-
- Academic Editors
-
-
-
†These authors contributed equally.
Background: High-grade serous ovarian cancer (HGSOC) treatment is
facing clinical challenges. The tumor immune microenvironment (TME) has recently
been shown to perform a critical function in the prediction of clinical outcomes
as well as the effectiveness of treatment. Leukocyte migration is enhanced in
malignant tumors and promotes immunity. However, its role in how to underlie the
migration of immune cells into the TME remains to be further explained in HGSOC.
Methods: We built a prognostic multigene signature with
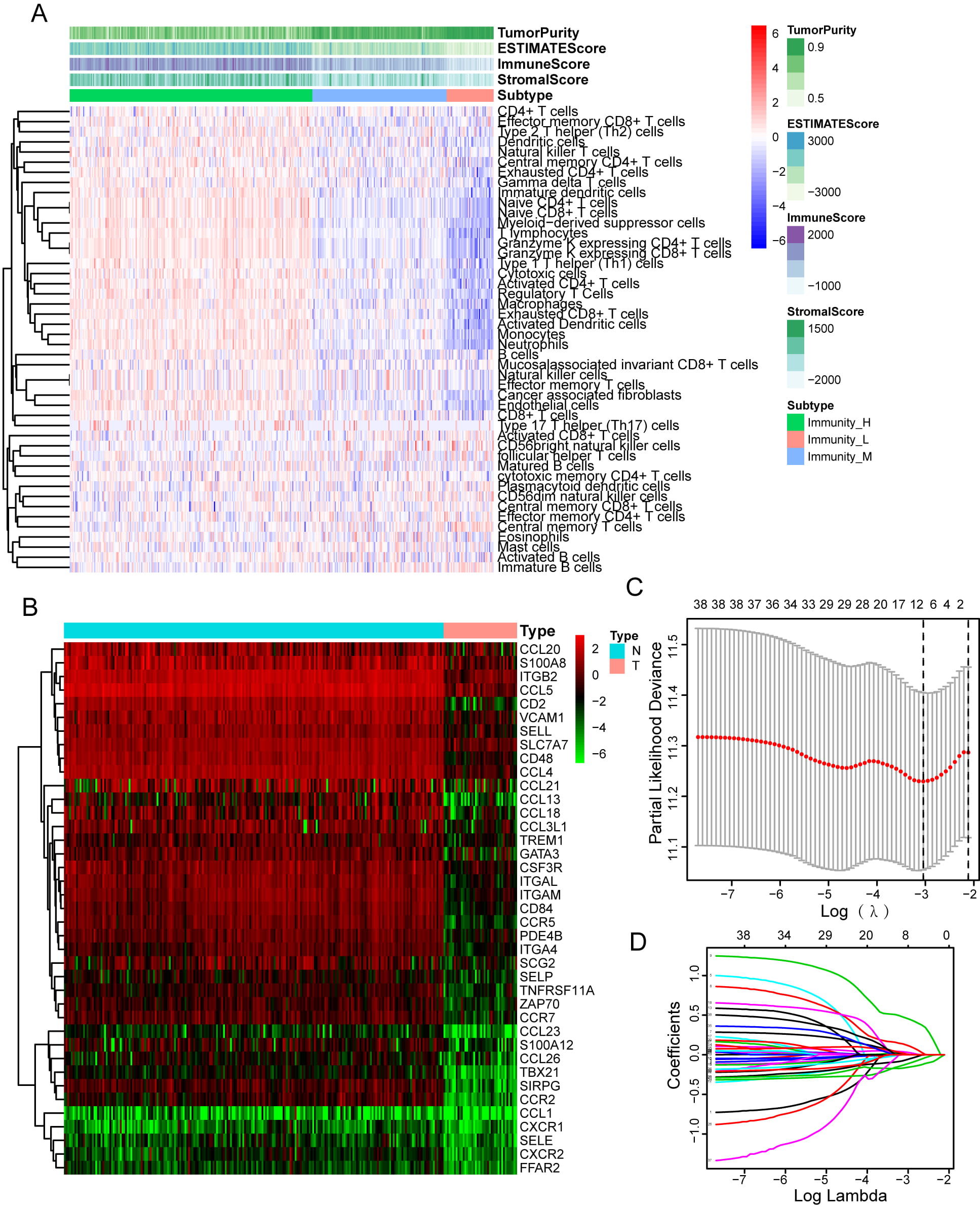
leukocyte migration-related differentially expressed genes (LMDGs), which is
associated with TME by single-sample gene set enrichment analysis (ssGSEA), in
the The Cancer Genome Atlas (TCGA) cohort. Furthermore, we systematically
correlated risk signature with immunological characteris-tics in TME, mutational
profiles of HGSOC, and potential value in predicting efficacy of platinum-based
chemotherapy and immunotherapy. Screening of the most important prognostic factor
among risk signatures by Friends analysis, and immunofluorescence was employed to
examine both the expression of CD2 as well as its relationship with CD8 and PD-1.
Results: LMDGs-related prognostic model showed good
prediction performance. Patients who had high-risk scores exhibited significantly
reduced progression-free survival (PFS) and overall survival (OS) than those with
low-risk scores, according to the results of the survival analysis (p