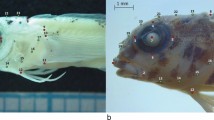
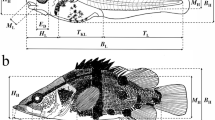
Abstract
Knowledge of the relationship between auditory form and function is necessary to understand the role of hearing in fish ecology, as well as the potential impacts of anthropogenic noise and environmental change on hearing-mediated functions. We explored the functional significance of otolith (ear stone) morphology in a large predatory sciaenid, Argyrosomus japonicus (Temminck and Schlegel), by comparing ontogenetic changes in otolith structure to changes in habitat, diet and swimming activity. We examined an otolith metric that has previously been linked to habitat use and activity in fishes, the relative sensory area (sulcus to otolith area ratio, S:O), and a new metric that quantifies the protuberant structure (calcareous concrescence) found on the distal surface of sciaenid otoliths (otolith perimeter to area ratio, P:SA). Strong allometric growth of the sulcus acusticus was detected in the sagitta of A. japonicus ranging from 7 to 80 cm TL. Both S:O and P:SA changed rapidly until A. japonicus reached 50 cm TL, after which only a gradual increase in relative sensory area was detected. The aspect ratio of the caudal fin (CAR, an index of swimming activity) abruptly increased in A. japonicus at 41–50 cm TL. These changes coincide with an observed shift in diet from benthic invertebrates to pelagic fish, a shift in foraging habitat and an increase in foraging activity.





Similar content being viewed by others
References
Aguirre WE (2003) Allometric growth of the sulcus in Cynoscion spp. (Sciaenidae). J Fish Biol 63:1341–1346
Aguirre H, Lombarte A (1999) Ecomorphological comparisons of sagittae in Mullus barbatus and M. surmuletus. J Fish Biol 55:105–114
Arellano RV, Hamerlynck O, Vincx M, Mees J, Hostens K, Gijselinck W (1995) Changes in the ratio of the sulcus acusticus area to the sagitta area of Pomatoschistus minutus and P. lozanoi (Pisces, Gobiidae). Mar Biol 122:355–360
Astrup JE, Møhl BE (1993) Detection of intense ultrasound by the cod Gadus morhua. J Exp Biol 182:71–80
Au WWL, Floyd RW, Penner RH, Murchison AE (1974) Measurement of echolocation signals of the Atlantic bottlenose dolphin, Tursiops truncatus Montagu, in open waters. J Acoust Soc Am 56:1280–1290
Bervian G, Fontoura NF, Haimovici M (2006) Statistical model of variable allometric growth: otolith growth in Micropogonias furnieri (Actinopterygii, Sciaenidae). J Fish Biol 68:196–208
Bignami S, Enochs IC, Manzello DP, Sponaugle S, Cowen RK (2013) Ocean acidification alters the otoliths of a pantropical fish species with implications for sensory function. Proc Natl Acad Sci USA 110:7366–7370
Campana SE, Casselman JM (1993) Stock discrimination using otolith shape analysis. Can J Fish Aquat Sci 50:1062–1083
Carroll AG, Przeslawski R, Duncan A, Gunning M, Bruce B (2017) A critical review of the potential impacts of marine seismic surveys on fish and invertebrates. Mar Pollut Bull 114:9–24
Chao LN (1978) A basis for classifying western Atlantic Sciaenidae (Teleostei: Perciformes). NOAA Tech Rep NMFS Tech Circ 415:1–64
Fay R (2009) Soundscapes and the sense of hearing of fishes. Integr Zool 4:26–32
Fay RR, Popper AN (2000) Evolution of hearing in vertebrates: the inner ears and processing. Hear Res 149:1–10
Gauldie RW (1988) Function, form and time-keeping properties of fish otoliths. Comp Biochem Physiol A Comp Physiol 91:395–402
Griffiths MH (1996) Life history of the dusky kob Argyrosomus japonicus (Sciaenidae) off the east coast of South Africa. S Afr J Mar Sci 17:135–154
Higgs DM, Plachta DTT, Rollo AK, Singheiser M, Hastings MC, Popper AN (2004) Development of ultrasound detection in American shad (Alosa sapidissima). J Exp Biol 207:155–163
Horodysky AZ, Brill RW, Fine ML, Musick JA, Latour RJ (2008) Acoustic pressure and particle motion thresholds in six sciaenid fishes. J Exp Biol 211:1504–1511
Hüssy K (2008) Otolith shape in juvenile cod (Gadus morhua): ontogenetic and environmental effects. J Exp Mar Biol Ecol 364:35–41
Leis JM, Siebeck U, Dixson DL (2011) How nemo finds home: the neuroecology of dispersal and of population connectivity in larvae of marine fishes. Integr Comp Biol 51:826–843
Lombarte A (1992) Changes in otolith area: sensory area ratio with body size and depth. Environ Biol Fishes 33:405–410
