Structural Aspects LiNbO3 Nanoparticles and Their Ferromagnetic Properties
Abstract
:1. Introduction
2. Results and Discussion
2.1. Differential Scanning Calorimetry

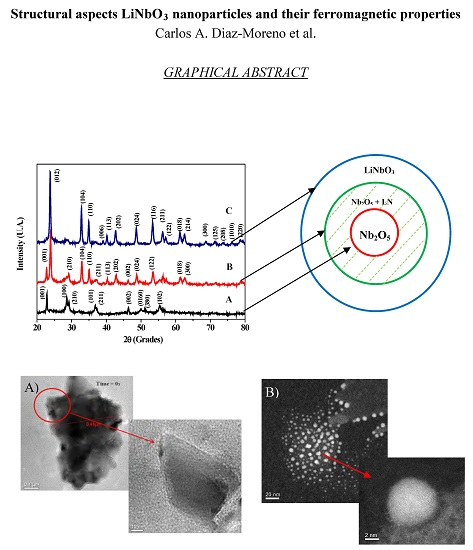
2.2. Powder X-ray Diffraction Pattern

2.3. Magnetometry Measurements

2.4. Structural Aspects by Cs-Corrected Scanning Electron Microscope



3. Experimental Section
3.1. LiNbO3 Nanoparticle Preparation

3.2. Powder X-ray Diffraction
3.3. Magnetometry Measurements
3.4. Experimental HRTEM Microanalysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zhu, X. Recent patents on perovskite ferroelectric nanostructures. Recent Pat. Nanotechnol. 2009, 3, 42–52. [Google Scholar] [PubMed]
- Xue, D.; Betzler, K.; Hesse, H. Dielectric properties of lithium niobate and tantalate crystals. Solid State Commun. 2000, 115, 581–585. [Google Scholar] [CrossRef]
- Volk, T.; Wöhlecke, M. Lithium Niobate, 1st ed.; Springer-Verlag: Berlin, Germany, 2008; p. 258. [Google Scholar]
- Knabe, B.; Schütze, D.; Jungk, T.; Svete, M.; Assenmacher, W.; Mader, W.; Buse, K. Synthesis and characterization of Fe-doped LiNbO3 nanocrystals from a triple-alkoxide method. Phys. Status Solidi A 2011, 208, 857–862. [Google Scholar] [CrossRef]
- Rüdiger, A.; Waser, R. Size effects in nanoscale ferroelectrics. J. Alloys Compd. 2008, 449, 2–6. [Google Scholar]
- Hölscher, R.; Schmidt, W.G.; Sanna, S. Modelling LiNbO3 surfaces at ambient conditions. Phys. Rev. C 2014, 118, 10213–10220. [Google Scholar]
- Veithen, M.; Ghosez, Ph. First-principles study of the dielectric and dynamical properties of lithium niobate. Phys. Rev. B 2002, 65. [Google Scholar] [CrossRef]
- Wei, D.T.Y.; Lee, W.W.; Bloom, L.R. Large refractive index change induced by ion implantation in lithium niobate. Appl. Phys. Lett. 1974, 25, 329–331. [Google Scholar] [CrossRef]
- Kong, Y.; Liu, S.; Xu, J. Recent advances in the photorefraction of doped lithium niobate crystals. Materials 2012, 5, 1954–1971. [Google Scholar] [CrossRef]
- Sundaresan, A.; Rao, C.N.R. Ferromagnetism as a universal feature of inorganic nanoparticle. Nanotoday 2009, 4, 96–106. [Google Scholar]
- Song, C.; Zeng, F.; Shen, Y.X.; Geng, K.W.; Xie, Y.N.; Wu, Z.Y.; Pan, F. Local Co structure and ferromagnetism in ion-implanted Co-doped LiNbO3. Phys. Rev. B 2006, 73. [Google Scholar] [CrossRef]
- Díaz-Moreno, C.A.; Farías-Mancilla, R.; Matutes-Aquino, J.A.; Elizalde-Galindo, J.; Espinosa-Magaña, F.; González-Hernández, J.; Hurtado-Macías, A. Magnetic behavior in LiNbO3 nanocrystallites caused by oxygen vacancies. J. Magn. Magn. Mater. 2014, 356, 82–86. [Google Scholar] [CrossRef]
- Díaz-Moreno, C.; Farias, R.; Hurtado-Macias, A.; Elizalde-Galindo, J.; Hernandez-Paz, J. Multiferroic response of nanocrystalline lithium niobate. J. Appl. Phys. 2012, 111. [Google Scholar] [CrossRef]
- Zhou, W.; Oxley, M.P.; Lupini, A.R.; Krivanek, O.L.; Pennycook, S.J.; Idrobo, J.-C. Single atom microscopy. Microsc. Microanal. 2012, 18, 1342–1354. [Google Scholar] [PubMed]
- Borisevich, A.Y.; Lupini, A.R.; He, J.; Eliseev, E.A.; Morozovska, A.N.; Svechnikov, G.S.; Yu, P.; Chu, Y.-H.; Ramesh, R.; Pantelides, S.T.; et al. Interface dipole between two metallic oxides caused by localized oxygen vacancies. Phys. Rev. B 2012, 86. [Google Scholar] [CrossRef]
- Sepulveda-Guzman, S.; Elizondo-Villarreal, N.; Ferrer, D.A.; Torres-Castro, A.; Gao, X.; Zhou, J.P.; José-Yacamán, M. In situ formation of bismuth nanoparticles trough beam irradiation in transmission electron microscope. Nanotechnology 2007, 18. [Google Scholar] [CrossRef] [PubMed]
- Shimada, S.; Kodaira, K.; Matsuchita, T. A study of the formation of LiNbO3 in the system Li2CO3-Nb2O5. Thermochim. Acta 1978, 23, 135–144. [Google Scholar]
- Stamenov, C.P.; Gunning, R.D.; Venkatesanand, M.; Paul, K. Ferromagnetism in defect-ridden oxides and related materials. New J. Phys. 2010, 12. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Diaz-Moreno, C.A.; Farias-Mancilla, R.; Elizalde-Galindo, J.T.; González-Hernández, J.; Hurtado-Macias, A.; Bahena, D.; José-Yacamán, M.; Ramos, M. Structural Aspects LiNbO3 Nanoparticles and Their Ferromagnetic Properties. Materials 2014, 7, 7217-7225. https://doi.org/10.3390/ma7117217
Diaz-Moreno CA, Farias-Mancilla R, Elizalde-Galindo JT, González-Hernández J, Hurtado-Macias A, Bahena D, José-Yacamán M, Ramos M. Structural Aspects LiNbO3 Nanoparticles and Their Ferromagnetic Properties. Materials. 2014; 7(11):7217-7225. https://doi.org/10.3390/ma7117217
Chicago/Turabian StyleDiaz-Moreno, Carlos A., Rurik Farias-Mancilla, Jose T. Elizalde-Galindo, Jesus González-Hernández, Abel Hurtado-Macias, Daniel Bahena, Miguel José-Yacamán, and Manuel Ramos. 2014. "Structural Aspects LiNbO3 Nanoparticles and Their Ferromagnetic Properties" Materials 7, no. 11: 7217-7225. https://doi.org/10.3390/ma7117217