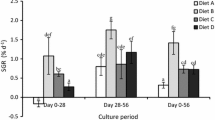
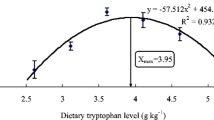
Abstract
Ecdysis is a common phenomenon that happens throughout the life phase of the giant freshwater prawn Macrobrachium rosenbergii. It is vital to better understand the correlation between cannibalism and biochemical compound that exists during the moulting process. The objective of the present study was to determine the amino acid profile released by M. rosenbergii during the ecdysis process that promotes cannibalism. To accomplish this, changes in amino acid levels (total amino acid (TAA) and free amino acid (FAA)) of tissue muscle, exoskeleton, and sample water of culture medium from the moulting (E-stage) and non-moulting (C-stage) prawns were analysed using high-performance liquid chromatography (HPLC). Comparison study revealed that among the TAA compounds, proline and sarcosine of tissues from moulting prawn were found at the highest levels. The level of FAA from water that contains moulting prawns (E-stage) was dominated by tryptophan and proline. Significant values obtained in the present study suggested that these amino acid compounds act as a chemical cue to promote cannibalism in M. rosenbergii during ecdysis. The knowledge of compositions and compounds that were released during the moulting process should be helpful for better understanding of the mechanism and chemical cues that play roles on triggering cannibalism, and also for future dietary manipulation to improve feeding efficiencies and feeding management, which indirectly impacts productivity and profitability.
摘要
目的
确定罗氏沼虾在蜕皮过程(该过程引起同类残杀) 中释放的氨基酸组分, 并明确其中差异的氨基酸 成分.
创新点
了解罗氏沼虾蜕皮过程中生化组分的变化对于理 解同类残杀的重要意义。目前, 还没有关于同类 残杀和蜕皮过程与生化组分之间关系的研究报 道。本研究以此为切入点, 采用统计学方法探究 二者之间的关联, 并鉴定其中差异的氨基酸成 分.
方法
在人工饲养条件下, 测定了蜕皮阶段和非蜕皮阶 段的罗氏沼虾肌肉、外骨骼和养殖水体中的总氨 基酸及自由氨基酸组分, 采用双因素方差分析法 比较分析了两个阶段氨基酸组分的差异, 同时鉴 定了差异的氨基酸成分。, 在蜕皮阶段, 组织中的总氨基酸以脯氨酸和肌氨 酸的含量最高, 而水体中的自由氨基酸含量以色 氨酸和脯氨酸为主。研究结果表明, 这些氨基酸 组分在罗氏沼虾蜕皮过程中释放了促进同类残 杀的化学信号.
Similar content being viewed by others
References
Abe H, Okuma E, Amano H, et al., 1999. Effects of seawater acclimation on the levels of free d- and l-alanine and other osmolytes in the Japanese mitten crab Eriocheir japonicus. Fisheries Sci, 65(6):949–954. https://doi.org/10.2331/fishsci.65.949
Augusto A, Greene LJ, Laure HJ, et al., 2007a. Adaptive shifts in osmoregulatory strategy and the invasion of freshwater by brachyuran crabs: evidence from Dilocarcinus pagei (Trichodactylidae). J Exp Zool Part A: Ecol Genet Physiol, 307A(12):688–698. https://doi.org/10.1002/jez.a.422
Augusto A, Greene LJ, Laure HJ, et al., 2007b. The ontogeny of isosmotic intracellular regulation in the diadromous, freshwater palaemonid shrimps, Macrobrachium amazonicum and M. olfersi (Decapoda). J Crustacean Biol, 27(4):626–634. https://doi.org/10.1651/S-2796.1
Augusto A, Pinheiro AS, Greene LJ, et al., 2009. Evolutionary transition to freshwater by ancestral marine palaemonids: evidence from osmoregulation in a tide pool shrimp. Aquat Biol, 7(1–2):113–122. https://doi.org/10.3354/ab00183
Azra MN, Chen JC, Hsu TH, et al., 2019. Growth, molting duration and carapace hardness of blue swimming crab, Portunus pelagicus, instars at different water temperatures. Aquacult Rep, 15:100226. https://doi.org/10.1016/j.aqrep.2019.100226
