Abstract
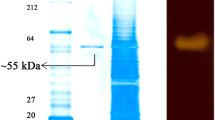
In the present study, metagenomic library of Western Ghats soil sample was constructed in a fosmid vector (pCC1FOS) and screened for biocatalytic properties. The clones showed amylolytic activity on Luria-Bertani starch agar plates and one of them was studied in detail. The enzyme exhibited stability at elevated temperature with 60°C being the optimal temperature. The enzyme retained more than 30% activity after 60 min incubation at 80°C. It also showed more than 70% activity retention in 1.5 M NaCl solution. The pH optimum of the enzyme was at pH = 5.0. The enzyme possesses good activity in the presence of chelating and strong reducing agents with activity enhancements or retention being observed at 5 mM β-mercaptoethanol, dithiothreitol and N-bromosuccinimide. However, almost complete loss of activity was observed with 5 mM EDTA, while activity enhancement was observed upon incubation with Ca2+ suggesting it to be a Ca2+-dependent α-amylase, which was further confirmed by a thin-layer chromatography (TLC). The TLC run revealed that digestion pattern was similar to commercial α-amylase. The 16S rRNA gene sequence (GenBank accession number HQ680979) BLAST showed 95% similarities with Exiguobacterium sp. AFB-11 and AFB 18, with query sequence coverage of 99%.
Similar content being viewed by others
Abbreviations
- CFU:
-
colony forming unit
- DTT:
-
dithiothreitol
- LB:
-
Luria-Bertani
- NBS:
-
N-bromosuccinimide
- TLC:
-
thin layer chromatography
References
Atichokudomchai N., Jane J. & Hazlewood G. 2006. Reaction pattern of a novel thermostable α-amylase. Carbohydr. Polym. 64: 582–588.
Beja O., Suzuki M.T., Koonin E.V., Aravind L., Hadd A. & Nguyen L.P. 2000. Construction and analysis of bacterial artificial chromosome libraries from a marine microbial assemblage. Environ. Microbiol. 2: 516–529.
Bilderback D.E. 1973. A simple method to differentiante between α and β amylase. Plant Physiol. 51: 594–595.
Chakraborty S., Khopade A., Kokare C., Mahadik K. & Chopade B. 2009. Isolation and characterization of novel α-amylase from marine Streptomyces sp. 1. J. Mol. Catal. B-Enzym. 58: 17–23.
DeSantis G., Zhu Z., Greenberg W.A., Wong K., Chaplin J., Hanson S.R., Farwell B., Nicholson L.W., Rand C.L., Weiner D.P., Robertson D.E. & Burk M.J. 2002. An enzyme library approach to biocatalysis: development of nitrilases for enantioselective production of carboxylic acid derivatives. J. Am. Chem. Soc. 124: 9024–9025.
Fernandez-Arrojo L., Guazzaroni M., Lopez-Cortes N., Beloqui A. & Ferrer M. 2010. Metagenomic era for biocatalyst identification. Curr. Opin. Biotechnol. 21: 725–733.
Henne A., Schmitz R.A., Bömeke M., Gottschalk G & Daniel R. 2000. SScreening of environmental DNA libraries for the presence of genes conferring lipolytic activity on Escherichia coli. Appl. Environ. Microbiol. 66: 3113–3116.
Hugenholz P., Goebel B.M. & Pace N.R. 1998. Impact of cultureindependent studies on the emerging phylogenetic view of bacterial diversity. J. Bacteriol. 180: 4765–4774.
Igarashi K., Hatada Y., Hagihara H., Saeki K., Takaiwa M. & Uemura T. 1998. Enzymatic properties of a novel liquefying α-amylase from an alkaliphilic Bacillus isolate and entire nucleotide and amino acid sequences. Appl. Environ. Microbiol. 64: 3282–3289.
JunGang L., KeGui Z. & WenJun H. 2010. Cloning and biochemical characterization of a novel lipolytic enzyme from the activated sludge metagenome, and its gene product. Microb. Cell Fact. 9: 83.
Kawaminami S., Ozaki K., Sumitomo N., Hayashi Y., Ito S. & Shimada I.A. 1994. Stable isotope-aided NMR study of the active site of an endoglucanase from a strain of Bacillus. J. Biol. Chem. 269: 28752–28756.
Kielak A.M., van Veen J.A. & Kowalchuk G.A. 2010. Comparative analysis of acidobacterial genomic fragments from terrestrial and aquatic metagenomic libraries, with emphasis on Acidobacteria subdivision. Appl. Environ. Microbiol. 76: 6769–6777.
