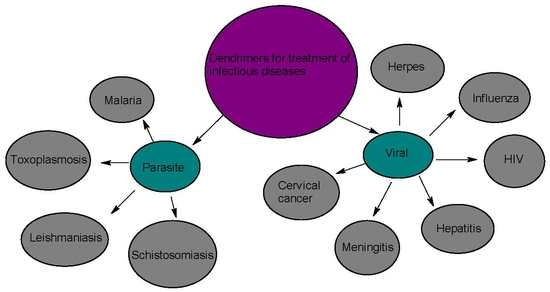
Application of Dendrimers for the Treatment of Infectious Diseases
Abstract
:1. Introduction
2. Parasitic Infections
2.1. Malaria
2.2. Leishmaniasis
2.3. Schistosomiasis
2.4. Toxoplasmosis
3. Viral Infection
3.1. HIV
3.2. Influenza
3.3. Meningitis
3.4. Herpes
3.5. Hepatitis
3.6. Cervical Cancer
4. Application of Dendrimers in the Treatment of Infectious Diseases
4.1. Various Dendrimers and Their Applications
4.2. Dendrimers for the Treatment of Leishmaniasis
4.3. Toxoplasmosis
4.4. Schistosomiasis
4.5. Malaria
5. Application of Dendrimers for the Treatment of Viral Infections
5.1. HIV
5.2. Herpes
5.3. Hepatitis
5.4. Influenza
5.5. Cervical Cancer
6. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Lewis, K. Antibiotics: Recover the lost art of drug delivery. Nature 2012, 485, 439–440. [Google Scholar] [CrossRef] [PubMed]
- Cole, S.T. Who will develop new antibacterial agents? Phil. Trans. R. Soc. B 2014, 369, 20130430. [Google Scholar] [CrossRef] [PubMed]
- Aderibigbe, B.A. Polymeric therapeutic delivery systems for the treatment of infectious diseases. Ther. Deliv. 2017, 8, 57–76. [Google Scholar] [CrossRef] [PubMed]
- Brownlie, J.; Peckham, C.; Waage, J.; Woolhouse, M.; Catherine Lyall, C.; Meagher, L.; Tait, J.; Baylis, M.; Nicoll, A. Infectious Diseases: Preparing for the Future—Future Threats; Office of Science and Innovation: London, UK, 2006. [Google Scholar]
- National Institutes of Health (US). Understanding Emerging and Re-emerging Infectious Diseases. 2007. Available online: https://www.ncbi.nlm.nih.gov/books/NBK20370/ (accessed on 30 December 2016).
- Jain, K.; Mehra, N.K.; Jain, N.K. Potentials and emerging trends in nanopharmacology. Curr. Opin. Pharmacol. 2014, 15, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Van den Bruel, A.; Haj-Hassan, T.; Thompson, M.; Buntinx, F.; Mant, D. Diagnostic value of clinical features at presentation to identify serious infection in children in developed countries: A systematic review. Lancet 2010, 375, 834–845. [Google Scholar] [CrossRef]
- Clinical Infectious Disease—Introduction. Available online: www.microbiologybook.org (accessed on 19 April 2017).
- Murillo, A.; Vera-Estrella, R.; Barkla, B.J.; Méndez, E.; Arias, C.F. Identification of host cell factors associated with astrovirus replication in Caco-2 cells. J. Virol. 2015, 89, 10359–10370. [Google Scholar] [CrossRef] [PubMed]
- Ljubin-Sternak, S.; Mestrovic, T.J. Review: Clamydia trachonmatis and genital mycoplasmias: Pathogens with an impact on human reproductive health. J. Pathog. 2014, 183167. [Google Scholar] [CrossRef]
- Potter, P. Summer buzz. Emerg. Infect. Dis. 2013, 19, 1184. [Google Scholar] [CrossRef]
- Rosenberg, R.; Beard, C.B. Vector-borne infections. Emerg. Infect. Dis. 2011, 17, 769–770. [Google Scholar] [CrossRef] [PubMed]
- Keen, E.C. Beyond phage therapy: Virotherapy of protozoal diseases. Future Microbiol. 2013, 8, 821–823. [Google Scholar] [CrossRef] [PubMed]
- Hyman, P.; Atterbury, R.; Barrow, P. Fleas and smaller fleas: Virotherapy for parasite infections. Trends Microbiol. 2013, 21, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Fernández de Mera, I.G.; Riz-Fons, F.; De La Fuente, G.; Mangold, A.J.; Gortázar, C.; De La Fuente, J. Spotted fever group rickettsiae in questing ticks, central Spain. Emerg. Infect. Dis. 2013, 19, 1163–1165. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carabollo, H. Emergency department management of mosquito—borne illness: Malaria, dengue, and West Nile virus. Emerg. Med. Pract. 2014, 16, 1–23. [Google Scholar]
- Collins, W.E. Plasmodium knowlesi: A malaria parasite of monkeys and humans. Annu. Rev. Entomol. 2012, 57, 107–121. [Google Scholar] [CrossRef] [PubMed]
- Beare, N.A.; Lewallen, S.; Taylor, T.E.; Molyneux, M.E. Redefining cerebral malaria by including malaria retinopathy. Future Microbiol. 2011, 6, 349–355. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- World Health Organization. Malaria Fact Sheet No. 94. 2014. Available online: https://reliefweb.int/report/world/malaria-fact-sheet-no-94-updated-march-2014 (accessed on 28 August 2014).
- Fenton Hall, B.; Antony, S. Malaria control, elimination and eradication: The role of the evolving biomedical research agenda. J. Infect. Dis. 2009, 200, 1639–1643. [Google Scholar] [CrossRef] [PubMed]
- Bartoloni, A.; Zammarchi, L. Clinical aspects of uncomplicated and severe malaria. Mediterr. J. Hematol. Infect. Dis. 2012, 4, e2012026. [Google Scholar] [CrossRef] [PubMed]
- Douglas, N.M.; Anstrey, N.M.; Buffet, P.A.; Poesspoprodjo, J.; Yeo, T.W.; Whiten, N.J. The anaemia of plasmodium vivax malaria. Malar. J. 2012, 11, 135. [Google Scholar] [CrossRef] [PubMed]
- Moore, E.M.; Lockwood, D.N. Treatment of visceral leishmaniasis. J. Glob. Infect. Dis. 2010, 2, 151–158. [Google Scholar] [PubMed]
- World Health Organization. Leishmaniasis Magnitude of the Problem. Available online: http://www.who.int/leishmaniasis/en/ (accessed on 17 February 2014).