Lombarte A, Fortuño JM (1992) Differences in the morphological features of the sacculus of the inner ear of two hakes (Merluccius capensis and M. paradoxus, Gadiformes) inhabits from different depth of sea. J Morphol 214:97–107
Lombarte A, Lleonart J (1993) Otolith size changes related with body growth, habitat, depth and temperature. Environ Biol Fishes 37:297–306
Lombarte A, Popper AN (2004) Quantitative changes in the otolithic organs of the inner ear during the settlement period in European hake Merluccius merluccius. Mar Ecol Prog Ser 267:233–240
Lombarte A, Olaso I, Bozzano A (2003) Ecomorphological trends in the Artedidraconidae (Pisces:Perciformes:Notothenioidei) of the Weddell Sea. Antarct Sci 15:211–218
Lombarte A, Palmer M, Matallanas J, Gómez-Zurita J, Morales-Nin B (2010) Ecomorphological trends and phylogenetic inertia of otolith sagittae in Nototheniidae. Environ Biol Fishes 89:607–618
Lychakov DV, Rebane YT (2000) Otolith regularities. Hear Res 143:83–102
Lychakov DV, Rebane YT, Lombarte A, Fuiman LA, Takabayashi A (2006) Fish otolith asymmetry: morphometry and modeling. Hear Res 219:1–11
Mackenzie KV (1981) Discussion of sea water sound–speed determinations. J Acoust Soc Am 70:801–806
Mann DA, Lu ZM, Popper AN (1997) A clupeid fish can detect ultrasound. Nature 389:341
Mann DA, Lu Z, Hastings MC, Popper AN (1998) Detection of ultrasonic tones and simulated dolphin echolocation clicks by a teleost fish, the American shad (Alosa sapidissima). J Acoust Soc Am 104:562–568
Miller EF (2010) Structural irregularities in sagittal otoliths of black croaker (Cheilotrema saturnum) from Southern California. Bull S Calif Acad Sci 109:18–22
Monteiro LR, Di Beneditto APM, Guillermo LH, Rivera LA (2005) Allometric changes and shape differentiation of sagitta otoliths in sciaenid fishes. Fish Res 74:288–299
Morales-Nin B, Tores GJ, Lombarte A, Recasens L (1998) Otolith growth and age estimation in the European hake. J Fish Biol 53:1155–1168
Myrberg AAJ (1981) Sound communication and interception in fishes. In: Tavolga WN, Popper AN, Fay RR (eds) Hearing and sound communication in fishes. Springer, New York, pp 395–426
Norton SF, Luczkovich JJ, Motta PJ (1995) The role of ecomorphological studies in the comparative biology of fishes. Environ Biol Fishes 44:287–304
Parisi-Baradad V, Lombarte A, Garcia-Ladona E, Cabestany J, Piera J, Chic O (2005) Otolith shape contour analysis using affine transformation invariant wavelet transforms and curvature scale space representation. Mar Freshw Res 56:795–804
Pauly D (1989) Food consumption by tropical and temperate fish populations: some generalisations. J Fish Biol 35(Supplement A):11–20
Payne NL, van der Meulen DE, Suthers IM, Gray CA, Taylor MD (2015) Foraging intensity of wild mulloway Argyrosomus japonicus decreases with increasing anthropogenic disturbance. Mar Biol 162:539–546
Ponton D (2006) Is geometric morphometrics efficient for comparing otolith shape of different fish species? J Morphol 267:750–757
Popper AN, Fay RR (1993) Sound detection and processing by fish: critical review and major research questions. Brain Behav Evol 41:14–38
Popper AN, Fay RR (2011) Rethinking sound detection by fishes. Hear Res 273:25–36
Popper AN, Hastings MC (2009) The effects of anthropogenic sources of sound on fishes. J Fish Biol 75:455–489
Popper AN, Lu Z (2000) Structure–function relationships in fish otolith organs. Fish Res 46:15–25
Popper AN, Platt C (1983) Sensory surface of the saccule and lagena in the ears of ostariophysan fishes. J Morphol 176:121–129
Popper AN, Fay RR, Platt C, Sand O (2003) Sound detection mechanisms and capabilities of teleost fishes. In: Collin SP, Marshall NJ (eds) Sensory processing in aquatic environments. Springer, New York, pp 3–38
Popper AN, Ramcharitar J, Campana SE (2005) Why otoliths? Insights from inner ear physiology and fisheries biology. Mar Freshw Res 56:497–504
Ramcharitar J, Higgs DM, Popper AN (2001) Sciaenid inner ears: a study in diversity. Brain Behav Evol 58:152–162
Ramcharitar JU, Deng X, Ketten D, Popper AN (2004) Form and function in the unique inner ear of a teleost: the silver perch Bairdiella chrysoura. J Comp Neurol 475:531–539
Ramcharitar JU, Higgs DM, Popper AN (2006) Audition in sciaenid fishes with different swim bladder-inner ear configurations. J Acoust Soc Am 119:439–443
Schulz-Mirbach T, Ladich F, Plath M, Heß M (2019) Enigmatic ear stones: what we know about the functional role and evolution of fish otoliths. Biol Rev 94:457–482
