1. Introduction
Dextranase (1,6-α-
d-glucan-6-glucanohydrolase; E.C. 3.2.1.11) is an inducible enzyme and can specially catalyse the endohydrolysis of α-(1,6)-
d-glycoside linkages at random sites of dextran. The main products are isomaltose, isomaltotriose, small amounts of
d-glucose and a series of low molecular-mass polysaccharides [
1]. Dextranase has been widely used in the production of specific clinical dextrans [
2,
3]. Dextran molecules produced by dextranase with relatively low weight-average molecular mass (
Mw) of 20–70 kDa can be used as blood extenders and those with
Mw of 6–8 kDa can form complexes with iron to treat severe anaemia [
4]. Since dextranase can effectively degrade dextran in dental plaques, the use of this enzyme has become an effective way to treat dental caries [
5,
6,
7,
8].
Currently, bacteria and fungi are two of the main sources to produce dextranases [
9]. Dextranases from bacteria usually have favorable thermal stability but relatively low dextranase activity. Hide et al. [
10] obtained a dextranase from
Paenibacillus sp. mutant with the optimal temperature of 60 °C. Rashida et al. [
11] purified a dextranase from
Bacillus licheniformis KIBGE-IB25, of which the specific activity was 1405 U/mg. Compared with dextranases from bacteria, those from fungi may achieve higher activities. Wu et al. [
4] obtained a dextranase from
Hypocrea lixii F1002, of which the specific enzyme activity was 2782 U/mg. Siwames et al. [
12] acquired a dextranase from
Aspergillus allahabadii X26, of which the specific activity reached 3009 U/mg. Dextranases from
Penicillium have been found to have high dextranase activity as well as excellent temperature and pH stability. Zhang et al. [
13] screened
Talaromyces pinophilus H6 from the soil, which was found to be stable at 35–60 °C and pH of 3.0–10.0. This special dextranase displayed activity as high as 14,894 U/mg.
Previous research about dextranases has mostly focused on the mutation [
9], screening [
14], construction of genetically engineered bacteria [
15,
16], the optimization of fermentation medium [
17] and fermentation conditions [
18] and the purification and enzymatic properties [
19,
20,
21]. To the best of our knowledge, there are few studies on how to regulate molecular mass of dextran during production. Since the molecular mass of dextrans largely determines their applications, it is essential to find an efficient way to control this parameter. Besides, we found that the determination of the molecular mass of dextrans was inaccurate in most reports [
4,
13]. In these reports, weight-average molecular weight (
Mw) was detected and determined by a refractive index detector and its accuracy relies on the flow rate and the standard curve. However, the standard curves were plotted according to the relationship between retention time and peak molecular weight (
Mp). This means that the determination of
Mw depended on the calculation of measured
Mp. Since the molecular mass distribution of dextran was uneven during the degradation,
Mp can hardly match
Mw. This might cause great errors in the measurement. Moreover, it is also worth mentioning that in most of the previous studies, the molecular mass distribution of dextrans was not measured, despite that this parameter is important in that it directly reflects the quality of dextran. The polydispersity index, which is defined as the ratio of weight-average molecular weight and number-average molecular weight (
Mw/
Mn), is an index to describe the molecular mass distribution.
In this work, a novel dextranase was purified from Penicillium cyclopium CICC-4022 and its properties were characterized. The effects of temperature, pH and metal ions on dextranase activity were evaluated. The dextranase was then applied to degrade dextran. We found that Mw of the degraded dextrans could be effectively controlled by adjusting the reaction time, concentrations of the dextranase and the substrate concentration during the degradation. Mw and Mw/Mn of the degraded dextrans was analysed using gel permeation chromatography (GPC) coupled with multiple-angle laser light scattering (GPC-MALS).
3. Materials and Methods
3.1. Materials
Dextrans T3, T5, T20, T40, T70, T500 and T2000 (Mw ≈ 3, 5, 20, 40, 70, 500 and 2000 kDa) and a series of standards (dextran 1.27, 5.52, 11.6, 23.8, 48.6, 80.9, 273.0, 667.8 kDa) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Sepharose 6B was a product from Henghui (Beijing, China). The protein marker was obtained from ShineGene Molecular Biotech (Shanghai, China). Penicillium cyclopium (CICC-4022) was purchased from the China Centre of Industrial Culture Collection (Beijing, China). All other chemicals were of analytical grade and purchased from Sinopharm Chemical Reagent Co., Ltd. (Beijing, China).
