Abstract
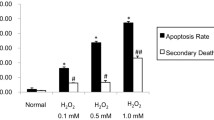
This study probed the protective effect of recombinant Lactobacillus plantarum against hydrogen peroxide (H2O2)-induced oxidative stress in human umbilical vein endothelial cells (HUVECs). We constructed a new functional L. plantarum (NC8-pSIP409-alr-angiotensin-converting enzyme inhibitory peptide (ACEIP)) with a double-gene-labeled non-resistant screen as an expression vector. A 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) colorimetric assay was carried out to determine the cell viability of HUVEC cells following pretreatment with NC8-pSIP409-alr-ACEIP. Flow cytometry (FCM) was used to determine the apoptosis rate of HUVEC cells. Cysteinyl aspartate specific proteinase (caspase)-3/8/9 activity was also assayed and western blotting was used to determine protein expression of B-cell lymphoma 2 (Bcl-2), Bcl-2-associated X protein (Bax), inducible nitric oxide synthase (iNOS), nicotinamide adenine dinucleotide phosphate oxidase 2 (gp91phox), angiotensin II (AngII), and angiotensin-converting enzyme 2 (ACE2), as well as corresponding indicators of oxidative stress, such as reactive oxygen species (ROS), mitochondrial membrane potential (MMP), malondialdehyde (MDA), and superoxide dismutase (SOD). NC8-pSIP409-alr-ACEIP attenuated H2O2-induced cell death, as determined by the MTT assay. NC8-pSIP409-alr-ACEIP reduced apoptosis of HUVEC cells by FCM. In addition, compared to the positive control, the oxidative stress index of the H2O2-induced HUVEC (Hy-HUVEC), which was pretreated by NC8-pSIP409-alr-ACEIP, iNOS, gp91phox, MDA, and ROS, was decreased obviously; SOD expression level was increased; caspase-3 or -9 was decreased, but caspase-8 did not change; Bcl-2/Bax ratio was increased; permeability changes of mitochondria were inhibited; and loss of transmembrane potential was prevented. Expression of the hypertension-related protein (AngII protein) in HUVEC cells protected by NC8-pSIP409-alr-ACEIP decreased and expression of ACE2 protein increased. These plantarum results suggested that NC8-pSIP409-alr-ACEIP protects against H2O2-induced injury in HUVEC cells. The mechanism for this effect is related to enhancement of antioxidant capacity and apoptosis.
概要
目的
本文研究新功能重组植物乳杆菌NC8-pSIP409-alr-ACEIP保护血管内皮细胞损伤的初步作用机制。
创新点
成功构建非抗性筛选标记的新功能植物乳杆菌NC8-pSIP409-alr-ACEIP, 其具有安全, 方便, 无毒副作用, 且以食疗方式达到降血压的优点。研究NC8-pSIP409-alr-ACEIP对相关蛋白血管紧张素II(AngII), 血管紧张素转化酶2(ACE2), B淋巴细胞瘤-2(Bcl-2), BCL2关联X蛋白(Bax)与氧化应激相关因子的变化情况, 为阐明NC8-pSIP409-alr-ACEIP保护血管内皮细胞损伤的作用机制提供了基础的理论依据。
方法
首次构建以双基因标记的非抗性筛选锚定为表达载体的新功能型植物乳杆菌(NC8-pSIP409-alr-ACEIP), 使用不同浓度的过氧化氢(H2O2)诱导人脐静脉内皮细胞(HUVEC)细胞, 构建氧化应激损伤细胞模型(Hy-HUVEC), 通过MTT, 流式细胞术等方法检测Hy-HUVEC的凋亡, 通过蛋白质印迹法(western blot)检测AngII, ACE2, 诱导型一氧化氮合酶(iNOS), 尼克酰胺腺嘌呤二核苷酸磷酸氧化酶(gp91phox), Bcl-2和Bax的蛋白表达情况, 同时检测氧化应激相应指标活性氧(ROS), 线粒体膜电位(MMP), 丙二醛(MDA)和超氧化物歧化酶(SOD)的变化情况。
结论
NC8-pSIP409-alr-ACEIP对H2O2诱导的HUVEC细胞氧化应激损伤具有保护作用, 其作用机制与增强抗氧化能力和降低由线粒体凋亡通路引发的细胞凋亡有关。
Article PDF
Similar content being viewed by others
References
Aldabbous L, Abdul-Salam V, McKinnon T, et al., 2016. Neutrophil extracellular traps promote angiogenesis. Arterioscler Thromb Vasc Biol, 36(10):2078–2087. https://doi.org/10.1161/ATVBAHA.116.307634
Bali A, Singh N, Jaggi AS, 2014. Renin-angiotensin system in pain: existing in a double life? J Renin Angiotensin Aldosterone Syst, 15(4):329–340. https://doi.org/10.1177/1470320313503694
Bialas AJ, Sitarek P, Milkowska-Dymanowska J, et al., 2016. The role of mitochondria and oxidative/antioxidative imbalance in pathobiology of chronic obstructive pulmonary disease. Oxid Med Cell Longev, 2016:7808576. https://doi.org/10.1155/2016/7808576
Bian YF, Yang HY, Yang ZM, et al., 2011. Amlodipine treatment prevents angiotensin II-induced human umbilical vein endothelial cell apoptosis. Arch Med Res, 42(1):22–27. https://doi.org/10.1016/j.arcmed.2011.01.012
