Abstract
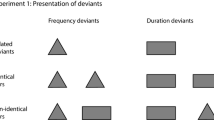
Research on brain mechanisms of deviance detection and sensory memory trace formation, best indexed by the mismatch negativity, mainly relied on the investigation of responses elicited by auditory stimuli. However, comparable less research reported the mismatch negativity elicited by somatosensory stimuli. More importantly, little is known on the functional features of mismatch deviant and standard responses across different sensory modalities. To directly compare different sensory modalities, we adopted a crossmodal roving paradigm and collected event-related potentials elicited by auditory, non-nociceptive somatosensory, and nociceptive trains of stimuli, during Active and Passive attentional conditions. We applied a topographical segmentation analysis to cluster successive scalp topographies with quasi-stable landscape of significant differences to extract crossmodal mismatch responses. We obtained three main findings. First, across different sensory modalities and attentional conditions, the formation of a standard sensory trace became robust mainly after the second stimulus repetition. Second, the neural representation of a modality deviant stimulus was influenced by the preceding sensory modality. Third, the mismatch negativity significantly covaried between Active and Passive attentional conditions within the same sensory modality, but not between different sensory modalities. These findings provide robust evidence that, while different modalities share a similar process of standard trace formation, the process of deviance detection is largely modality dependent.





Similar content being viewed by others
References
Akatsuka K, Wasaka T, Nakata H, Kida T, Hoshiyama M, Tamura Y, Kakigi R (2007) Objective examination for two-point stimulation using a somatosensory oddball paradigm: an MEG study. Clin Neurophysiol 118:403–411. doi:10.1016/j.clinph.2006.09.030
Baldeweg T, Klugman A, Gruzelier J, Hirsch SR (2004) Mismatch negativity potentials and cognitive impairment in schizophrenia. Schizophr Res 69:203–217
Bendixen A, Roeber U, Schroger E (2007) Regularity extraction and application in dynamic auditory stimulus sequences. J Cogn Neurosci 19:1664–1677. doi:10.1162/jocn.2007.19.10.1664
Bullmore ET, Suckling J, Overmeyer S, Rabe-Hesketh S, Taylor E, Brammer MJ (1999) Global, voxel, and cluster tests, by theory and permutation, for a difference between two groups of structural MR images of the brain. IEEE Trans Med Imaging 18:32–42
Butler JS, Molholm S, Fiebelkorn IC, Mercier MR, Schwartz TH, Foxe JJ (2011) Common or redundant neural circuits for duration processing across audition and touch. J Neurosci 31:3400–3406. doi:10.1523/JNEUROSCI.3296-10.2011
Butler JS, Foxe JJ, Fiebelkorn IC, Mercier MR, Molholm S (2012) Multisensory representation of frequency across audition and touch: high density electrical mapping reveals early sensory–perceptual coupling. J Neurosci 32:15338–15344. doi:10.1523/JNEUROSCI.1796-12.2012
Cacace AT, McFarland DJ (2003) Quantifying signal-to-noise ratio of mismatch negativity in humans. Neurosci Lett 341:251–255
Chen TL, Babiloni C, Ferretti A et al (2008) Human secondary somatosensory cortex is involved in the processing of somatosensory rare stimuli: an fMRI study. Neuroimage 40:1765–1771. doi:10.1016/j.neuroimage.2008.01.020
Cowan N, Winkler I, Teder W, Näätänen R (1993) Memory prerequisites of mismatch negativity in the auditory event-related potential (ERP). J Exp Psychol Learn Mem Cogn 19:909–921
Cruccu G, Aminoff MJ, Curio G et al (2008) Recommendations for the clinical use of somatosensory-evoked potentials. Clin Neurophysiol 119:1705–1719. doi:10.1016/j.clinph.2008.03.016
Delorme A, Makeig S (2004) EEGLAB: an open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J Neurosci Methods 134:9–21. doi:10.1016/j.jneumeth.2003.10.009
Doeller CF, Opitz B, Mecklinger A, Krick C, Reith W, Schroger E (2003) Prefrontal cortex involvement in preattentive auditory deviance detection: neuroimaging and electrophysiological evidence. Neuroimage 20:1270–1282. doi:10.1016/S1053-8119(03)00389-6
Garcia-Larrea L, Lukaszewicz AC, Mauguiere F (1995) Somatosensory responses during selective spatial attention: the N120-to-N140 transition. Psychophysiology 32:526–537
Garrido MI, Kilner JM, Kiebel SJ, Stephan KE, Friston KJ (2007) Dynamic causal modelling of evoked potentials: a reproducibility study. Neuroimage 36:571–580. doi:10.1016/j.neuroimage.2007.03.014