- Lozano, R. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2095–2128. [Google Scholar] [CrossRef]
- Ejazi, S.A.; Ali, N. Developments in diagnosis and treatment of visceral leishmaniasis during the last decade and future prospects. Expert Rev. Anti-Infect. Ther. 2013, 11, 79–98. [Google Scholar] [CrossRef] [PubMed]
- Dorlo, T.P.; Balasegaram, M.; Beijnen, J.H.; de Vries, P.J. Miltefosine: A review of its pharmacology and therapeutic efficacy in the treatment of leishmaniasis. J. Antimicrob. Chemother. 2012, 67, 2576–2597. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, H.; Bernstein, C.; Michod, R.E. Sex in microbial pathogens. Infect. Genet. Evol. 2018, 57, 8–25. [Google Scholar] [CrossRef] [PubMed]
- Xiao, S.H. Mefloquine, a new type of compound against schistosomes and other helminthes in experimental studies. J. Parasitol. Res. 2013, 112, 3723–3740. [Google Scholar] [CrossRef] [PubMed]
- Thétiot-Laurent, S.A.; Boissier, J.; Robert, A.; Meunier, B. Schistosomiasis chemotherapy. Angew. Chem. Int. Ed. 2013, 52, 7936–7956. [Google Scholar] [CrossRef] [PubMed]
- Gryseels, B.; Polman, K.; Clerinx, J.; Kestens, L. Human schistosomiasis. Lancet 2006, 368, 1106–1118. [Google Scholar] [CrossRef]
- Colley, D.G.; Bustinduy, A.L.; Secor, W.E.; King, C.H. Human schistosomiasis. Lancet 2014, 383, 2253–2264. [Google Scholar] [CrossRef] [Green Version]
- Cook, T.B.; Brenner, L.A.; Cloninger, C.R.; Langenberg, P.; Igbide, A.; Giegling, I.; Hartmann, A.M.; Konte, B.; Friedl, M.; Brundin, L.; et al. Latent infection with Toxoplasma gondii: Association with trait aggression and impulsivity in healthy adults. Psychiatry Res. 2015, 60, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Webster, J.P.; Kaushik, M.; Bristow, G.C. Toxoplasma gondii infection, from predation to schizophrenia: Can animal behaviour help us understand human behaviour. J. Exp. Biol. 2013, 216, 99–112. [Google Scholar] [CrossRef] [PubMed]
- Parlog, A.; Schlüter, D.; Dunay, I.R. Toxoplasma gondii-induced neuronal alterations. Parasite Immunol. 2015, 37, 159–170. [Google Scholar] [CrossRef] [PubMed]
- Blanchard, N.; Dunay, I.R.; Schlüter, D. Persistence of Toxoplasma gondii in the central nervous system: A fine-tuned balance between the parasite, the brain and the immune system. Parasite Immunol. 2015, 37, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Dubey, J.P.; Ferreira, L.R.; Martins, J.; Jones, J.L. Sporulation and survival of Toxoplasma gondii oocysts in different types of commercial cat litter. J. Parasotol. Res. 2011, 97, 751–754. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, B.; Voss, D.; Schlake, T.; Thess, A.; Kallen, K.J.; Stitz, L.; Kramps, T. Protective efficacy of in vitro synthesized, specific mRNA vaccines against influenza A virus infection. Nat. Biotechnol. 2012, 30, 1210–1216. [Google Scholar]
- Adams, M.J.; Carstens, E.B. Ratification vote on taxonomic proposals to the International Committee on Taxonomy of Viruses (2012). Arch. Virol. 2012, 157, 1411–1422. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stapleford, K.A.; Miller, D.J. Role of cellular lipids in positive-sense RNA virus replication complex assembly and function. Viruses 2010, 2, 1055–1068. [Google Scholar] [CrossRef] [PubMed]
- De Cock, K.M.; Jaffe, H.W.; Curran, J.W. The evolving epidemiology of HIV/AIDS. AIDS 2012, 26, 1205–1213. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pritchard, L.K.; Harvey, D.J.; Bonomelli, C.; Crispin, M.; Doores, K.J. Cell- and protein-directed glycosylation of native cleaved HIV-1 rnvelope. J. Virol. 2015, 89, 8932–8944. [Google Scholar] [CrossRef] [PubMed]
- Abdool Karim, S.S.; Baxter, C. Microbicides and their implications in HIV prevention. Indian J. Med. Res. 2010, 132, 656–659. [Google Scholar] [PubMed]
- Anton, P.A. Future prospects and perspectives on microbicides. Curr. HIV Res. 2012, 10, 113–115. [Google Scholar] [CrossRef] [PubMed]
- Permanyer, M.; Ballana, E.; Esté, J.A. Endocytosis of HIV: Anything goes. Trends Microbiol. 2010, 18, 543–551. [Google Scholar] [CrossRef] [PubMed]
- Gibson, R.M.; Arts, E.J. Past, present, and future of entry inhibitors as HIV microbicides. Curr. HIV Res. 2012, 10, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Nel, A.M.; Coplan, P.; Smythe, S.C. Pharmacokinetic assessment of dapivirine vaginal microbicide gel in healthy, HIV-negative women. AIDS Res. Hum. Retrovir. 2010, 26, 1181–1190. [Google Scholar] [CrossRef] [PubMed]
- Ebell, M.H.; Call, M.; Shinholser, J. Effectiveness of oseltamivir in adults: A meta-analysis of published and unpublished clinical trials. J. Fam. Pract. 2013, 30, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Robison, S.G.; Dunn, A.G.; Richards, D.L.; Leman, R.F. Changes in influenza vaccination rates after withdrawal of live vaccine. NUMA 2017, 48, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Shaman, J.; Kohn, M. Absolute humidity modulates influenza survival, transmission, and seasonality. Proc. Natl. Acad. Sci. USA 2009, 106, 3243–3248. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sheng, Z.; Ran, Z.; Wang, D.; Hoppe, A.D.; Simonson, R.; Chakravarty, S.; Hause, B.M.; Li, F. Genomic and evolutionary characterization of a novel influenza-C-like virus from swine. Arch. Virol. 2014, 159, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Udell, J.A.; Zawi, R.; Bhatt, D.L.; Keshtkar-Jahromi, M.; Gaughran, F.; Phrommintikul, A.; Ciszewski, A.; Vakili, H.; Hoffman, E.B.; Farkouh, M.E.; et al. Association between influenza vaccination and cardiovascular outcomes in high-risk patients: A meta-analysis. JAMA 2013, 310, 1711–1720. [Google Scholar] [CrossRef] [PubMed]
- Mulligan, M.J.; Bernstein, D.I.; Winokur, P.; Rupp, R.; Anderson, E.; Rouphael, N.; Dickey, M.; Stapleton, J.T.; Edupuganti, S.; Spearman, P.; et al. Serological responses to an avian influenza A/H7N9 vaccine mixed at the point-of-use with MF59 adjuvant: A randomized clinical trial. JAMA 2014, 312, 1409–1419. [Google Scholar] [CrossRef] [PubMed]
- Petsch, B.; Schnee, M.; Vogel, A.B.; Lange, E.; Salomon, R.; Webster, R.G. The influenza virus enigma. Cell 2009, 136, 402–410. [Google Scholar]
- Jit, M.; Newall, AT.; Beutels, P. Key issues for estimating the impact and cost-effectiveness of seasonal influenza vaccination strategies. Hum. Vaccin. Immunother. 2013, 9, 834–840. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Newall, A.T.; Dehollain, J.P.; Creighton, P.; Beutel, P.; Wood, J.G. Understanding the cost-effectiveness of influenza vaccination in children: Methodological choices and seasonal variability. Pharmacoeconomics 2013, 31, 693–702. [Google Scholar] [CrossRef] [PubMed]
- Raman, S.R. Fungal infections of the nervous system: current perspective and controversies in management. Int. J. Surg. 2010, 8, 591–601. [Google Scholar] [CrossRef] [PubMed]
- Kauffman, C.A.; Pappas, P.G.; Patterson, T.F. Fungal infections associated with contaminated methylprednisolone injections. N. Engl. J. Med. 2013, 368, 2495–2500. [Google Scholar] [CrossRef] [PubMed]
- Perfect, J.R.; Dismukes, W.E.; Dromer, F.; Goldman, D.L.; Graybill, J.R.; Hamill, R.J.; Harrison, T.S.; Larsen, R.A.; Lortholary, O.; Nguyen, M.H.; et al. Clinical practice guidelines for the management of cryptococcal disease: 2010 update by the infectious diseases society of America. Clin. Infect. Dis. 2010, 50, 291–322. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC). Viral Meningitis. Available online: http://www.cdc.gov/meningitis/viral.html (accessed on 5 March 2016).