Camini FC, da Silva Caetano CC, Almeida LT, et al., 2017. Oxidative stress in Mayaro virus infection. Virus Res, 236:1–8. https://doi.org/10.1016/j.virusres.2017.04.017
Chen JW, Mehta JL, Haider N, et al., 2004. Role of caspases in Ox-LDL-induced apoptotic cascade in human coronary artery endothelial cells. Circ Res, 94(3):370–376. https://doi.org/10.1161/01.RES.0000113782.07824.BE
Collister JP, Taylor-Smith H, Drebes D, et al., 2016. Angiotensin II-induced hypertension is attenuated by overexpressing copper/zinc superoxide dismutase in the brain organum vasculosum of the lamina terminalis. Oxid Med Cell Longev, 2016:3959087. https://doi.org/10.1155/2016/3959087
Deepak V, Ramachandran S, Balahmar RM, et al., 2016. In vitro evaluation of anticancer properties of exopolysaccharides from Lactobacillus acidophilus in colon cancer cell lines. In Vitro Cell Dev Biol Anim, 52(2): 163–173. https://doi.org/10.1007/s11626-015-9970-3
Du J, Leng JY, Zhang L, et al., 2016. Angiotensin II-induced apoptosis of human umbilical vein endothelial cells was inhibited by blueberry anthocyanin through Bax- and Caspase 3-dependent pathways. Med Sci Monit, 22:3223–3228. https://doi.org/10.12659/MSM.896916
Ettinger G, MacDonald K, Reid G, et al., 2014. The influence of the human microbiome and probiotics on cardiovascular health. Gut Microbes, 5(6):719–728. https://doi.org/10.4161/19490976.2014.983775
Fraga-Silva RA, Costa-Fraga FP, Murça TM, et al., 2013. Angiotensin-converting enzyme 2 activation improves endothelial function. Hypertension, 61(6): 1233–1238. https://doi.org/10.1161/HYPERTENSIONAHA.111.00627
Gordon JI, 2012. Honor thy gut symbionts redux. Science, 336(6086):1251–1253. https://doi.org/10.1126/science.1224686
Graham D, Hamilton C, Beattie E, et al., 2004. Comparison of the effects of omapatrilat and irbesartan/hydrochlorothiazide on endothelial function and cardiac hypertrophy in the stroke-prone spontaneously hypertensive rat: sex differences. J Hypertens, 22(2):329–337. https://doi.org/10.1097/00004872-200402000-00017
Huang H, Shan J, Pan XH, et al., 2006. Carvedilol protected diabetic rat hearts via reducing oxidative stress. Zhejiang Univ-Sci B (Biomed & Biotechnol), 7(9):725–731. https://doi.org/10.1631/jzus.2006.B0725
Kim S, Goel R, Kumar A, et al., 2018. Imbalance of gut microbiome and intestinal epithelial barrier dysfunction in patients with high blood pressure. Clin Sci, 132(6):701–718. https://doi.org/10.1042/CS20180087
Korsager Larsen M, Matchkov VV, 2016. Hypertension and physical exercise: the role of oxidative stress. Medicina, 52(1):19–27. https://doi.org/10.1016/j.medici.2016.01.005
Laurent S, 2017. Antihypertensive drugs. Pharmacol Res, 124:116–125. https://doi.org/10.1016/j.phrs.2017.07.026
Lee EJ, Ko HM, Jeong YH, et al., 2015. β-Lapachone suppresses neuroinflammation by modulating the expression of cytokines and matrix metalloproteinases in activated microglia. J Neuroinflammation, 12:133. https://doi.org/10.1186/s12974-015-0355-z
Liu CT, Chu FJ, Chou C, et al., 2011. Antiproliferative and anticytotoxic effects of cell fractions and exopolysaccharides from Lactobacillus casei 01. Mutat Res Genet Toxicol Environ Mutagen, 721(2): 157–162. https://doi.org/10.1016/j.mrgentox.2011.01.005
Liu J, Zhang FF, Li L, et al., 2013. ClC-3 deficiency prevents apoptosis induced by angiotensin II in endothelial progenitor cells via inhibition of NADPH oxidase. Apoptosis, 18(10):1262–1273. https://doi.org/10.1007/s10495-013-0881-z
Loperena R, Harrison DG, 2017. Oxidative stress and hypertensive diseases. Med Clin North Am, 101(1):169–193. https://doi.org/10.1016/j.mcna.2016.08.004
Lugo-Baruqui A, Muñoz-Valle JF, Arévalo-Gallegos S, et al., 2010. Role of angiotensin II in liver fibrosis-induced portal hypertension and therapeutic implications. Hepatol Res, 40(1):95–104. https://doi.org/10.1111/j.1872-034X.2009.00581.x