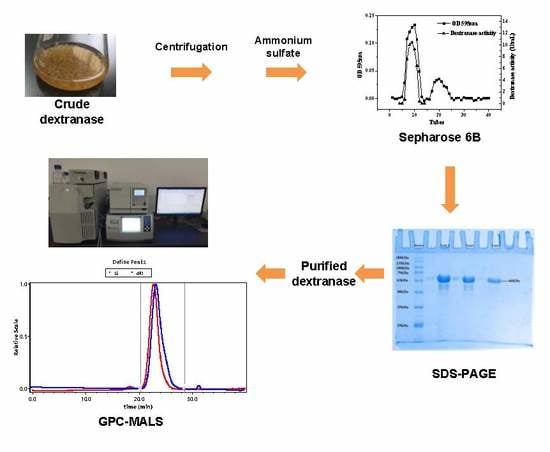
3.2. Preparation of Crude Dextranase
According to our preliminary experiments with dextran, sucrose, glucose, maltose and fructose for optimized carbon sources, we have assumed that that α-1,6 glycosidic bonds in dextran played an inducing role in the production of dextranase. A Czapek Dox medium was used as the solid medium and seed culture medium for Penicillium cyclopium. The fermentation medium was prepared by dextran T20 30 g/L, yeast extract 4 g/L, KCl:FeSO4·7H2O = 10:1 (concentration ratio) and K2HPO4 1.0 g/L. The mixture had an initial pH of 6.1. 60 mL of the fermentation medium was transferred into a 250mL flask, which was then sterilized at 121 °C for 30 min. Inoculum concentration was set as 3% and the fermentation medium was incubated in a rotary shaker at 160 r/min and 30 °C for 72 h. The thalli of Penicillium cyclopium were separated by centrifugation for 20 min at 4 °C and 10,000× g. The supernatant was stored at 4 °C for further dextranase purification.
3.3. Dextranase Activity Assay
Dextranase activity was assayed by the determination of the amount of substance of reducing sugar from Dextran T70. The diluted dextranase of 1.0 mL was incubated with 1.0 mL 3% dextran T70 (preparation with 0.02 M acetate buffer of pH 5) at 50 °C for 10 min, the amount of reducing sugar was obtained using the 3,5-dinitrosalicylic acid method as reported previously [
31]. One unit (1 U) of dextranase activity was defined as the amount of enzyme that degrades dextran T70 to produce 1 µmol glucose equivalent per min at 50 °C and pH of 5 [
32] and calculated according to Equation (1).
3.4. Determination of Protein Concentration
Protein concentration (mg/mL) was measured by the Bradford method [
33]. Crystalline bovine serum albumin was used as the protein standard.
3.5. Purification of Dextranase
The cell-free filtrate (CFF) was subjected to ammonium sulfate fractionation with a saturation of 20–90%. The precipitate was collected by centrifugation at 10,000×
g for 20 min at 4 °C. Then the precipitate was dissolved and dialyzed in 0.02 M acetate buffer (pH 5.0) overnight at 4 °C. The concentrated dextranase was loaded on a Sepharose 6B column (1.5 cm i.d. × 50 cm) and the dextranase was eluted with 0.02 M acetate buffer (pH 5.0) at a flow rate of 0.2 mL/min. The absorbance of each fraction (3 mL) was determined at 595 nm to monitor the proteins during chromatographic separation. The fractions with high dextranase activity were gathered to measure their dextranase activity and protein concentration as described in
Section 3.3 and
Section 3.4. Specific activity was calculated according to the above-measured results. The purity of the dextranase and its
Mw were determined by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE). The unstained protein marker was used as control samples,
Mw of the dextranase was analysed by a 5% concentrated gel and a 12% separated gel. Following electrophoresis, the gel was stained with Coomassie blue R-250 and decolorized several times with a destainer.
3.6. Dextranase Enzymatic Properties
3.6.1. Effect of Temperature and pH on Dextranase Activity and Stability
For investigating the effect of temperature, dextran T70 (30 mg/mL) and the dextranase were prepared with 0.02M acetate buffer (pH = 5.0). The effect of temperature on dextranase activity was measured at 35–65 °C. Relative activity was expressed as percentage of the highest activity while the highest dextranase activity is 100%. The thermal stability of dextranase was measured by exposing the dextranase in 0.02M acetate buffer (pH = 5.0) for 10–60 min at 45–65 °C without any substrate.
For studying the effect of pH, dextranase activity was determined at pH of 2.5–7.0 at 55 °C. Relative activity was expressed as percentage of the highest activity while the highest dextranase activity was 100%. The pH stability of the dextranase was evaluated by incubating the dextranase in reaction buffers at pH 2.5–7.5 at 4 °C for 1 h and 24 h respectively without any substrate.
For both thermal and pH stability, dextran T70 was added to the dextranase to measure the relative dextranase activity. The activity of non-heated and non-stored dextranase was taken as 100% dextranase activity, respectively.
3.6.2. Substrate Specificity and Analysis of Hydrolysis Products
Dextran T3, T5, T20, T40, T70, T500 and T2000, soluble starch, cellulose, sucrose, chitin, chitosan and β-cyclodextrin (30 mg/mL) were prepared with 0.02M acetate buffer (pH = 5.0) and the dextranase activity was measured with these different substrates. Relative activity was expressed as percentage of the highest activity so that the highest dextranase activity is 100%.
