Ampe F, Lindley ND (1996) Flux limitations in the ortho pathway of benzoate degradation of Alcaligenes eutrophus: metabolite overflow and induction of the meta pathway at high substrate concentrations. Microbiology 142:1807–1817
Bhushan B, Halasz A, Hawari M (2005) Biotransformation of CL-20 by a dehydrogenase enzyme from Clostridium sp. EDB2. Appl Microbiol Biotechnol 69:448–455
Bouwer EJ, Zehnder AJB (1993) Bioremediation of organic compounds—putting microbial metabolism to work. Trends Biotechnol 11:360–367
Caldwell ME, Suflita JM (2000) Detection of phenol and benzoate as intermediates of anaerobic benzene biodegradation under different terminal electron-accepting conditions. Environ Sci Technol 34:1216–1220
Cao B, Loh KC (2008) Catabolic pathways and cellular responses of Pseudomonas putida P8 during growth on benzoate with proteomics approach. Biotechnol Bioeng. doi:https://doi.org/10.1002/bit.21991
Denef VJ, Park J, Tsoi TV, Rouillard JM, Zhang H, Wibbenmeyer JA, Verstraete W, Gulari E, Hashsham SA, Tiedje JM (2004) Biphenyl and benzoate metabolism in a genomic context: outlining genome-wide metabolic networks in Burkholderia xenovorans LB400. Appl Environ Microbiol 70:4961–4970
Denef VJ, Patrauchan MA, Florizone C, Park J, Tsoi TV, Verstraete W, Tiedje JM, Eltis LD (2005) Growth substrate- and phase-specific expression of biphenyl, benzoate, and C-1 metabolic pathways in Burkholderia xenovorans LB400. J Bacteriol 187:7996–8005
Feist CF, Hegeman GD (1969) Phenol and benzoate metabolism by Pseudomonas putida: regulation of tangential pathways. J Bacteriol 100:1121–1123
Gescher J, Zaar A, Mohamed M, Schagger H, Fuchs G (2002) Genes coding for a new pathway of aerobic benzoate metabolism in Azoarcus evansii. J Bacteriol 184:6301–6315
Hamzah RY, Albaharna BS (1994) Catechol ring-cleavage in Pseudomonas cepacia—the simultaneous induction of ortho and meta pathways. Appl Microbiol Biotechnol 41:250–256
Harwood CS, Parales RE (1996) The beta-ketoadipate pathway and the biology of self-identity. Annu Rev Microbiol 50:553–590
Iwaki H, Hasegawa Y (2007) Degradation of 2-nitrobenzoate by Burkholderia terrae strain KU-15. Biosci Biotechnol Biochem 71:145–151
Jimenez JI, Minambres B, Garcia JL, Diaz E (2002) Genomic analysis of the aromatic catabolic pathways from Pseudomonas putida KT2440. Environ Microbiol 4:824–841
Kim SI, Song SY, Kim KW, Ho EM, Oh KH (2003) Proteomic analysis of the benzoate degradation pathway in Acinetobacter sp. KS-1. Res Microbiol 154:697–703
Kim SI, Kim JY, Yun SH, Kim JH, Leem SH, Lee C (2004) Proteome analysis of Pseudomonas sp. K82 biodegradation pathways. Proteomics 4:3610–3621
Kim YH, Cho K, Yun S-H, Kim JY, Kwon K-H, Yoo JS, Kim SI (2006) Analysis of aromatic catabolic pathways in Pseudomonas putida KT 2440 using a combined proteomic approach: 2-DE/MS and cleavable isotope-coded affinity tag analysis. Proteomics 6:1301–1318
Krishnan S, Prabhu Y, Phale PS (2004) O-phthalic acid, a dead-end product in one of the two pathways of phenanthrene degradation in Pseudomonas. sp strain PP2. Indian J Biochem Biophys 41:227–232
Kukor JJ, Olsen RH (1991) Genetic organization and regulation of a meta-cleavage pathway for catechols produced from catabolism of toluene, benzene, phenol, and cresols by Pseudomonas pickettii PKO1. J Bacteriol 173:4587–4594
Kurbatov L, Albrecht D, Herrmann H, Petruschka L (2006) Analysis of the proteome of Pseudomonas putida KT2440 grown on different sources of carbon and energy. Environ Microbiol 8:466–478
Lee N, Lee JM, Min KH, Kwon DY (2003) Purification and characterization of 2,3-dihydroxybiphenyl 1,2-dioxygenase from Comamonas sp. SMN4. J Microbiol Biotechnol 13:487–494
Loh KC, Chua SS (2002) Ortho pathway of benzoate degradation in Pseudomonas putida: induction of meta pathway at high substrate concentrations. Enzyme Microb Technol 30:620–626
Manjasetty BA, Powlowski J, Vrielink A (2003) Crvstal structure of a bifunctional aldolase-dehydrogenase: sequestering a reactive and volatile intermediate. Proc Natl Acad Sci U S A 100:6992–6997
Nakazawa T, Nakazawa A (1970) Pyrocatechase (Pseudomonas). Methods Enzymol 17A:518–522
Nozaki M (1970) Metapyrocatechase (Pseudomonas). Methods Enzymol 17A:522–525
Parales RE, Haddock JD (2004) Biocatalytic degradation of pollutants. Curr Opin Biotechnol 15:374–379
Parales RE, Harwood CS (1993) Regulation of the pcaIJ genes for aromatic acid degradation in Pseudomonas putida. J Bacteriol 175:5829–5838
Parales RE, Bruce NC, Schmid A, Wackett LP (2002) Biodegradation, biotransformation, and biocatalysis (B3). Appl Environ Microbiol 68:4699–4709
Shevchenko A, Wilm M, Vorm O, Mann M (1996) Mass spectrometric sequencing of proteins from silver stained polyacrylamide gels. Anal Chem 68:850–858
Shingler V, Franklin FCH, Tsuda M, Holroyd D, Bagdasarian M (1989) Molecular analysis of a plasmid-encoded phenol hydroxylase from Pseudomonas CF600. J Gen Microbiol 135:1083–1092
Shingler V, Powlowski J, Marklund U (1992) Nucleotide-sequence and functional-analysis of the complete phenol/3,4-dimethylphenol catabolic pathway of Pseudomonas sp. strain-CF600. J Bacteriol 174:711–724
Wackett LP (2003) Pseudomonas putida—a versatile biocatalyst. Nat Biotechnol 21:136–138
Wheelis ML, Ornston LN (1972) Genetic control of enzyme induction in the beta-ketoadipate pathway of Pseudomonas putida: deleting mapping of cat mutations. J Bacteriol 109:790–795
Acknowledgments
This research work was supported by a research grant from the Singapore Ministry of Education Academic Research Fund (R-279-000-181-112). The authors gratefully acknowledge the National University of Singapore for providing the research scholarship to Bin Cao.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cao, B., Geng, A. & Loh, KC. Induction of ortho- and meta-cleavage pathways in Pseudomonas in biodegradation of high benzoate concentration: MS identification of catabolic enzymes. Appl Microbiol Biotechnol 81, 99–107 (2008). https://doi.org/10.1007/s00253-008-1728-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-008-1728-3