- Chiang, S.S.; Khan, F.A.; Milstein, M.B.; Tolman, A.W.; Benedetti, A.; Starke, J.R.; Becerra, M.C. Treatment outcomes of childhood tuberculous meningitis: A systematic review and meta-analysis. Lancet Infect. Dis. 2014, 14, 947–957. [Google Scholar] [CrossRef]
- Durski, K.N.; Kuntz, K.M.; Yasukawa, K.; Virnig, B.A.; Meya, D.B.; Boulware, D.R. Cost-effective diagnostic checklists for meningitis in resource-limited settings. J. Acquir. Immune Dyndr. Hum. Retrovirol. 2013, 63, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Sakushima, K.; Hayashino, Y.; Kawaguchi, T.; Jackson, J.L.; Fukuhara, S. Diagnostic accuracy of cerebrospinal fluid lactate for differentiating bacterial meningitis from aseptic meningitis: A meta-analysis. J. Infect. Dis. 2011, 62, 255–262. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Centers for Disease Control and Prevention (CDC). Bacterial Meningitis. Available online: https://wikivisually.com/wiki/Meningitis (accessed on 5 March 2016).
- Chen, F.; Xu, H.; Liu, J.; Cui, Y.; Luo, X.; Zhou, Y.; Chen, Q.; Jiang, L. Efficacy and safety of nucleoside antiviral drugs for treatment of recurrent herpes labialis: A systematic review and meta-analysis. J. Oral Pathol. Med. 2017, 46, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Rotermann, M.; Langlois, K.A.; Severini, A.; Totten, S. Prevalence of Chlamydia trachomatis and herpes simplex virus type 2: Results from the 2009 to 2011 Canadian Health Measures Survey. Public Health Rep. 2013, 24, 10–15. [Google Scholar]
- Xu, F.; Sternberg, M.R.; Gottlieb, S.L.; Berman, S.M.; Markowitz, L.E. Seroprevalence of herpes simplex virus type 2 among persons aged 14–49 years—United States, 2005–2008. MMWR Morb. Mortal. Wkly. Rep. 2010, 59, 456–459. [Google Scholar]
- Xu, F.; Fujie, X.; Sternberg, M.R.; Kottiri, B.J.; McQuillan, G.M.; Lee, F.K.; Nahmias, A.J.; Berman, S.M.; Markowitz, L.E. Trends in herpes simplex virus type 1 and type 2 seroprevalence in the United States. JAMA 2006, 296, 964–973. [Google Scholar]
- Elad, S.; Zadik, Y.; Hewson, I.; Hovan, A.; Correa, M.E.; Logan, R.; Elting, L.S.; Spijkervet, F.K.; Brennan, M.T. A systematic review of viral infections associated with oral involvement in cancer patients: A spotlight on Herpesviridea. Support Care Cancer 2010, 18, 993–1006. [Google Scholar] [CrossRef] [PubMed]
- Balasubramaniam, R.; Kuperstein, A.S.; Stoopler, E.T. Update on oral herpes virus infections. Dent. Clin. N. Am. 2014, 58, 265–280. [Google Scholar] [CrossRef] [PubMed]
- Treister, N.S.; Woo, S.B. Topical n-docosanol for management of recurrent herpes labialis. Expert. Opin. Pharmacother. 2010, 11, 853–860. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Kourtis, A.P.; Ellington, S.; Legardy-Williams, J.; Bulterys, M. Safety of tenofovir during pregnancy for the mother and fetus: A systematic review. Clin. Infect. Dis. 2013, 57, 1773–1781. [Google Scholar] [CrossRef] [PubMed]
- Suk, K.T.; Kim, D.J. Drug-induced liver injury: Present and future. Clin. Mol. Hepatol. 2012, 18, 249–257. [Google Scholar] [CrossRef]
- Basra, S. Definition, epidemiology and magnitude of alcoholic hepatitis. World J. Hepatol. 2011, 3, 108–113. [Google Scholar] [CrossRef] [PubMed]
- Shi, Z.; Yang, Y.; Ma, L.; Li, X.; Schreiber, A. Lamivudine in late pregnancy to interrupt in utero transmission of hepatitis B virus: A systematic review and meta-analysis. Obstet. Gynecol. 2010, 116, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Ebert, G.; Preston, S.; Allison, C.; Cooney, J.; Toe, J.G.; Stutz, M.D.; Ojaimi, S.; Scott, H.W.; Baschuk, N. Cellular inhibitor of apoptosis proteins prevent clearance of hepatitis B virus. Proc. Natl. Acad. Sci. USA 2015, 112, 5797–5802. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Myers, R.P.; Krajden, M.; Bilodeau, M.; Kaita, K.; Marotta, P.; Peltekian, K.; Ramji, A.; Estes, C.; Razavi, H. Burden of disease and cost of chronic hepatitis C infection in Canada. Can. J. Gastroenterol. Hepatol. 2014, 28, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Younossi, Z.M.; Kanwal, F.; Saab, S.; Brown, K.A.; El-Serag, H.B.; Kim, W.R.; Ahmed, A.; Kugelmas, M.; Gordon, S.C. The impact of hepatitis C burden: An evidence-based approach. Aliment. Pharmacol. Ther. 2014, 39, 518–531. [Google Scholar] [CrossRef] [PubMed]
- Benova, L.; Mohamoud, Y.A.; Calvert, C.; Abu-Raddad, L.J. Vertical transmission of hepatitis C virus: Systematic review and meta-analysis. Clin. Infect. Dis. 2014, 59, 765–773. [Google Scholar] [CrossRef] [PubMed]
- Gadducci, A.; Barsotti, C.; Cosio, S.; Domenici, L.; Genazzani, R.A. Smoking habit, immune suppression, oral contraceptive use, and hormone replacement therapy use and cervical carcinogenesis: A review of the literature. Gynecol. Endocrinol. 2011, 27, 597–604. [Google Scholar] [CrossRef] [PubMed]
- Tran, N.P.; Hung, C.F.; Roden, R.; Wu, T.C. Control of HPV infection and related cancer through vaccination. Recent Results Cancer Res. 2014, 193, 149–171. [Google Scholar] [PubMed]
- Luhn, P.; Walker, J.; Schiffman, M.; Zuna, R.E.; Dunn, S.T.; Gold, M.A.; Smith, K.; Mathews, C.; Allen, R.A.; Zhang, R.; et al. The role of co-factors in the progression from human papillomavirus infection to cervical cancer. Gynecol. Oncol. 2013, 128, 265–270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Remschmidt, C.; Kaufmann, A.M.; Hagemann, I.; Vartazarova, E.; Wichmann, O.; Deleré, Y. Risk factors for cervical human papillomavirus infection and high-grade intraepithelial lesion in women aged 20 to 31 years in Germany. Int. J. Gynecol. Cancer 2013, 23, 519–526. [Google Scholar] [CrossRef] [PubMed]
- Mintzer, M.A.; Dane, E.L.; O’Toole, G.A.; Grinstaff, M.W. Exploiting dendrimer multivalency to combat emerging and re-emerging infectious diseases. Mol. Pharm. 2012, 9, 342–354. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, D.J.; Gould, I.M. Maximizing the impact of antimicrobial stewardship. Curr. Opin. Infect. Dis. 2013, 26, 352–358. [Google Scholar] [CrossRef] [PubMed]
- Klajnert, B.; Bryszewska, M. Dendrimers: Properties and applications. Acta Biochim. Pol. 2001, 48, 199–208. [Google Scholar] [PubMed]
- Esfand, R.; Tomalia, D.A. Poly(amidoamine) (PAMAM) dendrimers: From biomimicry to drug delivery and biomedical applications. Drug Discov. Today 2001, 6, 427–436. [Google Scholar] [CrossRef]
- World Health Organization (WHO). The Top 10 Causes of Death. Available online: http://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death (accessed on 24 September 2015).
