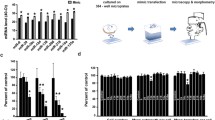
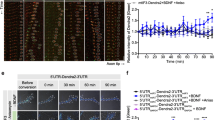
Abstract
MicroRNAs (miRNAs) constitute a novel class of small, non-coding RNAs that act as post-transcriptional regulators of gene expression. Remarkably, it has been shown that these small molecules can coordinately regulate multiple genes coding for proteins with related cellular functions. Previously, we reported that brain-specific miR-338 modulates the axonal expression of cytochrome c oxidase IV (COXIV), a nuclear-encoded mitochondrial protein that plays a key role in oxidative phosphorylation and axonal function. Here, we report that ATP synthase (ATP5G1), like COXIV mRNA, contains a putative miR-338 binding site, and that modulation of miR-338 levels in the axon results in alterations in both COXIV and ATP5G1 expression. Importantly, miR-338 modulation of local COXIV and ATP5G1 expression has a marked effect on axonal ROS levels, as well as axonal growth. These findings point to a mechanism by which miR-338 modulates local energy metabolism through the coordinate regulation of the expression of multiple nuclear-encoded mitochondrial mRNAs in the axon.





Similar content being viewed by others
Abbreviations
- H2DCFDA:
-
5-(and-6)-Carboxy-2′, 7′-dichlorodihydrofluorescein diacetate
- NDGA:
-
Nordihydraguaiaretic acid
- COX:
-
Cytochorome oxidase
- qRT-PCR:
-
Quantitative real-time reverse transcriptase polymerase chain reaction
- miRNA:
-
MicroRNA
References
Schuman E, Chan D (2004) Fueling synapses. Cell 119:738–740
Hollenbeck PJ (2005) Mitochondria and neurotransmission: evacuating the synapse. Neuron 47:331–333
Amiri M, Hollenbeck PJ (2008) Mitochondrial biogenesis in the axons of vertebrate peripheral neurons. Dev Neurobiol 68:1348–1361
Chang DT, Honick AS, Reynolds IJ (2006) Mitochondrial trafficking to synapses in cultured primary cortical neurons. J Neurosci 26:7035–7045
Ly CV, Verstreken P (2006) Mitochondria at the synapse. Neuroscientist 12:291–299
Bindokas VP, Lee CC, Colmers WF, Miller RJ (1998) Changes in mitochondrial function resulting from synaptic activity in the rat hippocampal slice. J Neurosci 18:4570–4587
Zenisek D, Matthews G (2000) The role of mitochondria in presynaptic calcium handling at a ribbon synapse. Neuron 25:229–237
Knott AB, Perkins G, Schwarzenbacher R, Bossy-Wetzel E (2008) Mitochondrial fragmentation in neurodegeneration. Nat Rev Neurosci 9:505–518
Gioio AE, Eyman M, Zhang H, Lavina ZS, Giuditta A, Kaplan BB (2001) Local synthesis of nuclear-encoded mitochondrial proteins in the presynaptic nerve terminal. J Neurosci Res 64:447–453
Gioio AE, Lavina ZS, Jurkovicova D, Zhang H, Eyman M, Giuditta A, Kaplan BB (2004) Nerve terminals of squid photoreceptor neurons contain a heterogeneous population of mRNAs and translate a transfected reporter mRNA. Eur J Neurosci 20:865–872
Hillefors M, Gioio AE, Mameza MG, Kaplan BB (2007) Axon viability and mitochondrial function are dependent on local protein synthesis in sympathetic neurons. Cell Mol Neurobiol 27:701–716
Wang W, van Niekerk E, Willis DE, Twiss JL (2007) RNA transport and localized protein synthesis in neurological disorders and neural repair. Dev Neurobiol 67:1166–1182
Willis DE, van Niekerk EA, Sasaki Y, Mesngon M, Merianda TT, Williams GG, Kendall M, Smith DS, Bassell GJ, Twiss JL (2007) Extracellular stimuli specifically regulate localized levels of individual neuronal mRNAs. J Cell Biol 178:965–980
Taylor AM, Berchtold NC, Perreau VM, Tu CH, Li JN, Cotman CW (2009) Axonal mRNA in uninjured and regenerating cortical mammalian axons. J Neurosci 29:4697–4707