Luo CF, Yuan DD, Zhao WC, et al., 2015. Sevoflurane ameliorates intestinal ischemia-reperfusion-induced lung injury by inhibiting the synergistic action between mast cell activation and oxidative stress. Mol Med Rep, 12(1):1082–1090. https://doi.org/10.3892/mmr.2015.3527
Maghraoui S, Clichici S, Ayadi A, et al., 2014. Oxidative stress in blood and testicle of rat following intraperitoneal administration of aluminum and indium. Acta Physiol Hung, 101(1):47–58. https://doi.org/10.1556/aphysiol.100.2013.021
Mendell JT, Olson EN, 2012. MicroRNAs in stress signaling and human disease. Cell, 148(6):1172–1187. https://doi.org/10.1016/j.cell.2012.02.005
Ott M, Gogvadze V, Orrenius S, et al., 2007. Mitochondria, oxidative stress and cell death. Apoptosis, 12(5):913–922. https://doi.org/10.1007/s10495-007-0756-2
Pavli F, Tassou C, Nychas GJE, et al., 2018. Probiotic incorporation in edible films and coatings: bioactive solution for functional foods. Int J Mol Sci, 19(1):150. https://doi.org/10.3390/ijms19010150
Präbst K, Engelhardt H, Ringgeler S, et al., 2017. Basic colorimetric proliferation assays: MTT, WST, and resazurin. Methods Mol Biol, 1601:1–17. https://doi.org/10.1007/978-1-4939-6960-9_1
Santos CMA, Pires MCV, Leão TL, et al., 2016. Selection of Lactobacillus strains as potential probiotics for vaginitis treatment. Microbiology, 162(7): 1195–1207. https://doi.org/10.1099/mic0.000302
Sharafedtinov KK, Plotnikova OA, Alexeeva RI, et al., 2013. Hypocaloric diet supplemented with probiotic cheese improves body mass index and blood pressure indices of obese hypertensive patients—a randomized double-blind placebo-controlled pilot study. Nutr J, 12:138. https://doi.org/10.1186/1475-2891-12-138
Shreiber DI, Enever PAJ, Tranquillo RT, 2001. Effects of PDGF-BB on rat dermal fibroblast behavior in mechanically stressed and unstressed collagen and fibrin gels. Exp Cell Res, 266(1):155–166. https://doi.org/10.1006/excr.2001.5208
Small HY, Migliarino S, Czesnikiewicz-Guzik M, et al., 2018. Hypertension: focus on autoimmunity and oxidative stress. Free Radic Biol Med, 125:104–115. https://doi.org/10.1016/j.freeradbiomed.2018.05.085
Upadrasta A, Madempudi RS, 2016. Probiotics and blood pressure: current insights. Integr Blood Press Control, 9:33–42. https://doi.org/10.2147/IBPC.S73246
van Thiel BS, van der Pluijm I, te Riet L, et al., 2015. The reninangiotensin system and its involvement in vascular disease. Eur J Pharmacol, 763:3–14. https://doi.org/10.1016/j.ejphar.2015.03.090
Vaziri ND, Wang XQ, Oveisi F, et al., 2000. Induction of oxidative stress by glutathione depletion causes severe hypertension in normal rats. Hypertension, 36(1): 142–146. https://doi.org/10.1161/01.HYP.36.1.142
Wali JA, Rondas D, McKenzie MD, et al., 2014. The proapoptotic BH3-only proteins Bim and Puma are downstream of endoplasmic reticulum and mitochondrial oxidative stress in pancreatic islets in response to glucotoxicity. Cell Death Dis, 5(3): e1124. https://doi.org/10.1038/cddis.2014.88
Wan L, Nie GJ, Zhang J, et al., 2011. β-Amyloid peptide increases levels of iron content and oxidative stress in human cell and Caenorhabditis elegans models of Alzheimer disease. Free Radic Biol Med, 50(1):122–129. https://doi.org/10.1016/j.freeradbiomed.2010.10.707
Wang JY, Sun PP, Bao YM, et al., 2012. Vitamin E renders protection to PC12 cells against oxidative damage and apoptosis induced by single-walled carbon nanotubes. Toxicol in Vitro, 26(1):32–41. https://doi.org/10.1016/j.tiv.2011.10.004
Wang ZX, Wang Y, Chen YF, et al., 2016. The IL-24 gene protects human umbilical vein endothelial cells against H2O2-induced injury and may be useful as a treatment for cardiovascular disease. Int J Mol Med, 37(3):581–592. https://doi.org/10.3892/ijmm.2016.2466
Wasilewska E, Złotkowska D, Pijagin ME, 2013. The role of intestinal microflora and probiotic bacteria in prophylactic and development of colorectal cancer. Postepy Hig Med Dosw, 67:837–847. https://doi.org/10.5604/17322693.1061847