- Reddy, M.; Gill, S.S.; Wu, W.; Kalkar, S.R.; Rochon, P.A. Does this patient have an infection of a chronic wound? JAMA 2012, 307, 605–611. [Google Scholar] [CrossRef] [PubMed]
- Svenson, S.; Tomalia, D.A. Dendrimers in biomedical applications-reflections on the field. Adv. Drug. Deliv. Rev. 2012, 64, 102–115. [Google Scholar] [CrossRef]
- Soto-Castro, D.; Cruz-Morales, J.A.; Ramírez Apan, M.T.; Guadarrama, P. Solubilization and anticancer-activity enhancement of Methotrexate by novel dendrimeric nanodevices synthesized in one-step reaction. Bioorg. Chem. 2012, 41–42, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Chahal, J.S.; Khan, O.F.; Cooper, C.L.; McPartlan, J.S.; Tsosie, J.K.; Tilley, L.D.; Sidik, S.M.; Lourido, S.; Langer, R.; Bavari, S.; et al. Dendrimer RNA nanoparticles generate protective immunity against lethal Ebola, H1N1 influenza, and Toxoplasma gondii challenges with a single dose. Proc. Natl. Acad. Sci. USA 2016, 113, 4133–4142. [Google Scholar] [CrossRef] [PubMed]
- Yandrapu, S.K.; Kanujia, P.; Chalasani, K.B.; Mangamoori, L.; Kolapalli, R.V.; Chauhan, A. Development and optimization of thiolated dendrimer as a viable mucoadhesive excipient for the controlled drug delivery: An acyclovir model formulation. Nanomed. Nanotechnol. Biol. Med. 2013, 9, 514–522. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.Y.; Rutka, J.T.; Chan, W.C. Nanomedicine. N. Engl. J. Med. 2010, 363, 2434–2443. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Kanamoto, T.; Nakashima, H.; Yoshida, T. Synthesis of a new amphiphilic glycodendrimer with antiviral functionality. Carbohydr. Polym. 2012, 90, 1061–1068. [Google Scholar] [CrossRef] [PubMed]
- Nanjwade, B.K.; Bechra, H.M.; Derkar, G.K.; Manvi, F.V.; Nanjwade, V.K. Dendrimers: Emerging polymers for drug-delivery systems. Eur. J. Pharm. Sci. 2009, 38, 185–196. [Google Scholar] [CrossRef] [PubMed]
- Gangadharan, D.; Dhandhala, N.; Dixit, D.; Thakur, R.S.; Popat, K.M.; Anand, P.S. Investigation of solid supported dendrimers for water disinfection. J. Appl. Polym. Sci. 2012, 124, 138491. [Google Scholar] [CrossRef]
- Lim, Y.; Kim, S.; Lee, Y.; Lee, W.; Yang, T.; Lee, M.; Suh, H.; Park, J. Cationic hyperbranched poly (amino ester): A novel class of DNA condensing molecule with cationic surface, biodegradable three-dimensional structure, and tertiary amine groups in the interior. J. Am. Chem. Soc. 2001, 123, 2460–2461. [Google Scholar] [CrossRef] [PubMed]
- Lalwani, S.; Chouai, A.; Perez, L.M.; Santiago, V.; Shaunak, S.; Simanek, E.E. Mimicking PAMAM dendrimers with ampholytic, hybrid triazine dendrimers: A comparison of dispersity and stability. Macromolecules 2009, 42, 6723–6732. [Google Scholar] [CrossRef] [PubMed]
- Kaur, D.; Jain, K.; Mehra, N.K.; Kesharwani, P.; Jain, N.K. A review on comparative study of PPI and PAMAM dendrimers. J. Nanopart. Res. 2016, 18, 146. [Google Scholar] [CrossRef]
- Kolhe, P.; Khandare, J.; Pillai, O.; Kannan, S.; Lieh-Lai, M.; Kannan, R.M. Preparation, cellular transport, and activity of polyamidoamine-based dendritic nanodevices with a high drug payload. Biomaterials 2006, 27, 660–669. [Google Scholar] [CrossRef] [PubMed]
- Shukla, S.K.; Govender, P.P.; Tiwari, A. Polymeric micellar structures for viosensor technology. Adv. Biomembr. Lipid Self Assem. 2016, 24, 143–161. [Google Scholar]
- Ohsaki, M.; Okuda, T.; Wada, A.; Hirayama, T.; Niidome, T.; Aoyagi, H. In vitro gene transfection using dendritic poly(l-lysine). Bioconj. Chem. 2002, 13, 510–551. [Google Scholar] [CrossRef]
- Den Boer, M.; Argaw, D.; Jannin, J.; Alvar, J. Leishmaniasis impact and treatment access. Clin. Microbiol. Infect. 2011, 17, 1471–1477. [Google Scholar] [CrossRef] [PubMed]
- Danesh-Bahreini, M.A.; Shokri, J.; Samiei, A.; Kamali-Sarvestani, E.; BarzegarJalali, M.; Mohammadi-Samani, S. Nanovaccine for leishmaniasis: Preparation of chitosan nanoparticles containing Leishmania superoxide dismutase and evaluation of its immunogenicity in BALB/c mice. Int. J. Nanomed. 2011, 6, 835–842. [Google Scholar]
- Carlsen, E.D.; Liang, Y.; Shelite, T.R.; Walker, D.H.; Melby, P.C.; Soong, L. Permissive and protective roles for neutrophils in leishmaniasis. Clin. Exp. Immunol. 2015, 182, 109–118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guerra, J.A.; Prestes, S.R.; Silveira, H.; Coelho, L.I.; Gama, P.; Moura, A.; Amato, V.; Barbosa, M.; Ferreira, L.C. Mucosal leishmaniasis caused by Leishmania (Viannia) braziliensis and Leishmania (Viannia) guyanensis in the Brazilian Amazon. PLoS Negl. Trop. Dis. 2011, 5, 980. [Google Scholar] [CrossRef] [PubMed]
- Mohamed-Ahmed, A.H.; Brocchini, S.; Croft, S.L. Recent advances in development of amphotericin B formulations for the treatment of visceral leishmaniasis. Curr. Opin. Infect. Dis. 2012, 25, 695–702. [Google Scholar] [CrossRef] [PubMed]
- Van Griensven, J.; Balasegaram, M.; Meheus, F.; Alvar, J.; Lynen, L.; Boelaert, M. Combination therapy for visceral leishmaniasis. Lancet Infect. Dis. 2010, 10, 184–194. [Google Scholar] [CrossRef]
- Ritmeijer, K.; TerHorst, R.; Chaneetal, S. Limited effectiveness of high-dose liposomal amphotericin B(AmBisome) for treatment of visceral leishmaniasis in anethiopian population with high HIV prevalence. Clin. Infect. Dis. 2011, 53, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Tiuman, T.S.; Santos, A.O.; Ueda-Nakamura, T.; Filho, B.P.D.; Nakamura, C.V. Recent advances in leishmaniasis treatment. Int. J. Infect. Dis. 2011, 15, 525–532. [Google Scholar] [CrossRef] [PubMed]
- Meheus, F.; Balasegaram, M.; Olliaroetal, P. Cost-effectiveness analysis of combination therapies for visceral leishmaniasis in the Indian subcontinent. PLoS Negl. Trop. Dis. 2010, 4, e818. [Google Scholar] [CrossRef] [PubMed]