Aschrafi A, Schwechter AD, Mameza MG, Natera-Naranjo O, Gioio AE, Kaplan BB (2008) MicroRNA-338 regulates local cytochrome c oxidase IV mRNA levels and oxidative phosphorylation in the axons of sympathetic neurons. J Neurosci 28:12581–12590
Natera-Naranjo O, Kar AN, Aschrafi A, Gervasi NM, MacGibeny MA, Gioio AE, Kaplan BB (2012) Local translation of ATP synthase subunit 9 mRNA alters ATP levels and the production of ROS in the axon. Mol Cell Neurosci 49:263–270
Aschrafi A, Natera-Naranjo O, Gioio AE, Kaplan BB (2010) Regulation of axonal trafficking of cytochrome c oxidase IV mRNA. Mol Cell Neurosci 43:422–430
Ambros V (2001) microRNAs: tiny regulators with great potential. Cell 107:823–826
Kim J, Krichevsky A, Grad Y, Hayes GD, Kosik KS, Church GM, Ruvkun G (2004) Identification of many microRNAs that copurify with polyribosomes in mammalian neurons. Proc Natl Acad Sci USA 101:360–365
Miska EA, Alvarez-Saavedra E, Townsend M, Yoshii A, Sestan N, Rakic P, Constantine-Paton M, Horvitz HR (2004) Microarray analysis of microRNA expression in the developing mammalian brain. Genome Biol 5:R68
Mathonnet G, Fabian MR, Svitkin YV, Parsyan A, Huck L, Murata T, Biffo S, Merrick WC, Darzynkiewicz E, Pillai RS, Filipowicz W, Duchaine TF, Sonenberg N (2007) MicroRNA inhibition of translation initiation in vitro by targeting the cap-binding complex eIF4F. Science 317:1764–1767
Campenot RB (1977) Local control of neurite development by nerve growth factor. Proc Natl Acad Sci USA 74:4516–4519
Natera-Naranjo O, Aschrafi A, Gioio AE, Kaplan BB (2010) Identification and quantitative analyses of microRNAs located in the distal axons of sympathetic neurons. RNA 16:1516–1529
An J, Choi KP, Wells CA, Chen YP (2010) Identifying co-regulating microRNA groups. J Bioinform Comput Biol 8:99–115
Lindow M (2011) Prediction of targets for microRNAs. Methods Mol Biol 703:311–317
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative C (T) method. Nat Protoc 3:1101–1108
Vonesch C, Unser M (2008) A fast thresholded landweber algorithm for wavelet-regularized multidimensional deconvolution. IEEE Trans Image Process 17:539–549
Griffiths-Jones S, Grocock RJ, van Dongen S, Bateman A, Enright AJ (2006) miRBase: microRNA sequences, targets and gene nomenclature. Nucleic Acids Res 34:D140–D144
Xiao F, Zuo Z, Cai G, Kang S, Gao X, Li T (2009) miRecords: an integrated resource for microRNA–target interactions. Nucl Acid Res 37:D105–D110
Zuker M (2003) Mfold web server for nucleic acid folding and hybridization prediction. Nucleic Acids Res 31:3406–3415
Kaplan BB, Gioio AE, Hillefors M, Aschrafi A (2009) Axonal protein synthesis and the regulation of local mitochondrial function. In: Koenig E (ed) Cell biology of the axon. Springer, Berlin, pp 225–242
Vives-Bauza C, Magrané J, Andreu AL, Manfredi G (2010) Novel role of ATPase subunit C targeting peptides beyond mitochondrial protein import. Mol Biol Cell 21:131–139
Loeb LA, Wallace DC, Martin GM (2005) The mitochondrial theory of aging and its relationship to reactive oxygen species damage and somatic mtDNA mutations. Proc Natl Acad Sci USA 102:18769–18770
Hong WK, Han EH, Kim DG, Ahn JY, Park JS, Han BG (2007) Amyloid-beta-peptide reduces the expression level of mitochondrial cytochrome oxidase subunits. Neurochem Res 32:1483–1488
Li Y, Park JS, Deng JH, Bai Y (2006) Cytochrome c oxidase subunit IV is essential for assembly and respiratory function of the enzyme complex. J Bioenerg Biomembr 38:283–291