Garrido MI, Friston KJ, Kiebel SJ, Stephan KE, Baldeweg T, Kilner JM (2008) The functional anatomy of the MMN: a DCM study of the roving paradigm. Neuroimage 42:936–944. doi:10.1016/j.neuroimage.2008.05.018
Garrido MI, Kilner JM, Kiebel SJ, Friston KJ (2009) Dynamic causal modeling of the response to frequency deviants. J Neurophysiol 101:2620–2631. doi:10.1152/jn.90291.2008
Haenschel C, Vernon DJ, Dwivedi P, Gruzelier JH, Baldeweg T (2005) Event-related brain potential correlates of human auditory sensory memory-trace formation. J Neurosci 25:10494–10501. doi:10.1523/JNEUROSCI.1227-05.2005
Hu L, Zhang ZG, Hung YS, Luk KD, Iannetti GD, Hu Y (2011) Single-trial detection of somatosensory evoked potentials by probabilistic independent component analysis and wavelet filtering. Clin Neurophysiol 122:1429–1439. doi:10.1016/j.clinph.2010.12.052
Hu L, Zhao C, Li H, Valentini E (2013) Mismatch responses evoked by nociceptive stimuli. Psychophysiology 50:158–173. doi:10.1111/psyp.12000
Inui K, Tran TD, Hoshiyama M, Kakigi R (2002) Preferential stimulation of Adelta fibers by intra-epidermal needle electrode in humans. Pain 96:247–252
Inui K, Tsuji T, Kakigi R (2006) Temporal analysis of cortical mechanisms for pain relief by tactile stimuli in humans. Cereb Cortex 16:355–365. doi:10.1093/cercor/bhi114
Jääskeläinen IP, Ahveninen J, Bonmassar G et al (2004) Human posterior auditory cortex gates novel sounds to consciousness. Proc Natl Acad Sci USA 101:6809–6814
Javitt DC, Strous RD, Grochowski S, Ritter W, Cowan N (1997) Impaired precision, but normal retention, of auditory sensory (“echoic”) memory information in schizophrenia. J Abnorm Psychol 106:315–324
Kekoni J, Hamalainen H, Saarinen M, Grohn J, Reinikainen K, Lehtokoski A, Näätänen R (1997) Rate effect and mismatch responses in the somatosensory system: ERP-recordings in humans. Biol Psychol 46:125–142
Kiiski H, Reilly RB, Lonergan R et al (2012) Only low frequency event-related EEG activity is compromised in multiple sclerosis: insights from an independent component clustering analysis. PLoS One 7:e45536. doi:10.1371/journal.pone.0045536
Lehmann D, Skrandies W (1980) Reference-free identification of components of checkerboard-evoked multichannel potential fields. Electroencephalogr Clin Neurophysiol 48:609–621
Lehmann D, Ozaki H, Pal I (1987) EEG alpha map series: brain micro-states by space-oriented adaptive segmentation. Electroencephalogr Clin Neurophysiol 67:271–288
Lehmann C, Koenig T, Jelic V et al (2007) Application and comparison of classification algorithms for recognition of Alzheimer’s disease in electrical brain activity (EEG). J Neurosci Methods 161:342–350. doi:10.1016/j.jneumeth.2006.10.023
Liang M, Mouraux A, Chan V, Blakemore C, Iannetti GD (2010) Functional characterisation of sensory ERPs using probabilistic ICA: effect of stimulus modality and stimulus location. Clin Neurophysiol 121:577–587. doi:10.1016/j.clinph.2009.12.012
Lozano-Soldevilla D, Marco-Pallares J, Fuentemilla L, Grau C (2012) Common N1 and mismatch negativity neural evoked components are revealed by independent component model-based clustering analysis. Psychophysiology 49:1454–1463. doi:10.1111/j.1469-8986.2012.01458.x
MacDonald DB, Stigsby B, Al Zayed Z (2004) A comparison between derivation optimization and Cz’–FPz for posterior tibial P37 somatosensory evoked potential intraoperative monitoring. Clin Neurophysiol 115:1925–1930. doi:10.1016/j.clinph.2004.03.008
Maris E, Oostenveld R (2007) Nonparametric statistical testing of EEG- and MEG-data. J Neurosci Methods 164:177–190. doi:10.1016/j.jneumeth.2007.03.024
May PJ, Tiitinen H (2004) Auditory scene analysis and sensory memory: the role of the auditory N100m. Neurol Clin Neurophysiol 2004:19
May PJ, Tiitinen H (2010) Mismatch negativity (MMN), the deviance-elicited auditory deflection, explained. Psychophysiology 47:66–122. doi:10.1111/j.1469-8986.2009.00856.x
Michel CM, Thut G, Morand S et al (2001) Electric source imaging of human brain functions. Brain Res Rev 36:108–118
Michel CM, Murray MM, Lantz G, Gonzalez S, Spinelli L, Grave de Peralta R (2004) EEG source imaging. Clin Neurophysiol 115:2195–2222. doi:10.1016/j.clinph.2004.06.001
Miura T, Sonoo M, Shimizu T (2003) Establishment of standard values for the latency, interval and amplitude parameters of tibial nerve somatosensory evoked potentials (SEPs). Clin Neurophysiol 114:1367–1378