- Menezes, J.P.B.; Almeida, T.F.; Petersen, A.L.O.A.; Guedes, C.E.S.; Mota, M.S.V.; Lima, J.G.B.; Palma, L.C.; Buck, G.A.; Krieger, M.A.; Probst, C.M.; et al. Proteomic analysis reveals differentially expressed proteins in macrophages infected with Leishmania amazonensis or Leishmania major. Microb. Infect. 2013, 15, 579–591. [Google Scholar] [CrossRef] [PubMed]
- Menezes, J.P.B.; Guedes, C.E.S.; Petersen, A.L.O.A.; Fraga, D.B.M.; Veras, P.S.T. Advances in development of new treatment for leishmaniasis. BioMed. Res. Int. 2015, 2015, 815023. [Google Scholar] [CrossRef] [PubMed]
- Croft, S.L.; Olliaro, P. Leishmaniasis chemotherapy—challenges and opportunities. Clin. Microbiol. Infect. 2011, 17, 1478–1483. [Google Scholar] [CrossRef] [PubMed]
- Jain, K.; Verma, A.K.; Mishra, P.R.; Jain, N.K. Characterization and evaluation of amphotericin B loaded conjugated poly (propylene imine) dendrimers. Nanomedicine 2015, 11, 705–713. [Google Scholar] [CrossRef] [PubMed]
- Daftarian, P.M.; Stone, G.W.; Kovalski, L.; Kumar, M.; Vosoughi, A.; Urbieta, M.; Blackwelder, P.; Dikici, E.; Serafini, P.; Duffort, S.; et al. A targeted and adjuvanted nanocarrier lowers the effective dose of liposomal amphotericin B and enhances adaptive immunity in murine cutaneous leishmaniasis. J. Infect. Dis. 2013, 208, 1914–1922. [Google Scholar] [CrossRef] [PubMed]
- Jain, K.; Verma, K.A.; Mishra, P.R.; Jaina, N.K. Surface-engineered dendrimeric nanoconjugates for macrophage targeted delivery of amphotericin B: Formulation development and in vitro and in vivo Evaluation. J. Infect. Dis. 2013, 208, 1914–1922. [Google Scholar]
- Behnke, M.S.; Wootton, J.C.; Lehmann, M.M.; Radke, J.B.; Lucas, O.; Nawas, J.; Sibley, L.D.; White, M.W. Coordinated progression through two subtranscriptomes underlies the tachyzoite cycle of Toxoplasma gondii. PLoS ONE 2010, 5, 12354. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Xing, Y.; Qu, G.; Li, X.; Dai, J.; Zhu, Y.; Zhang, X. PAMAM-Lys, a novel vaccine delivery vector, enhances the protective effects of the SjC23 DNA vaccine against schistosoma japonicum infection. PLoS ONE 2014, 9, 86578. [Google Scholar] [CrossRef] [PubMed]
- Movellan, J.; Urbán, P.; Moles, E.; de la Fuente, J.M.; Sierra, T.; Serrano, J.L.; Fernàndez-Busquets, X. Amphiphilic dendritic derivatives as nanocarriers for the targeted delivery of antimalarial drugs. Biomaterials 2014, 35, 7940–7950. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agrawal, P.; Gupta, U.; Jain, N.K. Glycoconjugated peptide dendrimers-based nanoparticulate system for the delivery of chloroquine phosphate. Biomaterials 2007, 28, 3349–3359. [Google Scholar] [CrossRef] [PubMed]
- Fomovska, A.; Wood, R.D.; Mui, E.J.; Dubey, J.P.; Ferriera, L.R.; Hickman, M.R.; Lee, P.J.; Leed, S.E.; Auschwitz, J.M.; Welsh, W.J.; et al. Salicylanilide inhibitors of Toxoplasma gondii. J. Med. Chem. 2012, 55, 8375–8391. [Google Scholar] [CrossRef] [PubMed]
- Astruc, D.; Boisselier, E.; Ornelas, C. Dendrimers designed for functions: From physical, photophysical, and supramolecular properties to applications in sensing, catalysis, molecular electronics, photonics, and nanomedicine. Chem. Rev. 2010, 110, 1857–1959. [Google Scholar] [CrossRef] [PubMed]
- Augagneur, Y.; Wesolowski, D.; Tae, H.S.; Altman, S.; Mamoun, C.B. Gene selective mRNA cleavage inhibits the development of Plasmodium falciparum. Proc. Natl. Acad. Sci. USA 2012, 109, 6235–6240. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moulton, H.M. Cell-penetrating peptides enhance systemic delivery of antisense morpholino oligomers. Methods Mol. Biol. 2012, 867, 407–414. [Google Scholar] [PubMed]
- Lai, B.; William, H.; Witola, W.H.; Bissati, K.E.; Zhou, Y.; Mui, E.; Fomovska, A.; McLeod, R. Molecular target validation, antimicrobial delivery, and potential treatment of Toxoplasma gondii infections. Proc. Natl. Acad. Sci. USA 2012, 109, 14182–14187. [Google Scholar] [CrossRef] [PubMed]
- Prieto, M.J.; Bacigalupe, D.; Pardini, O.; Amalvy, J.I.; Venturini, C.; Morilla, M.J.; Romero, E.L. Nanomolar cationic dendrimeric sulfadiazine as potential antitoxoplasmic agent. Int. J. Pharm. 2006, 326, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Grimes, J.E.T.; Croll, D.; Harrison, W.E.; Utzinger, J.; Freeman, M.C.; Templeton, M.R. The relationship between water, sanitation and schistosomiasis: A systematic review and meta-analysis. PLoS Negl. Trop. Dis. 2014, 8, e3296. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, L.D.; Guo, J.; Wu, X.; Chen, H.; Wang, T.; Zhu, S.; Zhang, Z.H.; Steinmann, P.; Yang, G.J.; Wang, S.P.; et al. China’s new strategy to block Schistosoma japonicum transmission: Experiences and impact beyond schistosomiasis. Trop. Med. Int. Health. 2009, 14, 1475–1483. [Google Scholar] [CrossRef] [PubMed]
- Fenwick, A.; Webster, J.P. Schistosomiasis: Challenges for control, treatment and drug resistance. Curr. Opin. Infect. Dis. 2006, 19, 577–582. [Google Scholar] [CrossRef] [PubMed]
- Doenhoff, M.J.; Mattoccia, P.L. Praziquantel for the treatment of schistosomiasis: Its use for control in areas with endemic disease and prospects for drug resistance. Expert Rev. Anti-Infect. 2006, 4, 199–210. [Google Scholar] [CrossRef] [PubMed]
- Sikwal, D.R.; Kalhapure, R.S.; Govender, T. An emerging class of amphiphilic dendrimers for pharmaceutical and biomedical applications: Janus amphiphilic dendrimers. Eur. J. Pharm. Sci. 2017, 97, 113–134. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Malaria Factsheet Updated January 2016. Available online: http://www.who.int/mediacentre/factsheets/fs094/en/ (accessed on 18 February 2016).