Abramov AY, Scorziello A, Duchen MR (2007) Three distinct mechanisms generate oxygen free radicals in neurons and contribute to cell death during anoxia and reoxygenation. J Neurosci 27:1129–1138
Xing SL, Yan J, Yu ZH, Zhu CQ (2011) Neuronal cell surface ATP synthase mediates synthesis of extracellular ATP and regulation of intracellular pH. Cell Biol Int 35:81–86
Backes C, Meese E, Lenhof HP, Keller A (2010) A dictionary on microRNAs and their putative target pathways. Nucleic Acids Res 38:4476–4486
Inui M, Martello G, Piccolo S (2010) MicroRNA control of signal transduction. Nat Rev Mol Cell Biol 11:252–263
Hengst U, Cox LJ, Macosko EZ, Jaffrey SR (2006) Functional and selective RNA interference in developing axons and growth cones. J Neurosci 26:5727–5732
Campenot RB (1987) Local control of neurite sprouting in cultured sympathetic neurons by nerve growth factor. Brain Res 465:293–301
Yoon YS, Lee JH, Hwang SC, Choi KS, Yoon G (2005) TGF beta1 induces prolonged mitochondrial ROS generation through decreased complex IV activity with senescent arrest in Mv1Lu cells. Oncogene 24:1895–1903
Carter BJ, Anklesaria P, Choi S, Engelhardt JF (2009) Redox modifier genes and pathways in amyotrophic lateral sclerosis. Antioxid Redox Signal 11:1569–1586
Granata S, Zaza G, Simone S, Villani G, Latorre D, Pontrelli P, Carella M, Schena FP, Grandaliano G, Pertosa G (2009) Mitochondrial dysregulation and oxidative stress in patients with chronic kidney disease. BMC Genomics 10:388
Pocock R, Hobert O (2008) Oxygen levels affect axon guidance and neuronal migration in Caenorhabditis elegans. Nat Neurosci 11:894–900
Press C, Milbrandt J (2008) Nmnat delays axonal degeneration caused by mitochondrial and oxidative stress. J Neurosci 28:4861–4871
Lin MT, Beal MF (2006) Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature 443:787–795
Konradi C, Eaton M, MacDonald ML, Walsh J, Benes FM, Heckers S (2004) Molecular evidence for mitochondrial dysfunction in bipolar disorder. Arch Gen Psychiatry 61:300–308
Shao L, Martin MV, Watson SJ, Schatzberg A, Akil H, Myers RM, Jones EG, Bunney WE, Vawter MP (2008) Mitochondrial involvement in psychiatric disorders. Ann Med 40:281–295
Hunsberger JG, Austin DR, Chen G, Manji HK (2009) MicroRNAs in mental health: from biological underpinnings to potential therapies. Neuro Mol Med 11:173–182
Jin P, Zarnescu DC, Ceman S, Nakamoto M, Mowrey J, Jongens TA, Nelson DL, Moses K, Warren ST (2004) Biochemical and genetic interaction between the fragile X mental retardation protein and the microRNA pathway. Nature Neurosci 7:113–117
Kuhn DE, Nuovo GJ, Terry AV Jr, Martin MM, Malana GE, Sansom SE, Pleister AP, Beck WD, Head E, Feldman DS, Elton TS (2010) Chromosome 21-derived microRNAs provide an etiological basis for aberrant protein expression in human Down syndrome brains. J Biol Chem 285:1529–1543
Szulwach KE, Li X, Smrt RD, Li Y, Luo Y, Lin L, Santistevan NJ, Li W, Zhao X, Jin P (2010) Cross talk between microRNA and epigenetic regulation in adult neurogenesis. J Cell Biol 189:127–141
Acknowledgments
This work was supported by the Division of Intramural Research Programs of the National Institute of Mental Health. We thank Ms. Sanah Vohra for her invaluable technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aschrafi, A., Kar, A.N., Natera-Naranjo, O. et al. MicroRNA-338 regulates the axonal expression of multiple nuclear-encoded mitochondrial mRNAs encoding subunits of the oxidative phosphorylation machinery. Cell. Mol. Life Sci. 69, 4017–4027 (2012). https://doi.org/10.1007/s00018-012-1064-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-012-1064-8