Abstract
Background
The pathogenesis of preeclampsia (PE) is suggested to be a consequence of inflammation. Previously conducted investigations on nod-like receptor pyrin domain-containing 3 (NLRP3) have shed light to its crucial role in PE. Furthermore, microRNA-520c-3p (miR-520c-3p) is observed to be implicated in inflammation. Therefore, the current study aimed to explore the role of miR-520c-3p in inflammatory cascade of PE by targeting NLRP3.
Methods
Microarray analyses were performed to screen differentially expressed genes associated with PE, and the potential relationship between miR-520c-3p and NLRP3 was analyzed. PE and normal placenta tissues were collected to determine the levels of inflammatory cytokines (IL-18, IL-33, IL-1β, IL-10, and TNF-α), miR-520c-3p and NLRP3. Hypoxic HTR8/SVneo cells were transfected with oe-NLRP3, si-NLRP3 or miR-520c-3p mimic to elucidate the functional role of NLRP3 or miR-520c-3p in the inflammatory cascade in PE, followed by the evaluation of levels of inflammatory cytokines and NLRP3 inflammasomes (NLRP3, ASC and caspase-1). Additionally, the HTR8/SVneo cell migration and invasion were evaluated.
Results
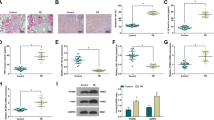
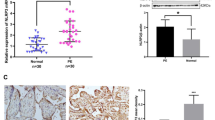
An upregulation of NLRP3, IL-18, IL-1β and TNF-α, and downregulation of miR-520c-3p, IL-33 and IL-10 were observed in PE placenta tissues. NLRP3 was found to be a target gene of miR-520c-3p. HTR8/SVneo cells after hypoxia transfected with si-NLRP3 or miR-520c-3p mimic exhibited decreased levels of inflammatory cytokines and NLRP3 inflammasomes, in addition to increased IL-10 and IL-33 levels. Moreover, enhanced migration and invasion abilities were observed in cells transfected with si-NLRP3.
Conclusion
Collectively, miR-520c-3p could potentially inhibit NLRP3 inflammasome activation and inflammatory cascade in PE by downregulating NLRP3, highlighting the potential of miR-520c-3p as a therapeutic target for PE treatment.




Similar content being viewed by others
References
Kohli S, Ranjan S, Hoffmann J, Kashif M, Daniel EA, Al-Dabet MM, et al. Maternal extracellular vesicles and platelets promote preeclampsia via inflammasome activation in trophoblasts. Blood. 2016;128:2153–64.
Leavey K, Benton SJ, Grynspan D, Kingdom JC, Bainbridge SA, Cox BJ. Unsupervised placental gene expression profiling identifies clinically relevant subclasses of human preeclampsia. Hypertension. 2016;68:137–47.
Brien ME, Boufaied I, Soglio DD, Rey E, Leduc L, Girard S. Distinct inflammatory profile in preeclampsia and postpartum preeclampsia reveal unique mechanisms. Biol Reprod. 2018;100(1):187–94.
Winship A, Dimitriadis E. Interleukin 11 is upregulated in preeclampsia and leads to inflammation and preeclampsia features in mice. J Reprod Immunol. 2018;125:32–8.
Harmon AC, Cornelius DC, Amaral LM, Faulkner JL, Cunningham MW Jr, Wallace K, et al. The role of inflammation in the pathology of preeclampsia. Clin Sci (Lond). 2016;130:409–19.
Valencia-Ortega J, Zarate A, Saucedo R, Hernandez-Valencia M, Cruz JG, Puello E. Placental proinflammatory state and maternal endothelial dysfunction in preeclampsia. Gynecol Obstet Invest. 2018;84(1):12–9.
Yin Y, Zhou Z, Liu W, Chang Q, Sun G, Dai Y. Vascular endothelial cells senescence is associated with NOD-like receptor family pyrin domain-containing 3 (NLRP3) inflammasome activation via reactive oxygen species (ROS)/thioredoxin-interacting protein (TXNIP) pathway. Int J Biochem Cell Biol. 2017;84:22–34.
Weel IC, Romao-Veiga M, Matias ML, Fioratti EG, Peracoli JC, Borges VT, et al. Increased expression of NLRP3 inflammasome in placentas from pregnant women with severe preeclampsia. J Reprod Immunol. 2017;123:40–7.
Hu J, Zhu Y, Zhang JZ, Zhang RG, Li HM. A novel mutation in the pyrin domain of the NOD-like receptor family pyrin domain containing protein 3 in Muckle-Wells syndrome. Chin Med J (Engl). 2017;130:586–93.
Wang Y, Dong Q, Gu Y, Groome LJ. Up-regulation of miR-203 expression induces endothelial inflammatory response: potential role in preeclampsia. Am J Reprod Immunol. 2016;76:482–90.
Keklikoglou I, Koerner C, Schmidt C, Zhang JD, Heckmann D, Shavinskaya A, et al. MicroRNA-520/373 family functions as a tumor suppressor in estrogen receptor negative breast cancer by targeting NF-kappaB and TGF-beta signaling pathways. Oncogene. 2012;31:4150–63.
Lee YH, Na HS, Jeong SY, Jeong SH, Park HR, Chung J. Comparison of inflammatory microRNA expression in healthy and periodontitis tissues. Biocell. 2011;35:43–9.
Donker RB, Mouillet JF, Chu T, Hubel CA, Stolz DB, Morelli AE, et al. The expression profile of C19MC microRNAs in primary human trophoblast cells and exosomes. Mol Hum Reprod. 2012;18:417–24.