- World Health Organization (WHO). World Malaria Report 2015. Available online: http://www.who.int/malaria/media/world-malaria-report-2015/en/ (accessed on 19 April 2016).
- Baird, J.K. Evidence and implications of mortality associated with acute Plasmodium vivax malaria. Clin. Microbiol. Rev. 2013, 26, 36–57. [Google Scholar] [CrossRef] [PubMed]
- Hartman, T.K.; Rogerson, S.J.; Fischer, P.R. The impact of maternal malaria on newborns. Ann. Trop. Pediatr. 2010, 30, 271–282. [Google Scholar] [CrossRef] [PubMed]
- Taylor, W.R.; Hanson, J.; Turner, G.D.; White, N.J.; Dondorp, A.M. Respiratory manifestations of malaria. Chest 2012, 142, 492–505. [Google Scholar] [CrossRef] [PubMed]
- Aditya, N.P.; Vathsala, P.G.; Vieirra, V.; Murthym, R.S.R.; Souto, E.B. Andvances in nanomedicines for malaria treatment. Adv. Colloid Interface Sci. 2013, 201–202, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Doitsh, G.; Galloway, N.L.K.; Geng, X.; Yang, Z.; Monroe, K.M.; Zepeda, O.; Hunt, P.W.; Hatano, H.; Sowinski, S.; Muñoz-Arias, I.; et al. Cell death by pyroptosis drives CD4 T-cell depletion in HIV-1 infection. Nature 2014, 505, 509–514. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Buckheit, R.W.; Watson, K.M.; Morrow, K.M.; Ham, A.S. Development of topical microbicides to prevent the sexual transmission of HIV. Antivir. Res. 2010, 85, 142–158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peng, J.; Wu, Z.; Qi, X.; Chen, Y.; Li, X. Dendrimers as potential therapeutic tools in HIV inhibition. Molecules 2013, 18, 7912–7929. [Google Scholar] [CrossRef] [PubMed]
- Tefanidou, M.; Herrera, C.; Armanasco, N.; Shattock, R.J. Saquinavir inhibits early events associated with establishment of HIV-1 infection: Potential role for protease inhibitors in prevention. Antimicrob. Agents Chemother. 2012, 56, 4381–4390. [Google Scholar] [CrossRef] [PubMed]
- Veazey, R.S.; Ketas, T.J. Dufour, J.; Moroney-Rasmussen, T.; Green, L.C.; Klasse, P.J.; Moore, J.P. Protection of rhesus macaques from vaginal infection by vaginally delivered maraviroc, an inhibitor of HIV-1 entry via the CCR5 co-receptor. J. Infect. Dis. 2010, 202, 739–744. [Google Scholar] [CrossRef] [PubMed]
- De Clercq, E. Where rilpivirine meets with tenofovir, the start of a new anti-HIV drug combination era. Biochem. Pharmacol. 2012, 84, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Tyssen, D.; Henderson, S.A.; Johnson, A.; Sterjovski, J.; Moore, K.; La, J.; Zanin, M.; Sonza, S.; Karellas, P.; Giannis, M.P.; et al. Structure activity relationship of dendrimer microbicides with dual action antiviral activity. PLoS ONE 2010, 5, e12309. [Google Scholar] [CrossRef] [PubMed]
- Mallipeddi, R.; Rohan, L.C. Progress in antiretroviral drug delivery using nanotechnology. Int. J. Nanomed. 2010, 5, 533–547. [Google Scholar]
- Enrique Vacas-Córdoba, E.V.; Galán, M.; De La Mata, F.J.; Gómez, R.; Pion, M.; Muñoz-Fernández, M.A. Enhanced activity of carbosilane dendrimers against HIV when combined with reverse transcriptase inhibitor drugs: Searching for more potent microbicides. Int. J. Nanomed. 2014, 9, 3591–3600. [Google Scholar]
- Landers, J.J.; Cao, Z.; Lee, I.; Piehler, L.T.; Myc, P.P.; Myc, A.; Hamouda, T.; Galecki, A.T.; Baker, J.R. Prevention of influenza pneumonitis by sialic acid—Conjugated dendritic polymers. J. Infect. Dis. 2002, 186, 1222–1230. [Google Scholar] [CrossRef] [PubMed]
- Luganini, A.; Nicoletto, S.F.; Pizzuto, L.; Pirri, G.; Giuliani, A.; Landolfo, S.; Gribaudo, G. Inhibition of herpes simplex virus type 1 and type 2 infections by peptide-derivatized dendrimers. Antimicrob. Agents Chemother. 2011, 55, 3231–3239. [Google Scholar] [CrossRef] [PubMed]
- Dutta, T.; Burgess, M.; McMillan, N.A.; Parekh, H.S. Dendrosome-based delivery of siRNA against E6 and E7 oncogenes in cervical cancer. Nanomedicine 2010, 6, 463–470. [Google Scholar] [CrossRef] [PubMed]
- Mekuria, S.L.; Debele, T.A.; Chou, H.Y.; Tsai, H.C. IL-6 antibody and RGD peptide conjugated poly (amidoamine) dendrimer for targeted drug delivery of HeLa cells. J. Phys. Chem. B 2016, 120, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Sepúlveda-Crespo, D.; Sánchez-Rodríguez, J.; Serramía, M.J.; Gómez, R.; De La Mata, F.J.; José Luis Jiménez, J.L.; Muñoz-Fernández, M.A. Triple combination of carbosilane dendrimers, tenofovir and maraviroc as potential microbicide to prevent HIV-1 sexual transmission. Nanomedicine 2015, 10, 899–914. [Google Scholar] [CrossRef] [PubMed]
- Ceña-Dıez, R.; Sepúlveda-Crespo, D.; Malyde, M.; Muñoz-Fern´andez, M. Dendrimeric based microbicides against sexual transmitted infections associated to heparin sulfate. RSC Adv. 2016, 6, 46755–46764. [Google Scholar] [CrossRef]
- Ken Hatano, K.; Teruhiko Matsubara, T.; Yosuke Muramatsu, Y.; Masakazu Ezure, M.; Tetsuo Koyama, T.; Koji Matsuoka, K.; Ryunosuke Kuriyama, R.; Haruka Kori, H.; Toshinori Sato, T. Synthesis and influenza virus inhibitory activities of carbosilane dendrimers peripherally functionalized with hemagglutinin-binding peptide. J. Med. Chem. 2014, 57, 8332–8339. [Google Scholar] [CrossRef] [PubMed]
- Sepúlveda-Crespo, D.; Jiménez, J.L.; Gómez, R.; De La Mata, F.J.; Majano, P.L.; Muñoz-Fernández, M.; Gastaminza, P. Polyanionic carbosilane dendrimers prevent hepatitis C virus infection in cell culture. Nanomedicine 2017, 13, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Ceña-Diez, R.; Vacas-Córdoba, E.; García-Broncano, P.; de la Mata, F.J.; Gómez, R.; Maly, M.; Muñoz-Fernández, M.Á. Prevention of vaginal and rectal herpes simplex virus type 2 transmission in mice: Mechanism of antiviral action. Int. J. Nanomed. 2016, 11, 2147. [Google Scholar]
