Abstract
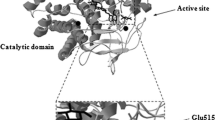
The implication of the original alanine 63 (Ala63) and the unique cysteine 306 (Cys306) residues in the thermostability of the Streptomyces sp. SK glucose isomerase (SKGI) were investigated by site-directed mutagenesis and homology modelling. The Cys306 to Ala mutation within SKGI dramatically affected its thermal stability by decreasing the half-life from 80 to 15 min at 90°C while the Ala63 to Ser replacement shifted this half-life to 65 min. The electrophoretic analysis proves that the residue Cys306 participates in oligomerization of the SKGI. Its stabilizing role is materialized by hydrogen bonds established with arginines at positions 284 and 259, as deduced from the constructed three-dimensional model. We have also shown that the presence of an Ala63 instead of Ser63 seems to be more suitable for enzyme thermostability by maintaining hydrophobic pocket that contributes to the protection of the enzyme active site.
Similar content being viewed by others
Abbreviations
- GI:
-
glucose isomerase
- SKGI:
-
Streptomyces sp. SK glucose isomerase
- WT:
-
wild-type
References
Altschul S.F., Madden T.L., Schaffer A.A., Zhang J., Zhang Z., Miller W. & Lipman D.J. 1997. Gapped BLAST and PSIBLAST: a new generation of protein database search programs. Nucleic Acids Res. 25: 3389–3402.
Asbóth B. & Naray-Szabó G. 2000. Mechanism of action of D-xylose isomerase. Curr. Protein Pept. Sci. 1: 237–254.
Belguith S.K. & Bejar S. 1998. A thermostable glucose isomerase having a relatively low optimum pH: study of activity and molecular cloning of the corresponding gene. Biotechnol. Lett. 20: 553–556.
Belguith S.K., Ellouze R. & Bejar S. 2002. Polypeptides having glucose isomerase activity and amino acids encoding the same. US Patent No: 6, 372, 476.
Bentley I.S. & Williams E.C. 1996. Starch conversion, pp. 339–357. In: Godfrey T. & West S.I., (eds), Industrial Enzymology, Stockton Press, New York.
Benson D.A., Karsch-Mizrachi I., Lipman D.J., Ostell J. & Sayers E.W. 2009. GenBank. Nucleic Acids Res. 37 (Database Issue): D26–D31.
Bhosale S.H., Rao M.B. & Deshpande V.V. 1996. Molecular and industrial aspects of glucose isomerase. Microbiol. Rev. 60: 280–300.
Borgi M.A., Belguith S.K., Ben Ali M., Mezghani M., Tranier S., Haser R. & Bejar S. 2004. Glucose isomerase of the Streptomyces sp. SK strain: purification, sequence analysis and implication of alanine 103 residue in the enzyme thermostability and acidotolerance. Biochimie 86: 561–568.
Borgi M.A., Rhimi M. & Bejar S. 2007. Involvement of alanine 103 residue in kinetic and physicochemical properties of glucose isomerases from Streptomyces species. Biotechnol. J. 2: 254–259.
Bradford M.M. 1976. A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72: 248–254.
Caniuguira A., Cabrera R., Baez M., Vasquez C.C., Babula J. & Guixéa V. 2005. Role of Cys-295 on subunit interactions and allosteric regulation of phosphofructokinase-2 from Escherichia coli. FEBS Lett. 579: 2313–2318.
Combet C., Jambon M., Deleage G. & Geourjon C. 2002. Geno3D: automatic comparative molecular modelling of protein. Bioinformatics 18: 213–214.
Chang C., Park B.C., Lee D.S. & Suh S.W. 1999. Crystal structures of thermostable xylose isomerases from Thermus caldophilus and Thermus thermophilus: possible structural determinants of thermostability. J. Mol. Biol. 288: 623–634.
Daniel R.M., Dines M. & Petach H.H. 1996. The denaturation and degradation of stable enzymes at high temperatures. Biochem. J. 317: 1–11.
Danson M.J. & Hough D.W. 1998. Structure, function and stability of enzymes from the archaea. Trends Microbiol. 6: 307–314.
Dische Z. & Borenfreund E. 1951. A new spectrophotometric method for the detection and determination of keto sugars and trioses. J. Biol. Chem. 192: 583–587.
Epting K.L., Vieille C., Zeikus G.J. & Kelly R.M. 2005. Influence of divalent cations on the structural thermostability and thermal inactivation kinetics of class II xylose isomerases FEBS J. 272: 1454–1464.
Hartley B.S., Hanlon N., Robin J.J. & Rangarajan M. 2000. Glucose isomerase: insights into protein engineering for increased thermostability. Biochim. Biophys. Acta 1543: 294–335.
Jenkins J., Janin J., Rey F., Chiadmi M., van Tilbeurgh H., Lasters I., De Maeyer M., Van Belle D., Wodak S.J., Lauwereys M., Stanssens P., Mrabet N.T., Snawaert J., Matthyssens G. & Lambeir A.M. 1992. Protein engineering of xylose (glucose) isomerase from Actinoplanes missouriensis 1. Crystallography and site-directed mutagenesis of metal binding sites. Biochemistry 31: 5449–5458.