Wu Q, Liu XX, Lu DY, et al., 2018. Protective effect of polygonum orientale flower extract on H2O2-induced oxidative damage of HUVEC cells. China J Chin Mat Med, 43(5):1008–1013 (in Chinese). https://doi.org/10.19540/j.cnki.cjcmm.2018.0033
Wu YC, Wang YM, Nabi X, 2019. Protective effect of Ziziphora clinopodioides flavonoids against H2O2-induced oxidative stress in HUVEC cells. Biomed Pharmacother, 117:109156. https://doi.org/10.1016/j.biopha.2019.109156
Yang GL, Jiang YL, Yang WT, et al., 2015. Effective treatment of hypertension by recombinant Lactobacillus plantarum expressing angiotensin converting enzyme inhibitory peptide. Microb Cell Fact, 14:202. https://doi.org/10.1186/s12934-015-0394-2
Yang T, Santisteban MM, Rodriguez V, et al., 2015. Gut dysbiosis is linked to hypertension. Hypertension, 65(6): 1331–1340. https://doi.org/10.1161/HYPERTENSIONAHA.115.05315
Young CN, Davisson RL, 2015. Angiotensin-II, the brain, and hypertension. Hypertension, 66(5):920–926. https://doi.org/10.1161/HYPERTENSIONAHA.115.03624
Zeng MY, Miralda I, Armstrong CL, et al., 2019. The roles of NADPH oxidase in modulating neutrophil effector responses. Mol Oral Microbiol, 34(2):27–38. https://doi.org/10.1111/omi.1225
Zhang SS, Qin FX, Yang LY, et al., 2016. Nucleophosmin mutations induce chemosensitivity in THP-1 leukemia cells by suppressing NF-κB activity and regulating Bax/Bcl-2 expression. Cancer, 7(15):2270–2279. https://doi.org/10.7150/jca.16010
Zhang Y, Zou CS, Yang SW, et al., 2016. p120 catenin attenuates the angiotensin II-induced apoptosis of human umbilical vein endothelial cells by suppressing the mitochondrial pathway. Int Mol Med, 37(3):623–630. https://doi.org/10.3892/ijmm.2016.2476
Zhou B, Bentham J, di Cesare M, et al., 2017. Worldwide trends in blood pressure from 1975 to 2015: a pooled analysis of 1479 population-based measurement studies with 19·1 million participants. Lancet, 389(10064):37–55. https://doi.org/10.1016/S0140-6736(16)31919-5
Zittermann A, Pilz S, 2019. Vitamin D and cardiovascular disease: an update. Anticancer Res, 39(9):4627–4635. https://doi.org/10.21873/anticanres.13643
Acknowledgments
This work is supported the National Key Research and Development Program of China (Nos. 2017YFD0501000 and 2017YFD0501200), the National Natural Science Foundation of China (Nos. 31672528, 31941018, and 32072888), the Science and Technology Project of Jilin Provincial Department of Education (No. JJKH20190942KJ), and the Science and Technology Development Program of Jilin Province (Nos. 20180201040NY and 20190301042NY), China.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Author contributions
Guan WANG and Mingyue HAO performed the experimental research and data analysis, and wrote and edited the manuscript. Mingyue HAO performed the establishment of cell models. Qiong LIU, Yanlong JIANG, and Haibin HUANG contributed to the study design, data analysis, and writing and editing of the manuscript. Guilian YANG and Chunfeng WANG performed experimental design. All authors have read and approved the final manuscript and, therefore, have full access to all the data in the study and take responsibility for the integrity and security of the data.
Compliance with ethics guidelines
Guan WANG, Mingyue HAO, Qiong LIU, Yanlong JIANG, Haibin HUANG, Guilian YANG, and Chunfeng WANG declare that they have no conflict of interest.
This article does not contain any studies with human or animal subjects performed by any of the authors.
Rights and permissions
About this article
Cite this article
Wang, G., Hao, M., Liu, Q. et al. Protective effect of recombinant Lactobacillus plantarum against H2O2-induced oxidative stress in HUVEC cells. J. Zhejiang Univ. Sci. B 22, 348–365 (2021). https://doi.org/10.1631/jzus.B2000441
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B2000441