Abstract
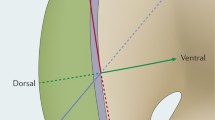
Understanding limb development not only gives insights into the outgrowth and differentiation of the limb, but also has clinical relevance. Limb development begins with two paired limb buds (forelimb and hindlimb buds), which are initially undifferentiated mesenchymal cells tipped with a thickening of the ectoderm, termed the apical ectodermal ridge (AER). As a transitional embryonic structure, the AER undergoes four stages and contributes to multiple axes of limb development through the coordination of signalling centres, feedback loops, and other cell activities by secretory signalling and the activation of gene expression. Within the scope of proximodistal patterning, it is understood that while fibroblast growth factors (FGFs) function sequentially over time as primary components of the AER signalling process, there is still no consensus on models that would explain proximodistal patterning itself. In anteroposterior patterning, the AER has a dual-direction regulation by which it promotes the sonic hedgehog (Shh) gene expression in the zone of polarizing activity (ZPA) for proliferation, and inhibits Shh expression in the anterior mesenchyme. In dorsoventral patterning, the AER activates Engrailed-1 (En1) expression, and thus represses Wnt family member 7a (Wnt7a) expression in the ventral ectoderm by the expression of Fgfs, Sp6/8, and bone morphogenetic protein (Bmp) genes. The AER also plays a vital role in shaping the individual digits, since levels of Fgf4/8 and Bmps expressed in the AER affect digit patterning by controlling apoptosis. In summary, the knowledge of crosstalk within AER among the three main axes is essential to understand limb growth and pattern formation, as the development of its areas proceeds simultaneously.
摘要
理解胚胎肢体发育有助于更加全面地了解肢体的 发生和分化,同时对于相关基因疗法和肢体再生 具有临床指导意义。本文综述了顶端外胚层嵴 (AER)参与胚胎肢体发育调控的研究进展及机 理,重点总结了AER 对肢体发育三个轴向的立 体调控,包括基因表达调控、信号通路及相关调 控中心,并进一步讨论了各信号中心之间的交互 作用及现有研究待解决问题,从而加深对肢体发 育异常、肢端异常等疾病的理解。
Similar content being viewed by others
References
Barrow JR, Thomas KR, Boussadia-Zahui O, et al., 2003. Ectodermal Wnt3/β-catenin signaling is required for the establishment and maintenance of the apical ectodermal ridge. Genes Dev, 17(3):394–409. https://doi.org/10.1101/gad.1044903
Bouldin CM, Gritli-Linde A, Ahn S, et al., 2010. Shh pathway activation is present and required within the vertebrate limb bud apical ectodermal ridge for normal autopod patterning. Proc Natl Acad Sci USA, 107(12):5489–5494. https://doi.org/10.1073/pnas.0912818107
Casanova JC, Uribe V, Badia-Careaga C, et al., 2011. Apical ectodermal ridge morphogenesis in limb development is controlled by Arid3b-mediated regulation of cell movements. Development, 138(6):1195–1205. https://doi.org/10.1242/dev.057570
Choi KS, Lee C, Maatouk DM, et al., 2012. Bmp2, Bmp4 and Bmp7 are co-required in the mouse AER for normal digit patterning but not limb outgrowth. PLoS ONE, 7(5):e37826. https://doi.org/10.1371/journal.pone.0037826
Cooper KL, Hu JKH, Ten Berge D, et al., 2011. Initiation of proximal-distal patterning in the vertebrate limb by signals and growth. Science, 332(6033):1083–1086. https://doi.org/10.1126/science.1199499
Danopoulos S, Parsa S, al Alam D, et al., 2013. Transient inhibition of FGFR2b-ligands signaling leads to irreversible loss of cellular β-catenin organization and signaling in AER during mouse limb development. PLoS ONE, 8(10):e76248. https://doi.org/10.1371/journal.pone.0076248
Delgado I, Torres M, 2017. Coordination of limb development by crosstalk among axial patterning pathways. Dev Biol, 429(2):382–386. https://doi.org/10.1016/j.ydbio.2017.03.006