- Chonco, L.; Pion, M.; Vacas, E.; Rasines, B.; Maly, M.; Serramia, M.J.; Lopez-Fernandez, L.; de la Mata, J.; Alvarez, S.; Gomez, R.; et al. Carbosilane dendrimer nanotechnology outlines of the broad HIV blocker profile. J. Control. Release 2012, 161, 949–958. [Google Scholar] [CrossRef] [PubMed]
- Jiménez, J.L.; Clemente, M.I.; Weber, N.D.; Sanchez, J.; Ortega, P.; de la Mata, F.J.; Gómez, R.; García, D.; López-Fernández, L.A.; Muñoz-Fernández, M.Á. Carbosilane dendrimers to transfect human astrocytes with small interfering RNA targeting human immunodeficiency virus. BioDrugs 2010, 24, 331–343. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.K.; Sharma, A.; Mahajan, M.; Sankar, R. In-vitro and in-vivo evaluation of poly(propyl ether imine) (PETIM) dendrimer for sustained delivery of zdovudine. J. Antivir. Antiretrovir. 2013. [Google Scholar] [CrossRef]
- Lakshminarayanan, A.; Reddy, B.U.; Raghav, N.; Ravi, V.K.; Kumar, A.; Maiti, P.K.; Sood, A.K.; Jayaraman, N.; Das, S. A galactose-functionalized dendritic siRNA-nanovector to potentiate hepatitis C inhibition in liver cells. Nanoscale 2015, 7, 16921–16931. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tarallo, R.; Carberry, T.P.; Falanga, A.; Vitiello, M.; Galdiero, S.; Galdiero, M.; Weck, M. Dendrimers functionalized with membrane-interacting peptides for viral inhibition. Int. J. Nanomed. 2013, 8, 521–534. [Google Scholar] [Green Version]
- Zhou, J.; Neff, C.P.; Liu, X.; Zhang, J.; Li, H.; Smith, D.D.; Swiderski, P.; Aboellail, T.; Huang, Y.; Du, Q.; et al. Systemic administration of combinatorial dsiRNAs via nanoparticles efficiently suppresses HIV-1 infection in humanized mice. Mol. Ther. 2011, 19, 2228–2238. [Google Scholar] [CrossRef] [PubMed]
- Telwatte, S.; Moore, K.; Johnson, A.; Tyssen, D.; Sterjovski, J.; Aldunate, M.; Gorry, P.R.; Ramsland, P.A.; Lewis, G.R.; Paull, J.R.; et al. Virucidal activity of the dendrimer microbicide SPL7013 against HIV-1. Antivir. Res. 2011, 90, 195–199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Han, S.; Yoshida, D.; Kanamoto, T.; Nakashima, H.; Uryu, T.; Yoshida, T. Sulfated oligosaccharide cluster with polylysine core scaffold as a new anti-HIV dendrimer. Carbohydr. Polym. 2010, 80, 1111–1115. [Google Scholar] [CrossRef]
- Borges, A.R.; Wieczorek, L.; Johnson, B.; Benesi, A.J.; Brown, B.K.; Kensinger, R.D.; Krebs, F.C.; Wigdahl, B.; Blumenthal, R.; Puri, A.; et al. Multivalent dendrimeric compounds containing carbohydrates expressed on immune cells inhibit infection by primary isolates of HIV-1. Virology 2010, 408, 80–88. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Doménech, R.; Abian, O.; Bocanegra, R.; Correa, J.; Sousa-Hervesm, A.; Riguera, R.; Mateu, M.G.; Fernandez-Megia, E.; Velázquez-Campoy, A.; Neira, J.L. Dendrimers as potential inhibitors of the dimerization of the capsid protein of HIV-1. Biomacromolecules 2010, 11, 2069–2078. [Google Scholar] [CrossRef] [PubMed]
- Price, C.F.; Tyssen, D.; Sonza, S.; Davie, A.; Evans, S.; Lewis, G.R.; Xia, S.; Spelman, T.; Hodsman, P.; Moench, T.R.; et al. SPL7013 Gel (VivaGel®) retains potent HIV-1 and HSV-2 inhibitory activity following vaginal administration in humans. PLoS ONE 2011, 6, e24095. [Google Scholar] [CrossRef] [PubMed]
- Ionov, M.; Ciepluch, K.; Klajnert, B.; Glińska, S.; Gomez-Ramirez, R.; de la Mata, F.J.; Munoz-Fernandez, M.A.; Bryszewska, M. Complexation of HIV derived peptides with carbosilane dendrimers. Colloids Surf. B. Biointerfaces 2013, 101, 236–242. [Google Scholar] [CrossRef] [PubMed]
- De Las Cuevas, N.; Garcia-Gallego, S.; Rasines, B.; de la Mata, F.J.; Guijarro, G.L.; Munoz-Fernandez, M.A.; Gómez, R. In vitro studies of water-stable cationic carbosilane dendrimers as delivery vehicles for gene therapy against HIV and hepatocarcinoma. Curr. Med. Chem. 2012, 19, 5052–5061. [Google Scholar] [CrossRef] [PubMed]
- Briz, V.; Serramia, M.; Madrid, R.; Hameau, A.; Caminade, A.-M.; Majoral, J.; Munoz-Fernandez, M. Validation of a generation 4 phosphorus-containing polycationic dendrimer for gene delivery against HIV-1. Curr. Med. Chem. 2012, 19, 5044–5051. [Google Scholar] [CrossRef] [PubMed]
- Suazo, P.A.; Tognarelli, E.I.; Kalergis, A.M.; González, P.A. Herpes simplex virus 2 infection: Molecular association with HIV and novel microbicides to prevent disease. Med. Microbiol. Immunol. 2015, 204, 161–176. [Google Scholar] [CrossRef] [PubMed]
- Noguchi, L.; Hillier, S.; Richardson, B.; Chirenje, Z.; Balkus, J.; Piper, J.; Marrazzo, J. 018-2 Injectable progestin contraception and acquisition of hsv-2 infection among South African women participating in the voice trial. Sex. Transm. Infect. 2015, 91, e65. [Google Scholar] [CrossRef]
- Bastien, S.; Mason-Jones, A.; De Koker, P.; Mmbaga, E.; Ross, D.; Mathews, C. Herpes simplex virus type 2 infection as a biomarker for sexual debut among young people in sub-Saharan Africa: A literature review. Int. J. STD AIDS 2012, 23, e761. [Google Scholar] [CrossRef] [PubMed]
- Astruc, D.; Liang, L.; Rapakousiou, A.; Ruiz, J. Click dendrimers and triazole-related aspects: Catalysts, mechanism, synthesis, and functions. A bridge between dendritic architectures and nanomaterials. Acc. Chem. Res. 2012, 45, 630–640. [Google Scholar] [CrossRef] [PubMed]
- Carberry, T.P.; Tarallo, R.; Falanga, A.; Finamore, E.; Galdiero, M.; Weck, M.; Galdiero, S. Dendrimer functionalization with a membrane-interacting domain of herpes simplex virus type 1: Towards intracellular delivery. Chemistry 2012, 18, 13678–13685. [Google Scholar] [CrossRef] [PubMed]
- Tarallo, R.; Accardo, A.; Falanga, A.; Guarnieri, D.; Vitiello, G.; Netti, P.; D’Errico, G.; Morelli, G.; Galdiero, S. Clickable functionalization of liposomes with the gH625 peptide from Herpes simplex virus type I for intracellular drug delivery. Chemistry 2011, 17, 12659–12668. [Google Scholar] [CrossRef] [PubMed]