Mouraux A, Iannetti GD, Plaghki L (2010) Low intensity intra-epidermal electrical stimulation can activate Adelta-nociceptors selectively. Pain 150:199–207. doi:10.1016/j.pain.2010.04.026
Murray MM, Brunet D, Michel CM (2008) Topographic ERP analyses: a step-by-step tutorial review. Brain Topogr 20:249–264. doi:10.1007/s10548-008-0054-5
Näätänen R, Winkler I (1999) The concept of auditory stimulus representation in cognitive neuroscience. Psychol Bull 125:826–859
Näätänen R, Gaillard AW, Mantysalo S (1978) Early selective-attention effect on evoked potential reinterpreted. Acta Psychol 42:313–329
Näätänen R, Teder W, Alho K, Lavikainen J (1992) Auditory attention and selective input modulation: a topographical ERP study. NeuroReport 3:493–496
Näätänen R, Paavilainen P, Rinne T, Alho K (2007) The mismatch negativity (MMN) in basic research of central auditory processing: a review. Clin Neurophysiol 118:2544–2590. doi:10.1016/j.clinph.2007.04.026
Näätänen R, Kujala T, Kreegipuu K, Carlson S, Escera C, Baldeweg T, Ponton C (2011) The mismatch negativity: an index of cognitive decline in neuropsychiatric and neurological diseases and in ageing. Brain 134:3435–3453. doi:10.1093/brain/awr064
Opitz B, Schröger E, Von Cramon D (2005) Sensory and cognitive mechanisms for preattentive change detection in auditory cortex. Eur J Neurosci 21:531–535
Pakarinen S, Huotilainen M, Näätänen R (2010) The mismatch negativity (MMN) with no standard stimulus. Clin Neurophysiol 121:1043–1050
Pascual-Marqui RD, Michel CM, Lehmann D (1995) Segmentation of brain electrical activity into microstates: model estimation and validation. IEEE Trans Biomed Eng 42:658–665. doi:10.1109/10.391164
Paxinos G, Mai JK (2004) The human nervous system. Academic Press, San Diego
Ploner M, Schmitz F, Freund HJ, Schnitzler A (1999) Parallel activation of primary and secondary somatosensory cortices in human pain processing. J Neurophysiol 81:3100–3104
Ploner M, Schmitz F, Freund HJ, Schnitzler A (2000) Differential organization of touch and pain in human primary somatosensory cortex. J Neurophysiol 83:1770–1776
Ploner M, Pollok B, Schnitzler A (2004) Pain facilitates tactile processing in human somatosensory cortices. J Neurophysiol 92:1825–1829. doi:10.1152/jn.00260.2004
Sams M, Hamalainen M, Antervo A, Kaukoranta E, Reinikainen K, Hari R (1985) Cerebral neuromagnetic responses evoked by short auditory stimuli. Electroencephalogr Clin Neurophysiol 61:254–266
Stelzer J, Chen Y, Turner R (2013) Statistical inference and multiple testing correction in classification-based multi-voxel pattern analysis (MVPA): random permutations and cluster size control. Neuroimage 65:69–82. doi:10.1016/j.neuroimage.2012.09.063
Sussman ES (2007) A new view on the MMN and attention debate. J Psychophysiol 21:164–175
Torta DM, Liang M, Valentini E, Mouraux A, Iannetti GD (2012) Dishabituation of laser-evoked EEG responses: dissecting the effect of certain and uncertain changes in stimulus spatial location. Exp Brain Res 218:361–372. doi:10.1007/s00221-012-3019-6
Valentini E, Torta DM, Mouraux A, Iannetti GD (2011) Dishabituation of laser-evoked EEG responses: dissecting the effect of certain and uncertain changes in stimulus modality. J Cogn Neurosci 23:2822–2837. doi:10.1162/jocn.2011.21609
Winkler I, Karmos G, Näätänen R (1996) Adaptive modeling of the unattended acoustic environment reflected in the mismatch negativity event-related potential. Brain Res 742:239–252
Yucel G, McCarthy G, Belger A (2007) fMRI reveals that involuntary visual deviance processing is resource limited. Neuroimage 34:1245–1252. doi:10.1016/j.neuroimage.2006.08.050
Acknowledgments
LH and CZ were supported by the National Natural Science Foundation of China (31200856, 31471082), Natural Science Foundation Project of CQ CSTC, and Fundamental Research Funds for the Central Universities (SWU1409105). CZ was supported by the program of China Scholarship Council (201406990026).
Conflict of interest
The authors declare no competing financial interests.
Author information
Authors and Affiliations
Corresponding author
Additional information
Chen Zhao and Elia Valentini have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhao, C., Valentini, E. & Hu, L. Functional features of crossmodal mismatch responses. Exp Brain Res 233, 617–629 (2015). https://doi.org/10.1007/s00221-014-4141-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-014-4141-4