Kim S.J., Lee C.M., Han B.R., Kim M.Y., Yeo Y.S. & Yoon S.H. 2008. Characterization of a gene encoding cellulase from uncultured soil bacteria. FEMS Microbiol. Lett. 282: 44–51.
Kiran K.K. & Chandra T.S. 2008. Production of surfactant and detergent-stable, halophilic, and alkalitolerant α-amylase by a moderately halophilic Bacillus sp. strain TSCVKK. Appl. Microbiol. Biotechnol. 77: 1023–1031.
Miller G.L. 1959. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Chem. 31: 426–429.
Monteiro de Souza P. & de Oliveirae e Magalhaes P.2010. Application of microbial α amylases in industry — a review. Braz. J. Microbiol. 41: 850–861.
Okolo B.N., Ezeogu L.I. & Mba C.N. 1995. Production of raw starch digesting amylase by Aspergillus niger grown on native starch sources. J. Sci. Food Agric. 69: 109–115.
Pandey A., Nigam P., Soccol C.R., Singh D., Soccol V.T. & Mohan R. 2000. Advances in microbial amylases. Biotechnol. Appl. Biochem. 31: 135–152.
Pang H., Zhang P., Duan C.J., Mo X.C., Tang J.L. & Feng J.X. 2009. Identification of cellulase genes from the metagenomes of compost soils and functional characterization of one novel endoglucanase. Curr. Microbiol. 58: 404–408.
Ranjan R., Grover A., Kapardar R.K. & Sharma R. 2005. Isolation of novel lipolytic genes from uncultured bacteria of pond water. Biochem. Biophys. Res. Commun. 335: 57–65.
Rao J. & Satyanarayana T. 2007. Purification and characterization of a hyperthermostable and high maltogenic α-amylase of an extreme thermophile Geobacillus thermoleovorans. Appl. Biochem. Biotechnol. 142: 179–193.
Rhee J.K., Ahn D.G., Kim Y.G. & Oh J.W. 2005. New thermophilic and thermostable esterase with sequence similarity to the hormone-sensitive lipase family, cloned from a metagenomic library. Appl. Environ. Microbiol. 71: 817–825.
Richardson T.H., Tan X., Frey G., Callen W., Cabell M. & Lam D. 2002. A novel, high performance enzyme for starch liquefaction discovery and optimization of a low pH, thermostable α-amylase. J. Biol. Chem. 277: 26501–26507.
Rondon M.R., August P.R., Bettermann A.D., Brady S.F., Grossman T.H. & Liles M.R. 2000. Cloning the soil metagenome: a strategy for accessing the genetic and functional diversity of uncultured microorganisms. Appl. Environ. Microbiol. 66: 2541–2547.
Sharma S., Khan F.G. & Qazi G.N. 2010. Molecular cloning and characterization of amylase from soil metagenomic library derived from North-Western Himalayas. Appl. Microbiol. Biotechnol. 86: 1821–1828.
Singh A., Chauhan N.S., Thulasiram H.V., Taneja V. & Sharma R. 2010. Identification of two flavin monooxygenases from an effluent treatment plant sludge metagenomic library. Bioresource Technol. 101: 8481–8484.
van der Maarel, M.J.E.C., van der Veen B., Uitdehaag, J.C.M., Leemhuis H. & Dijkhuizen L. 2002. Properties and applications of starch-converting enzymes of the α-amylase family. J. Biotechnol. 94: 137–155.
Venter J.C., Remington K., Heidelberg J.F., Halpern A.L., Rusch D. & Eisen J.A. 2004. Environmental genome shotgun sequencing of the Sargasso Sea. Science 304: 66–74.
Yun J., Kang S., Park S., Yoon H., Kim M.J. & Heu S. 2004. Characterization of a novel amylolytic enzyme encoded by a gene from a soil-derived metagenomic library. Appl. Environ. Microbiol. 70: 7229–7235.
Yun J. & Ryu S. 2005. Screening for novel enzymes from metagenome and SIGEX, as a way to improve it. Microb. Cell Fact. 4: 8.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vidya, J., Swaroop, S., Singh, S.K. et al. Isolation and characterization of a novel α-amylase from a metagenomic library of Western Ghats of Kerala, India. Biologia 66, 939–944 (2011). https://doi.org/10.2478/s11756-011-0126-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11756-011-0126-y