- Shivkumar, S.; Peeling, R.; Jafari, Y.; Joseph, L.; Pant Pai, N. Accuracy of rapid and point-of-care screening tests for hepatitis C: A systematic review and meta-analysis. Ann. Intern. Med. 2012, 157, 558–566. [Google Scholar] [CrossRef] [PubMed]
- Singh, S. Nanomedicine-nanoscale drugs and delivery systems. J. Nanosci. Nanotechnol. 2010, 10, 7906–7918. [Google Scholar] [CrossRef] [PubMed]
- Walter, M.V.; Malkoch, M. Simplifying the synthesis of dendrimers: Accelerated approaches. Chem. Soc. Rev. 2012, 41, 4593–4609. [Google Scholar] [CrossRef] [PubMed]
- Heegaard, P.M.H.; Boas, U.; Sorensen, N.S. Dendrimers for vaccine and immunostimulatory uses: A review. Bioconjug. Chem. 2010, 21, 405–418. [Google Scholar] [CrossRef] [PubMed]
- Khosravy, M.S.; Ardestani, M.S.; Cohan, R.A.; Doroud, D.; Amini, S.; Momen, S.B.; Atyabi, S.M.; Heydari, H.; Vahabpour, R. Design, synthesis, physicochemical and immunological characterization of dendrimer-HBsAg conjugate. Vacres 2014, 1, 24–28. [Google Scholar] [CrossRef]
- Beck, C.R.; McKenzie, B.C.; Hashim, A.B.; Harris, R.C.; Nguyen-Van-TDesam, J.S. Influenza vaccination for immunocompromised patients: Systematic review and meta-analysis by etiology. J. Infect. Dis. 2012, 206, 1250–1259. [Google Scholar] [CrossRef] [PubMed]
- Newall, A.T.; Kelly, H.; Harsley, S.; Scuffham, P.A. Cost effectiveness of influenza vaccination in older adults: A critical review of economic evaluations for the 50- to 64-year age group. Pharmacoeconomics 2009, 27, 439–450. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, F.; Lindley, M.C.; Allred, N.; Weinbaum, C.M.; Grohskopf, L. Effect of influenza vaccination of healthcare personnel on morbidity and mortality among patients: Systematic review and grading of evidence. Clin. Infect. Dis. 2014, 58, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Dolan, G.P.; Harris, R.C.; Clarkson, M.; Sokal, R.; Morgan, G.; Mukaigawara, M.; Horiuchi, H.; Hale, R.; Stormont, L.; Béchard-Evans, L.; et al. Vaccination of healthcare workers to protect patients at increased risk of acute respiratory disease: Summary of a systematic review. Influenza Other Respir. Viruses 2013, 7, 93–96. [Google Scholar] [CrossRef] [PubMed]
- Shazly, S.A.M.; Murad, M.H.; Dowdy, S.C.; Gostout, B.S.; Famuyide, A.O. Robotic radical hysterectomy in early stage cervical cancer: A systematic review and meta-analysis. Gynecol. Oncol. 2015, 138, 457–471. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, P. Cervical cancer: Can it be prevented? World. J. Clin. Oncol. 2014, 5, 775–780. [Google Scholar] [CrossRef] [PubMed]
- Lorusso, D.; Petrelli, F.; Coinu, A.; Raspagliesi, F.; Barni, S. A systematic review comparing cisplatin and carboplatin plus paclitaxel-based chemotherapy for recurrent or metastatic cervical cancer. Gynecol. Oncol. 2014, 133, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Pfaendler, K.S.; Tewari, K.S. Changing paradigms in the systemic treatment of advanced cervical cancer. Am. J. Obstet. Gynecol. 2016, 214, 22–30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Joshi, J.R.; Patel, R.P. Role of biodegradable polymers in drug delivery. Int. J. Curr. Pharm. Res. 2012, 4, 74–81. [Google Scholar]
- Kesharwani, P.; Banerjee, S.; Gupta, U.; Mohd Amin, M.C.I.; Padhye, S.; Sarkar, F.H.; Iyer, A.K. Pamam dendrimers as promising nanocarriers for RNAi therapeutics. Mater. Today 2015, 18, 565–572. [Google Scholar] [CrossRef]
- Liu, T.Y.; Hussein, W.M.; Jia, Z.; Ziora, Z.M.; McMillan, N.A.; Monteiro, M.J.; Toth, I.; Skwarczynski, M. Self-adjuvanting polymer-peptide conjugates as therapeutic vaccine candidates against cervical cancer. Biomacromolecules 2013, 14, 2798–2806. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hussein, W.M.; Liu, T.Y.; Jia, Z.; McMillan, N.A.J.; Monteiro, M.J.; Toth, I.; Skwarczynski, M. Multiantigenic peptide-polymer conjugates as therapeutic vaccines against cervical cancer. Bioorg. Med. Chem. 2016, 24, 4372–4380. [Google Scholar] [CrossRef] [PubMed]



















| Dendrimers Classification | Combination with Type of Drugs | Type of Infection | References |
|---|---|---|---|
| PPI | Amphotericin B | Leishmaniasis | [116,118] |
| PAA | Sulfadoxine, chloroquine and primaquine | Toxoplamosis | [118,119] |
| PAMAM | DNA Chloroquine and primaquine | Schistosomiasis Malaria | [120] [121] |
| Poly-l-lysine | Chloroquine | Malaria | [122] |
| Dendrimers Classification | Combination with Type of Drugs | Type of Infection | References |
|---|---|---|---|
| PAA | DNA | Influenza | [149] |
| Peptide dendrimers | Acyclovir | Herpes | [150] |
| siRNA | Cervical cancer | [151] | |
| Doxorubicin | Cervical cancer | [152] | |
| Carbosilane dendrimers | Zidovudine, efarvenz and tenofovir | HIV | [148] |
| Maraviroc and tenofovir | HIV | [153] | |
| Heparan sulfate | Herpes | [154] | |
| Oseltamivir | Influenza | [155] | |
| Sofosbuvir | Hepatitis | [156] | |
| Acyclovir and tenofovir | Herpes | [157] | |
| Microbicide | HIV | [158] | |
| siRNA | HIV | [159] | |
| PPI | Zidovudine | HIV | [160] |
| PETIM | siRNA | Hepatitis | [161] |
| PA | Glycoprotein H | Herpes | [162] |
| PAMAM | Heparan sulphate | Herpes | [154] |
| siRNA | HIV | [163] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mhlwatika, Z.; Aderibigbe, B.A. Application of Dendrimers for the Treatment of Infectious Diseases. Molecules 2018, 23, 2205. https://doi.org/10.3390/molecules23092205
Mhlwatika Z, Aderibigbe BA. Application of Dendrimers for the Treatment of Infectious Diseases. Molecules. 2018; 23(9):2205. https://doi.org/10.3390/molecules23092205
Chicago/Turabian StyleMhlwatika, Zandile, and Blessing Atim Aderibigbe. 2018. "Application of Dendrimers for the Treatment of Infectious Diseases" Molecules 23, no. 9: 2205. https://doi.org/10.3390/molecules23092205