Zhou R, Tardivel A, Thorens B, Choi I, Tschopp J. Thioredoxin-interacting protein links oxidative stress to inflammasome activation. Nat Immunol. 2010;11:136–40.
Atkins D, Reiffen KA, Tegtmeier CL, Winther H, Bonato MS, Storkel S. Immunohistochemical detection of EGFR in paraffin-embedded tumor tissues: variation in staining intensity due to choice of fixative and storage time of tissue sections. J Histochem Cytochem. 2004;52:893–901.
Wang B, Shen ZL, Gao ZD, Zhao G, Wang CY, Yang Y, et al. MiR-194, commonly repressed in colorectal cancer, suppresses tumor growth by regulating the MAP4K4/c-Jun/MDM2 signaling pathway. Cell Cycle. 2015;14:1046–58.
Hardy DB, Yang K. The expression of 11 beta-hydroxysteroid dehydrogenase type 2 is induced during trophoblast differentiation: effects of hypoxia. J Clin Endocrinol Metab. 2002;87:3696–701.
Staff AC, Johnsen GM, Dechend R, Redman CWG. Preeclampsia and uteroplacental acute atherosis: immune and inflammatory factors. J Reprod Immunol. 2014;101–102:120–6.
Kwaan HC, Wang J, Boggio L, Weiss I, Grobman WA. The thrombogenic effect of an inflammatory cytokine on trophoblasts from women with preeclampsia. Am J Obstet Gynecol. 2004;191:2142–7.
Matias ML, Romao M, Weel IC, Ribeiro VR, Nunes PR, Borges VT, et al. Endogenous and uric acid-induced activation of nlrp3 inflammasome in pregnant women with preeclampsia. PLoS One. 2015;10:e0129095.
Bauernfeind F, Rieger A, Schildberg FA, Knolle PA, Schmid-Burgk JL, Hornung V. NLRP3 inflammasome activity is negatively controlled by miR-223. J Immunol. 2012;189:4175–81.
Qiu C, Li Y, Li M, Li M, Liu X, McSharry C, et al. Anti-interleukin-33 inhibits cigarette smoke-induced lung inflammation in mice. Immunology. 2013;138:76–82.
Chen H, Zhou X, Han TL, Baker PN, Qi H, Zhang H. Decreased IL-33 production contributes to trophoblast cell dysfunction in pregnancies with preeclampsia. Mediat Inflamm. 2018;2018:9787239.
Naruse K, Akasaka J, Shigemitsu A, Tsunemi T, Koike N, Yoshimoto C, et al. Involvement of visceral adipose tissue in immunological modulation of inflammatory cascade in preeclampsia. Mediat Inflamm. 2015;2015:325932.
Xie C, Yao MZ, Liu JB, Xiong LK. A meta-analysis of tumor necrosis factor-alpha, interleukin-6, and interleukin-10 in preeclampsia. Cytokine. 2011;56:550–9.
Huang X, Huang H, Dong M, Yao Q, Wang H. Serum and placental interleukin-18 are elevated in preeclampsia. J Reprod Immunol. 2005;65:77–87.
Lai Z, Kalkunte S, Sharma S. A critical role of interleukin-10 in modulating hypoxia-induced preeclampsia-like disease in mice. Hypertension. 2011;57:505–14.
Lim R, Lappas M. NOD-like receptor pyrin domain-containing-3 (NLRP3) regulates inflammation-induced pro-labor mediators in human myometrial cells. Am J Reprod Immunol. 2018;79:e12825.
Tschopp J, Schroder K. NLRP3 inflammasome activation: the convergence of multiple signalling pathways on ROS production? Nat Rev Immunol. 2010;10:210–5.
De Nardo D, Latz E. NLRP3 inflammasomes link inflammation and metabolic disease. Trends Immunol. 2011;32:373–9.
Chen C, Ma X, Yang C, Nie W, Zhang J, Li H, et al. Hypoxia potentiates LPS-induced inflammatory response and increases cell death by promoting NLRP3 inflammasome activation in pancreatic beta cells. Biochem Biophys Res Commun. 2018;495:2512–8.
Wang F, Yan J. MicroRNA-454 is involved in regulating trophoblast cell proliferation, apoptosis, and invasion in preeclampsia by modulating the expression of ephrin receptor B4. Biomed Pharmacother. 2018;107:746–53.
Nizyaeva NV, Kulikova GV, Nagovitsyna MN, Kan NE, Prozorovskaya KN, Shchegolev AI, et al. Expression of microRNA-146a and microRNA-155 in placental villi in early- and late-onset preeclampsia. Bull Exp Biol Med. 2017;163:394–9.
Takahashi H, Ohkuchi A, Kuwata T, Usui R, Baba Y, Suzuki H, et al. Endogenous and exogenous miR-520c-3p modulates CD44-mediated extravillous trophoblast invasion. Placenta. 2017;50:25–31.
Wang Y, Han Z, Fan Y, Zhang J, Chen K, Gao L, et al. MicroRNA-9 inhibits NLRP3 inflammasome activation in human atherosclerosis inflammation cell models through the JAK1/STAT signaling pathway. Cell Physiol Biochem. 2017;41:1555–71.
Acknowledgements
We would like show sincere appreciation to the reviewers for critical comments on this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors’ declares that they have no conflict of interest.
Additional information
Responsible Editor: John Di Battista.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liu, Z., Zhao, X., Shan, H. et al. microRNA-520c-3p suppresses NLRP3 inflammasome activation and inflammatory cascade in preeclampsia by downregulating NLRP3. Inflamm. Res. 68, 643–654 (2019). https://doi.org/10.1007/s00011-019-01246-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-019-01246-8