The final hydrolysis products of dextran T70 were analysed by high-performance liquid chromatography (HPLC) with a mobile phase of 80% acetonitrile. Glucose, isomaltose and isomaltotriose were used as the standards. The hydrolysates were diluted to 10-fold with the mobile phase and then centrifuged at 11,000× g for 20 min. The supernatant was filtered with a 0.22 μm filter and analysed by HPLC, in which a Polaris NH2 (4.6 mm × 250 nm, Agilent Technologies, Santa Clara, CA, USA) column was used. The chromatograph was operated under a flow rate of 1.0 mL/min at 35 °C and then connected to an RID-10A refractive index detector (Shimadzu Corp. Kyoto, Japan).
3.6.3. Effects of Metal ions and other Compounds on Dextranase Activity
Metal ions and reagents (namely, Cu2+ (CuSO4), Fe3+ (FeCl3), Zn2+ (ZnSO4), Ca2+ (CaCl2), Co2+ (CoCl2), K+ (KCl), Li+ (LiCl), Na+ (NaCl), Pb2+ (Pb(NO3)2), Ni2+ (NiCl2), NH4+ (NH4Cl), Fe2+ (FeSO4), Tris, SDS, urea and EDTA) were dissolved with 0.02 M acetate buffer (pH = 5.0). The concentrations of these solutions were 0.001, 0.005 and 0.01 M, respectively. The effects of different ions and reagents on dextranase activity were measured. Dextranase activity without any compounds was considered as 100% to calculate the relative activity of the above compounds.
3.6.4. Enzyme Kinetics
In order to determine the kinetic constants, the initial velocity (
v) was measured with various concentrations (0.1–0.6%) of Dextran T5, T70 and T2000 in 0.02M acetate buffer (pH = 5.0) at 55 °C for 6 min. The kinetic constants were calculated from Lineweaver–Burk plots [
34].
3.7. Determination of Mw and Mw/Mn
3.7.1. Gel permeation chromatography (GPC)
The chromatographic conditions used were as follows: the mobile phases: 0.03% NaN3 and 0.1 M NaNO3; the columns: UltrahydrogelTM 2000 (7.8 mm × 300 mm, Shimadzu, Japan), UltrahydrogelTM 250 (7.8 mm × 300 mm, Shimadzu, Japan) and UltrahydrogelTM DP120A (7.8 mm × 300 mm, Shimadzu, Japan); flow rate: 1 mL/min; the detector: Waters 2414 refractive index detector (Waters Corporation, Milford, MA, USA); column temperature: 35 °C; and detector temperature: 35°C.
The dextran standards with Mw of 1.27, 5.52, 11.6, 23.8, 48.6, 80.9, 273.0 and 667.8 kDa (Mp = 1.08, 4.44, 9.89, 21.4, 43.5, 66.7, 196.3 and 401.3 kDa, respectively) were prepared with a concentration of 1 mg/mL using the mobile phase. The samples were injected after filtration with a 0.22 μm water filter, GPC was performed to detect the retention time for different Mw. Waters Breeze 2 software was used to generate the standard curve of dextran.
Both GPC and GPC-MALS techniques were used to determine Mw and Mw/Mn of dextran. The relative errors of Mw and Mw/Mn determined by the two techniques were analysed and compared. Except that GPC-MALS had a refractive index detector and a laser detector, the two techniques used the same chromatographic conditions.
3.7.2. Degradation of dextrans by dextranase
GPC-MALS was used to measure
Mw and
Mw/
Mn of dextrans.
Mw,
Mn and
Mw/
Mn can be obtained without depending on the pump speed and the standard curve. Pawcenis et al. [
35] have used this method to measure
Mw of cellulose.
Dextran T2000 (1%, 3%, 5%) and
Penicillium cyclopium dextranase (dextranase activity: 0.2, 0.4 and 0.6 U/mL) were prepared with 0.02 M acetate buffer (pH = 5.0). The dextranase was incubated with the substrate at 50 °C. Samples were obtained at intervals, boiled for at least 3 min to stop the reaction and diluted to 10 folds with the mobile phase. The samples were filtered with a 0.22 μm water filter.
Mw and
Mw/
Mn of dextran were analysed by GPC-MALS (Waters Corporation, Milford, MA, USA; Wyatt Technology Corporation, Santa Barbara, CA, USA) according to the conditions stated in
Section 3.7.1.
4. Conclusions
In this work, we obtained a novel dextranase from Penicillium cyclopium CICC-4022. After purification to 16.09-fold concentration, the dextranase showed specific enzymatic activity of 3780.13 U/mg and molecular weight of 66 kDa. For this dextranase, the optimum temperature and pH were 55 °C and 5.0, respectively. The dextranase was stable below 45 °C under pH from 3.5 to 7. We found that this dextranase could specifically degrade α-1,6 glucosidic bonds of dextran and could be identified as an endodextranase. Besides, some metal ions such as Li+, Na+ and Fe2+ could effectively promote the dextranase activity. The obtained dextranases were then applied to degrade dextrans with excellent performance. Moreover, we found Mw of dextran could be effectively controlled by adjusting the dextranase activity, substrate concentration and reaction time during degradation. Therefore, our findings show the high potential of this dextranase from Penicillium cyclopium CICC-4022, which can be potentially applied to the production of specific dextrans for clinical applications.