Duboc V, Logan MPO, 2009. Building limb morphology through integration of signalling modules. Curr Opin Genet Dev, 19(5):497–503. https://doi.org/10.1016/j.gde.2009.07.002
Dudley AT, Ros MA, Tabin CJ, 2002. A re-examination of proximodistal patterning during vertebrate limb development. Nature, 418(6897):539–544. https://doi.org/10.1038/nature00945
Fernandez-Teran M, Ros MA, 2008. The apical ectodermal ridge: morphological aspects and signaling pathways. Int J Dev Biol, 52(7):857–871. https://doi.org/10.1387/ijdb.072416mf
Gros J, Tabin CJ, 2014. Vertebrate limb bud formation is initiated by localized epithelial-to-mesenchymal transition. Science, 343(6176):1253–1256. https://doi.org/10.1126/science.1248228
Hajihosseini MK, Heath JK, 2002. Expression patterns of fibroblast growth factors-18 and -20 in mouse embryos is suggestive of novel roles in calvarial and limb development. Mech Dev, 113(1):79–83. https://doi.org/10.1016/S0925-4773(01)00656-6
Haro E, Delgado I, Junco M, et al., 2014. Sp6 and Sp8 transcription factors control AER formation and dorsalventral patterning in limb development. PLoS Genet, 10(8):e1004468. https://doi.org/10.1371/journal.pgen.1004468
Irvine KD, Rauskolb C, 2001. Boundaries in development: formation and function. Annu Rev Cell Dev Biol, 17:189–214. https://doi.org/10.1146/annurev.cellbio.17.L189
Itoh N, Ohta H, 2014. Fgf10: a paracrine-signaling molecule in development, disease, and regenerative medicine. Curr Mol Med, 14(4):504–509. https://doi.org/10.2174/1566524014666140414204829
Jin LB, Wu J, Bellusci S, et al., 2019. Fibroblast growth factor 10 and vertebrate limb development. Front Genet, 9:705. https://doi.org/10.3389/fgene.2018.00705
Kawakami Y, Capdevila J, Büscher D, et al., 2001. WNT signals control FGF-dependent limb initiation and AER induction in the chick embryo. Cell, 104(6):891–900. https://doi.org/10.1016/S0092-8674(01)00285-9
Laufer E, Nelson CE, Johnson RL, et al., 1994. Sonic hedgehog and Fgf-4 act through a signaling cascade and feedback loop to integrate growth and patterning of the developing limb bud. Cell, 79(6):993–1003. https://doi.org/10.1016/0092-8674(94)90030-2
Lettice LA, Williamson I, Wiltshire JH, et al., 2012. Opposing functions of the ETS factor family define Shh spatial expression in limb buds and underlie polydactyly. Dev Cell, 22(2):459–467. https://doi.org/10.1016/j.devcel.2011.12.010
Lewandoski M, Sun X, Martin GR, 2000. Fgf8 signalling from the AER is essential for normal limb development. Nat Genet, 26(4):460–463. https://doi.org/10.1038/82609
Logan C, Hornbruch A, Campbell I, et al., 1997. The role of Engrailed in establishing the dorsoventral axis of the chick limb. Development, 124(12):2317–2324.
Loomis CA, Harris E, Michaud J, et al., 1996. The mouse Engrailed-1 gene and ventral limb patterning. Nature, 382(6589):360–363. https://doi.org/10.1038/382360a0
Mallick A, 2013. The function of apical ectodermal ridge in the formation of limb. Bangladesh J Sci Res, 26(1–2):95–99. https://doi.org/10.3329/bjsr.v26i1-2.20237
Mariani FV, Ahn CP, Martin GR, 2008. Genetic evidence that FGFs have an instructive role in limb proximal-distal patterning. Nature, 453(7193):401–405. https://doi.org/10.1038/nature06876
Martin GR, 1998. The roles of FGFs in the early development of vertebrate limbs. Genes Dev, 12(11):1571–1586. https://doi.org/10.1101/gad.12.11.1571
Mercader N, Leonardo E, Azpiazu N, et al., 1999. Conserved regulation of proximodistal limb axis development by Meis1/Hth. Nature, 402(6760):425–429. https://doi.org/10.1038/46580
Mercader N, Leonardo E, Piedra ME, et al., 2000. Opposing RA and FGF signals control proximodistal vertebrate limb development through regulation of Meis genes. Development, 127(18):3961–3970.