Silberschneider V, Gray CA (2005) Arresting the decline of the commercial and recreational fisheries for mulloway (Argyrosomus japonicus). NSW Department of Primary Industries-Fisheries Final Report Series, No. 82, p 66
Silberschneider V, Gray CA (2008) Synopsis of biological, fisheries and aquaculture-related information on mulloway Argyrosomus japonicus (Pisces:Sciaenidae), with particular reference to Australia. J Appl Ichthyol 24:7–17
Simpson SD, Munday PL, Wittenrich ML, Manassa R, Dixson DL, Gagliano M, Yan HY (2011) Ocean acidification erodes crucial auditory behaviour in a marine fish. Biol Lett 7:917–920
Simpson SD, Radford AN, Nedelec SL, Ferrari MC, Chivers DP, McCormick MI, Meekan MG (2016) Anthropogenic noise increases fish mortality by predation. Nat Commun 7:10544
Slabbekoorn H, Bouton N, van Opzeeland I, Coers A, ten Cate C, Popper AN (2010) A noisy spring: the impact of globally rising underwater sound levels on fish. Trends Ecol Evol 25:419–427
Taylor MD, Palmer PJ, Fielder DS, Suthers IM (2005) Responsible estuarine finfish stock enhancement: an Australian perspective. J Fish Biol 67:299–331
Taylor MD, Fielder DS, Suthers IM (2006a) Spatial and ontogenetic variation in the diet of wild and stocked mulloway (Argyrosomus japonicus, Sciaenidae) in Australian estuaries. Estuar Coast 29:785–793
Taylor MD, Laffan SD, Fielder DS, Suthers IM (2006b) Key habitat and home range of mulloway Argyrosomus japonicus in a south-east Australian estuary: Finding the estuarine niche to optimise stocking. Mar Ecol Prog Ser 328:237–247
Taylor MD, Fielder DS, Suthers IM (2009) Growth and viability of hatchery-reared Argyrosomus japonicus released into open and semi-closed systems. Fish Manag Ecol 16:478–483
Torres GJ, Lombarte A, Morales-Nin B (2000) Variability of the sulcus acusticus in the sagittal otolith of the genus Merluccius (Merlucciidae). Fish Res 46:5–13
Tuset VM, Rosin PL, Lombarte A (2006) Sagittal otolith shape used in the identification of fishes of the genus Serranus. Fish Res 81:316–325
Tuset VM, Piretti S, Lombarte A, González JA (2010) Using sagittal otoliths and eye diameter for ecological characterization of deep-sea fish: Aphanopus carbo and A. intermedius from NE Atlantic waters. Sci Mar 74:807–814
Tuset VM, Otero-Ferrer JL, Gómez‐Zurita J, Venerus LA, Stransky C, Imondi R, Orlov AM, Ye Z, Santschi L, Afanasiev PK, Zhuang L (2016) Otolith shape lends support to the sensory drive hypothesis in rockfishes. J Evol Biol 29:2083–2097
Ueng JP, Huang BQ, Mok HK (1999) Ontogenetic changes of sound production in Japanese croaker, Argyrosomus japonicus (Sciaenidae). J Fish Soc Taiwan 26:13–21
Ueng JP, Huang BQ, Mok HK (2007) Sexual differences in the spawning sounds of the Japanese croaker, Argyrosomus japonicus (Sciaenidae). Zool Stud 46:103–110
Vignon M (2012) Ontogenetic trajectories of otolith shape during shift in habitat use: Interaction between otolith growth and environment. J Exp Mar Biol Ecol 420:26–32
Waessle JA, Lasta CA, Favero M (2003) Otolith morphology and body size relationships for juvenile Sciaenidae in the Rio de la Plata estuary (35–36° S). Sci Mar 67:233–240
Watkinson DA, Gillis DM (2005) Stock discrimination of Lake Winnipeg walleye based on Fourier and wavelet description of scale outline signals. Fish Res 72:193–203
Acknowledgements
We thank T. Lombarte, A. Popper, J. Ramcharitar, M. Hastings and A. Bass for advice provided whilst preparing this manuscript. The authors also wish to thank T. Mullaney, H. El-Hassan, A. Pursche and K. Taylor for assistance in various areas of this study.
Funding
This work was supported in part by a grant from the New South Wales Recreational Saltwater Fishing Trust and an Australian Research Council Linkage Grant (#LP0219596).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the University of New South Wales (UNSW), and were reviewed and approved by the UNSW Animal Care and Ethics Committee (Permit Number: ACEC #02/115).
Additional information
Communicated by E. Hunter.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Reviewed by K. S. Boyle and an undisclosed expert.
Rights and permissions
About this article
Cite this article
Taylor, M.D., Fowler, A.M. & Suthers, I.M. Insights into fish auditory structure–function relationships from morphological and behavioural ontogeny in a maturing sciaenid. Mar Biol 167, 21 (2020). https://doi.org/10.1007/s00227-019-3619-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-019-3619-9