Abstract
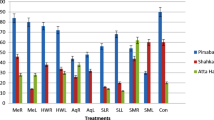
A filed experiment was done to develop management practices to reduce the use of synthetic herbicides, in the arid and semi-arid agricultural ecosystems of north east of Iran. Five treatments including I: chopped residues of canola (Brasica vulgaris), II: chopped residues of wheat (Triticum aestivum) both were separately incorporated to 25 cm depth soil, 20 days before sowing, III: shoot aqueous extract of canola, IV: shoot aqueous extract of wheat which were separately sprayed at post emergence stage and V: without any residues and spraying as control. The weed control treatments reduced the total weed cover, weed density and biomass of weed. The reduction in weed density with canola and wheat residues incorporation were up to 67.5 and 62.2% respectively, at 40 days after sowing and 65.3 and 75.6%, respectively, at 90 days after sowing, compared to control. However, post emergence spraying of shoot aqueous extract of canola and wheat, suppressed weed density up to 41.8 and 36.6% at 40 days after sowing and 54.2 and 52.7% at 90 days after sowing respectively, compared to control. Weed control treatments reduced weed cover (%), weed biomass and weeds stem length. Incorporation of canola and wheat residues in soil reduced weed cover (%) by 62.5 and 63% respectively, while spraying of shoot water extract of canola and wheat suppressed weed cover (%) by 39.6 and 40.4% respectively at 90 days after sowing. Application of canola and wheat residues increased soybean yield by 45.4 and 69.5% respectively, compared to control while post emergence application of shoot aqueous extract of canola and wheat increased soybean yield by 22 and 29.8% respectively.
Similar content being viewed by others
References
Akemo, M.C., Regnier, E.E., and Bennett, M.A., Weed suppression in spring-sown rye (Secale cereale)–Pea (Pisum sativum) cover crop mixes, Weed Tech., 2000, Vol. 14, pp. 545–549.
Ben-Hammounda, M., Kremer, R.J., Minor, H.C., and Sarwar, M., A chemical basis for differential allelo-pathic potential of Sorghum hybrids on wheat, J. Chem. Ecol., 1995, Vol. 21, pp. 775–785.
Bertholdsson, N.O., Variation in allelopathic activity in spring wheat, in Proc. Second European Allelopathy Symp. “Allelopathy: from Understanding to Application,” Pulawy, Poland, June 4, 2004, 2004.
Cheema, Z.A., Asim, M., and Khaliq, A., Sorghum allelopathy for weed control in cotton (Gossypium arboretum L.), Int. J. Agric. Biol., 2000, Vol. 2, No. 1, pp. 37–41.
Dadkhah, A., Phytotoxic effect of aqueous extract of eucalyptus, sunflower, and sugar beet on seed germination, growth and photosynthesis of Amarabthus regrof-lexus, Allelopathy. J., 2012, Vol. 29, No. 2, pp. 287–296.
Dayan, F., Romagni, J., Tellez, M., Rimando, A., and Duke, S., Managing weeds with natural products, Pesticide Outlook, 1000, Vol. 10, pp. 185–188.
De-Herralde, F., Biel, C., Save, R., Morales, M.A., Torrecillas, A., and Alarcon, J.J., Effects of water and salt stresses on growth, gas exchange and water relations in Argyranthemum coronopiflium plants, Crop Sci., 1998, Vol. 139, pp. 9–17.
Elijarrat, E. and Barcelo, D., Sample handling and analysis of allelochemical compounds in plants, Trends Anal. Chem., 2001, Vol. 20, No. 10, pp. 584–590.
Fenwick, G.R., Griffiths, N.M., and Heaney, R.K., Bitterness in Brussels sprouts (Brassica oleracea L. var. gemmifera): The role of glucosinolates and their break-down products, J. Sci. Food Agric., 1983, Vol. 34, No. 1, pp. 73–80.
Florentine, S.K., Westbrooke, M. E., Gosney, K, Ambrose, G., and O’Keefe, M., The arid lands invasive weed Nicotiana glauca R. Graham (Solanaceae): popu-lation and soil seed bank dynamics, seed germination patterns and seedling response to flood and drought, J. Arid Environ., 2006, Vol. 66, pp. 218–230.
Hegab, M.M., Khodary, S.E.A., Hammouda, O., and Ghareib, H.R., Autotoxicity of chard and its allelo-pathic potentiality on germination and some metabolic activities associated with growth of wheat seedlings, Afr. J. Biotechnol., 2008, Vol. 7, No. 7, pp. 884–892.
Jefferson, L.V. and Pennacchio, M., Allelopathic effects of foliage extracts from four Chenopodiaceae species on seed germination, J. Arid Environ., 2003, Vol. 55, pp. 275–285.
Oyun, M.B., Allelopathic potentialities of Gliricidia sepium and Acacia auriculiformis on the germination and seedling vigor of maize (Zea mays L.), Am. J. Agric. Biol. Sci., 2006, Vol. 1, No. 3, pp. 44–47.
Rice, E.L., Allelopathy, New York: Academic, 1984, 2nd ed.
Rizvi, S.J.H., Ketata, H., Bazazi, D., Roostaii, M., and Pala, M., Weed suppressing ability of bread wheat gen-otypes under greenhouse and field conditions, in Proc. Second European Allelopathy Symp. “Allelopathy: from Understanding to Application,” Pulawy, Poland, June 4, 2004, 2004, p. 25.
Singh, H.P., Daizy, R., and Kohli, R.K., Allelopathic interactions and allelochemicals: New possibilities for sustainable weed management, Crit. Rev. Plant Sci., 2003, Vol. 22, pp. 239–311.
Smith, A.E., The potential importance of allelopathy in the pasture ecosystem, a review, Adv. Agron., 1991, Vol. 45, pp. 27–37.
Wu, H., Pratley, J., Lemerle, D., and Haig, T., Labora-tory screening for allelopathic potential of wheat (Triti-cum aestivum) accessions against annual ryegrass, Aust. J. Agric. Res., 2000, Vol. 51, pp. 259–266.
Wu, H., Pratley, J., Lemerle, D., and Haig, T., Allelop-athy in wheat (Triticum aestivum), Ann. Appl. Biol., 2001, Vol. 139, pp. 1–9.
Xuan, T.D., Eiji, T., and Khan, T.D., Methods to deter-mine allelopathic potential of crop for weed control, Allelopathy J., 2004, Vol. 13, pp. 149–164.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
About this article
Cite this article
Dadkhah, A. Allelopathic potential of canola and wheat to control weeds in soybean (Glycine max). Russ. Agricult. Sci. 41, 111–114 (2015). https://doi.org/10.3103/S1068367415020081
Received:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S1068367415020081