Mercader N, Selleri L, Criado LM, et al., 2009. Ectopic Meis1 expression in the mouse limb bud alters P-D patterning in a Pbx1-independent manner. Int J Dev Biol, 53(8–10):1483–1494. https://doi.org/10.1387/ijdb.072430nm
Moon AM, Capecchi MR, 2000. Fgf8 is required for outgrowth and patterning of the limbs. Nat Genet, 26(4):455–459. https://doi.org/10.1038/82601
Moon AM, Boulet AM, Capecchi MR, 2000. Normal limb development in conditional mutants of Fgf4. Development, 127(5):989–996.
Nelson CE, Morgan BA, Burke AC, et al., 1996. Analysis of Hox gene expression in the chick limb bud. Development, 122(5):1449–1466.
Niederreither K, Fraulob V, Garnier JM, et al., 2002. Differential expression of retinoic acid-synthesizing (RALDH) enzymes during fetal development and organ differentiation in the mouse. Mech Dev, 110(1–2):165–171. https://doi.org/10.1016/S0925-4773(01)00561-5
Nishimoto S, Wilde SM, Wood S, et al., 2015. RA acts in a coherent feed-forward mechanism with Tbx5 to control limb bud induction and initiation. Cell Rep, 12(5):879–891. https://doi.org/10.1016/j.celrep.2015.06.068
Niswander L, Martin GR, 1992. Fgf-4 expression during gastrulation, myogenesis, limb and tooth development in the mouse. Development, 114(3):755–768.
Niswander L, Tickle C, Vogel A, et al., 1993. FGF-4 replaces the apical ectodermal ridge and directs outgrowth and patterning of the limb. Cell, 75(3):579–587. https://doi.org/10.1016/0092-8674(93)90391-3
Niswander L, Jeffrey S, Martin GR, et al., 1994. A positive feedback loop coordinates growth and patterning in the vertebrate limb. Nature, 371(6498):609–612. https://doi.org/10.1038/371609a0
Pajni-Underwood S, Wilson CP, Elder C, et al., 2007. BMP signals control limb bud interdigital programmed cell death by regulating FGF signaling. Development, 134(12):2359–2368. https://doi.org/10.1242/dev.001677
Pizette S, Abate-Shen C, Niswander L, 2001. BMP controls proximodistal outgrowth, via induction of the apical ectodermal ridge, and dorsoventral patterning in the vertebrate limb. Development, 128(22):4463–4474.
Pownall ME, Isaacs HV, 2010. FGF Signalling in Vertebrate Development. Morgan and Claypool Life Sciences, San Rafael, USA. https://doi.org/10.4199/C00011ED1V01Y201004DEB002
Rodriguez-Esteban C, Tsukui T, Yonei S, et al., 1999. The T-box genes Tbx4 and Tbx5 regulate limb outgrowth and identity. Nature, 398(6730):814–818. https://doi.org/10.1038/19769
Rodriguez-Leon J, Tomas AR, Johnson A, et al., 2013. Recent advances in the study of limb development: the emergence and function of the apical ectodermal ridge. In: Reyes D, Casales A (Eds.), Embryo Development: Stages, Mechanisms and Clinical Outcomes. Nova Science Publishers Inc., New York, p.77–112.
