Abstract
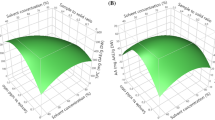
Functional phenolic compounds were extracted from Chinese quince using highly efficient ultrasonic-assisted extraction (UAE) technology. The experiment was carried out in accordance with a small central composite design (SCCD) five-level and three-variable approach. The optimum combination of solvent concentration, extraction temperature and time with the application of ultrasonic, for the maximum extraction of phenolic compounds from Chinese quince, was obtained by response surface methodology. Responses of phenolic compounds such as total phenolics, total flavonoids, and total flavan-3-ols were determined by the spectrophotometer assay. High significance of each response revealed that this SCCD model well-fitted the experimental parameters as well as the reliability of the predicted value. The optimal UAE conditions obtained by superimposed contour plot were 50% ethanol concentration, 63.9°C extraction temperature, and extraction time 11.4 min. Under the above-mentioned conditions, the experimental results for three responses were total phenolic content 2367.16 mg gallic acid equivalents /100 g, total flavonoid content 544.12 mg rutin equivalents /100 g, and total flavan-3-ols content 709.07 mg catechin equivalents /100 g, which well-matched with the predicted values. The optimal conditions attained from the present study would provide scientific references to economical production of relative phenolic compounds products.
Similar content being viewed by others
References
Bakar MFA, Mohamed M, Rahmat M, and Fry F (2009) Phytochemicals and antioxidant activity of different parts of bambangan (Mangifera pajang) and tarap (Artocarpus odoratissimus). Food Chem 113, 479–483.
Bazerra MA, Santelli RE, Oliveira EP, Villar LS, and Escaleira LA (2008) Response surface methodology (RSM) as a tool for optimization in analytical chemistry. Talanta 76, 956–977.
Dvorakova M, Moreira M, Dostalek P, Skulilova Z, Guido LF, and Barros AA (2008) Characterization of monomeric and oligomeric flavan-3-ols from barley and malt by liquid chromatography-ultraviolet detection-electrospray ionization mass spectrometry. J Chromatogr A 1189, 398–405.
Ghafoor K, Choi YH, Jeon JY, and Jo IH (2009) Optimization of ultrasound-assisted extraction of phenolic compounds, antioxidants, and anthocyanins from Grape (Vitis vinifera) seeds. J Agric Food Chem 57, 4988–4994.
Hamauzu Y, Inno T, Kume C, Irie M, and Hiramatsu K (2006) Antioxidant and antiulcerative properties of phenolics from Chinese quince, quince, and apple fruit. J Agric Food Chem 54, 765–772.
Hamauzu Y, Irie M, Kondo M, and Fujita T (2008) Antiucerative properties of crude polyphenols and juice of apple, and Chinese quince extracts. Food Chem 108, 488–495.
Hamauzu Y, Yasui H, Inno T, Kume C, and Omanyuda M (2005) Phenolic profile, antioxidant property, and anti-influenza viral activity of Chinese quince (Pseudocydonia sinenesis Schneid.), quince (Cydonia oblonga Mill.) and apple (Malus domestica Mill.) fruits. J Agric Food Chem 53, 928–934.
Hertog MGL, Feskens EJM, Hollman PCH, Katan MB, and Kromhout D (1993) Dietary antioxidant flavonoids and risk of coronary heart disease: the Zutphen elderly study. Lancet 342, 1007–1011.
Karvela E, Makris DP, Kalogeropoulos N, and Karathanos VT (2009) Deployment of response surface methodology to optimize recovery of grape (Vitis vinifera) stem polyphenolos. Talanta 76, 1131–1321.
Khan MK, Abert-Vian M, Fabiano-Tixier AS, Dangles O, and Chemat F (2010) Ultrasound-assisted extraction of polyphenols (flavanone glycosides) from orange (Citrus sinensis L.) peel. Food Chem 119, 851–858.
Knekt P, Jarvinen R, Runanen A, and Maatela J (1996) Flavonoids intake and coronary mortality in finland: a cohort study. Br Med J 312, 478–481.
Lee SY, Kang MJ, Kwon JH, Shinr SR, Lee GD, and Kim KS (2003) Monitoring of total phenolics, electron donating ability and nitrite scavenging ability in microwave-assisted extraction for angelica gigas nakai. Food Sci Biotechnol 12, 491–496.
Liu SC, Yang F, Zhang CH, Ji HW, Hong PZ, and Deng CJ (2009) Optimization of process parameters for supercritical carbon dioxide extraction of passiflora seed oil by response surface methodology. J Supercrit Fluids 48, 9–14.
Neptoe V, Grosso NR, and Guzman CA (2005) Optimization of extraction of phenolic antioxidants from peanut skins. J Sci Food Agric 85, 33–38.
Osawa K, Miyazaki K, Imai H, Arakawa T, Yasuda H, and Takeya K (1999) Inhibitory effects of Chinese quince (Chaenomeles sinensis) on hyaluronidase and histamine release from rat mast cells. Nat Med 53, 188–193.
Prommuak C, Eknamkul WD, and Shotipruk A (2008) Extraction of flavonoids and carotenoids from Thai silk waste and antioxidant activity of extracts. Sep Purif Technol 62, 444–448.
Romanik G, Gilgenast E, Przyjazny A, and Kaminski M (2007) Techniques of preparing plant material for chromatographic separation and analysis. J Biochem Biophys Methods 70, 253–261.
Rostagno MA, Plama M, and Barroso CG (2003) Ultrasound-assisted extraction of soy isoflavones. J Chromatogr A 1012, 119–128.
Sawai R, Kuroda K, Shibata T, Gomyou R, Osawa K, and Shimizu K (2008) Anti-influenza virus activity of Chaenomeles sinensis. J Ethnopharmacol 118, 108–112.
Vilkhu K, Mawson R, Simons L, and Bates D (2008) Applications and opportunities for ultrasound assisted extraction in the food industry-a review. Innov Food Sci Emerg Technol 9, 161–169.
Wang J, Sun BG, Cao YP, Tian Y, and Li XH (2008) Optimization of ultrasound-assisted extraction of phenolic compounds from wheat bran. Food Chem 106, 804–810.
Xie XF, Cai XQ, and Zhu SY (2007) Chemical composition and antimicrobial activity of essential oils of Chaenomeles speciosa from China. Food Chem 100, 1312–1315.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Teng, H., Jo, I.H. & Choi, Y.H. Optimization of ultrasonic-assisted extraction of phenolic compounds from Chinese quince (Chaenomeles sinensis) by response surface methodology. J. Korean Soc. Appl. Biol. Chem. 53, 618–625 (2010). https://doi.org/10.3839/jksabc.2010.094
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.3839/jksabc.2010.094