Karimaki J., Parkkinen T., Santa H., Pastinen O., Leisola M., Rouvinen J. & Turunen O. 2004. Engineering the substrate specificity of xylose isomerase. Protein Eng. 17: 861–869.
Katz A.K., Li X., Carrell H.L., Hanson B.L., Langan P., Coates L., Schoenborn B.P., Glusker J.P. & Bunick G.J. 2006. Locating active-site hydrogen atoms in D-xylose isomerase: time-of-flight neutron diffraction. Proc. Natl. Acad. Sci. USA 103: 8342–8347.
Ladenstein R. & Antranikian G. 1998. Proteins from hyperthermophiles: stability and enzymatic catalysis close to the boiling point of water. Adv. Biochem. Eng. Biotechnol. 61: 37–85.
Laemmli U.K. 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685.
Lebbink J.H.G., Kengen S.W.M., Van der Oost J. & De Vos W.M. 1999. Glutamate dehydrogenase from hyperthermophilic bacteria and archaea: determinants of thermostability and catalysis at extremely high temperatures. J.Mol. Catal. B: Enzym. 7: 133–145.
Li Y., Yang X., Chang M., Yager J.D., van Breemen R.B. & Bolton J.L. 2005. Functional and structural comparisons of cysteine residues in the Val108 wild type and Met108 variant of human soluble catechol O-methyltransferase. Chem. Biol. Interact. 152: 151–163.
Meng M., Bagdasarian M. & Zeikus J.G. 1993. Thermal stabilization of xylose isomerase from Thermoanaerobacterium thermosulfurigenes. Bio/Technology 11: 1157–1161.
Nordberg Karlsson E., Labes A., Turner P., Fridjonsson O.H., Wennerberg C., Pozzo T., Hreggvidson G.O., Kristjansson J.K. & Schönheit P. 2008. Differences and similarities in enzymes from the neopullulanase subfamily isolated from thermophilic species. Biologia 63: 1006–1014.
Petsko G.A. 2001. Structural basis of thermostability in hyperthermophilic proteins, or ‘there’s more than one way to skin a cat. Methods Enzymol. 334: 469–478.
Quax W.J., Mrabet N.T., Luiten R.G.M., Schuurhuizen P.W., Stanssens P. & Lasters I. 1991. Enhancing the thermostability of glucose isomerase by protein engineering. Bio/Technology 9: 738–742.
Rhimi M. & Bejar S. 2006. Cloning, purification and biochemical characterization of metallic-ions independent and thermoactive L-arabinose isomerase from the Bacillus stearothermophilus US100 strain. Biochim. Biophys. Acta. 1760: 191–199.
Roussel A. & Cambillau C. 1992. Architecture et fonction des macromolecules biologiques. Biographics, Marseille, France.
Sambrook J., Fritsch E.F. & Maniatis T. 1989. Molecular Cloning: A Laboratory Manual, 2nd Ed., Cold Spring Harbor Laboratory Press, Cold Spring Harbor.
Segura L.G., García R.V., Pińera E.R., Jiménez C.M., Rosario A. & Clares M. 2005. Site-directed mutagenesis and homology modeling indicate an important role of cysteine 439 in the stability of betaine aldehyde dehydrogenase from Pseudomonas aeruginosa. Biochimie 87: 1056–1064.
Takasaki Y., Kosugi Y. & Kanbayashi A. 1969. Studies on sugarisomerizing enzyme: purification, crystallization, and some properties of glucose isomerase from Streptomyces sp. Agric. Biol. Chem. 33: 1527–1534.
Thompson J.D., Higgins D.G. & Gibson T.J. 1994. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 22: 4673–4680.
Vieille C., Hess J.M., Kelly R.M., Zeikus J.G. 1995. xylA cloning and sequencing and biochemical characterization of xylose isomerase from Thermotoga neapolitana. Appl. Environ. Microbiol. 61: 1867–1875.
Vieille C. & Zeikus G.J. 2001. Hyperthermophilic enzymes: sources, uses and molecular mechanisms for thermostability. Microbiol. Mol. Biol. Rev. 65: 1–43.
Zhu G.P., Xu C., Teng M.K., Tao L.M., Zhu X.Y., Wu C.J., Hang J., Niu L.W. & Wang Y.Z. 1999. Increasing the thermostability of D-xylose isomerase by introduction of a proline into a turn of a random coil. Protein Eng. 12: 635–638.
Xu W., Yan M., Xu L., Ding L. & Ouyang P. 2009. Engineering the activity of thermophilic xylose isomerase by site-directed mutation at subunit interfaces. Enzyme Microb. Technol. 44: 77–83.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Borgi, M.A., Rhimi, M., Aghajari, N. et al. Involvement of cysteine 306 and alanine 63 in the thermostability and oligomeric organization of glucose isomerase from Streptomyces sp. SK. Biologia 64, 845–851 (2009). https://doi.org/10.2478/s11756-009-0155-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11756-009-0155-y