Roselló-Díez A, Arques CG, Delgado I, et al., 2014. Diffusible signals and epigenetic timing cooperate in late proximodistal limb patterning. Development, 141(7):1534–1543. https://doi.org/10.1242/dev.106831
Saunders JW Jr, 1948. The proximo-distal sequence of origin of the parts of the chick wing and the role of the ectoderm. J Exp Zool, 108(3):363–403. https://doi.org/10.1002/jez.1401080304
Scherz PJ, Harfe BD, McMahon AP, et al., 2004. The limb bud Shh-Fgf feedback loop is terminated by expansion of former ZPA cells. Science, 305(5682):396–399. https://doi.org/10.1126/science.1096966
Sekine K, Ohuchi H, Fujiwara M, et al., 1999. Fgf10 is essential for limb and lung formation. Nat Genet, 21(1):138–141. https://doi.org/10.1038/5096
Sheeba CJ, Logan MPO, 2017. The roles of T-box genes in vertebrate limb development. Curr Top Dev Biol, 122:355–381. https://doi.org/10.1016/bs.ctdb.2016.08.009
Summerbell D, Lewis JH, Wolpert L, 1973. Positional information in chick limb morphogenesis. Nature, 244(5417):492–496. https://doi.org/10.1038/244492a0
Sun X, Lewandoski M, Meyers EN, et al., 2000. Conditional inactivation of Fgf4 reveals complexity of signalling during limb bud development. Nat Genet, 25(1):83–86. https://doi.org/10.1038/75644
Sun X, Mariani FV, Martin GR, 2002. Functions of FGF signalling from the apical ectodermal ridge in limb development. Nature, 418(6897):501–508. https://doi.org/10.1038/nature00902
Tabin C, Wolpert L, 2007. Rethinking the proximodistal axis of the vertebrate limb in the molecular era. Genes Dev, 21(12):1433–1442. https://doi.org/10.1101/gad.1547407
Tickle C, 2003. Patterning systems—from one end of the limb to the other. Dev Cell, 4(4):449–458. https://doi.org/10.1016/S1534-5807(03)00095-9
Tickle C, Wolpert L, 2002. The progress zone—alive or dead?. Nat Cell Biol, 4(9):E216–E217. https://doi.org/10.1038/ncb0902-e216
Towers M, Tickle C, 2009. Growing models of vertebrate limb development. Development, 136(2):179–190. https://doi.org/10.1242/dev.024158
Wolpert L, 2002. Limb patterning: reports of model’s death exaggerated. Curr Biol, 12(18):R628–R630. https://doi.org/10.1016/S0960-9822(02)01137-5
Yashiro K, Zhao XL, Uehara M, et al., 2004. Regulation of retinoic acid distribution is required for proximodistal patterning and outgrowth of the developing mouse limb. Dev Cell, 6(3):411–422. https://doi.org/10.1016/S1534-5807(04)00062-0
Zeller R, López-Ríos J, Zuniga A, 2009. Vertebrate limb bud development: moving towards integrative analysis of organogenesis. Nat Rev Genet, 10(12):845–858. https://doi.org/10.1038/nrg2681
Zhang Z, Verheyden JM, Hassell JA, et al., 2009. FGF-regulated Etv genes are essential for repressing Shh expression in mouse limb buds. Dev Cell, 16(4):607–613. https://doi.org/10.1016/j.devcel.2009.02.008
Author information
Authors and Affiliations
Contributions
Guo-hao LIN performed the literature research, wrote and edited the manuscript. Lan ZHANG provided expert comments, edited and revised the manuscript. Both authors have read and approved the final manuscript.
Corresponding author
Additional information
Compliance with ethics guidelines
Guo-hao LIN and Lan ZHANG declare that they have no conflict of interest.
This article does not contain any studies with human or animal subjects performed by either of the authors.
Project supported by the Key Research and Development Project of Shandong Province (No. 2017G006043), China
Rights and permissions
About this article
Cite this article
Lin, Gh., Zhang, L. Apical ectodermal ridge regulates three principal axes of the developing limb. J. Zhejiang Univ. Sci. B 21, 757–766 (2020). https://doi.org/10.1631/jzus.B2000285
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B2000285
Key words
- Apical ectodermal ridge (AER)
- Limb development
- Fibroblast growth factor (FGF)
- Zone of polarizing activity (ZPA)