Barki A, Jones C, Karplus I, 2011. Chemical communication and aquaculture of decapod crustaceans: needs, problems, and possible solutions. In: Breithaupt T, Thiel M (Eds.), Chemical Communication in Crustaceans. Springer, New York, p.485–506. https://doi.org/10.1007/978-0-387-77101-4_25
Bhavan PS, Radhakrishnan S, Seenivasan C, et al., 2010. Proximate composition and profiles of amino acids and fatty acids in the muscle of adult males and females of commercially viable prawn species Macrobrachium rosenbergii collected from natural culture environments. Int J Biol, 2(2):107–119. https://doi.org/10.5539/ijb.v2n2p107
Brodsky VY, Malchenko LA, Butorina NN, et al., 2017. Glutamic acid as enhancer of protein synthesis kinetics in hepatocytes from old rats. Biochemistry (Moscow), 82(8):957–961. https://doi.org/10.1134/S0006297917080119
Caprio J, Derby CD, 2010. Aquatic animal models in the study of chemoreception. Senses: A Compr Ref, 4:97–134. https://doi.org/10.1016/B978-012370880-9.00073-6
Carter CG, Mente E, 2014. Protein synthesis in crustaceans: a review focused on feeding and nutrition. Central Eur J Biol, 9(1):1–10. https://doi.org/10.2478/s11535-013-0134-0
Chang ES, Bruce MJ, Tamone SL, 1993. Regulation of crustacean molting: a multi-hormonal system. Am Zoologist, 33(3):324–329. https://doi.org/10.1093/icb/33.3.324
Cuzon G, Cahu C, Aldrin JF, et al., 1980. Starvation effect on metabolism of Penaeus japonicus. Proc World Mariculture Soc, 11(1–4):410–423. https://doi.org/10.1111/j.1749-7345.1980.tb00135.x
de Faria SC, Augusto AS, McNamara JC, 2011. Intra- and extracellular osmotic regulation in the hololimnetic Caridea and Anomura: a phylogenetic perspective on the conquest of fresh water by the decapod Crustacea. J Comp Physiol B, 181(2):175–186. https://doi.org/10.1007/s00360-010-0522-6
Derby CD, Sorensen PW, 2008. Neural processing, perception, and behavioral responses to natural chemical stimuli by fish and crustaceans. J Chem Ecol, 34(7):898–914. https://doi.org/10.1007/s10886-008-9489-0
Fujimori T, Abe H, 2002. Physiological roles of free d- and L-alanine in the crayfish Procambarus clarkii with special reference to osmotic and anoxic stress responses. Comp Biochem Physiol Part A: Mol Integr Physiol, 131(4):893–900. https://doi.org/10.1016/S1095-6433(02)00006-5
Gäde G, Marco HG, 2006. Structure, function and mode of action of select arthropod neuropeptides. Stud Nat Prod Chem, 33:69–139. https://doi.org/10.1016/S1572-5995(06)80026-8
Hay ME, 2011. Crustaceans as powerful models in aquatic chemical ecology. In: Breithaupt T, Thiel M (Eds.), Chemical Communication in Crustaceans. Springer, New York, p.41–62. https://doi.org/10.1007/978-0-387-77101-4_3
Henderson JW, Ricker RD, Bidlingmeyer BA, et al., 2000. Rapid, accurate, sensitive, and reproducible HPLC analysis of amino acids: amino acid analysis using Zorbax Eclipse-AAA columns and the Agilent 1100 HPLC. Agilent Technologies, USA. https://www.agilent.com/cs/library/chromatograms/59801193.pdf [Accessed on Jun. 18, 2019].
Höglund E, Bakke MJ, Øverli O, et al., 2005. Suppression of aggressive behaviour in juvenile Atlantic cod (Gadus morhua) by l-tryptophan supplementation. Aquaculture, 249(1–4):525–531. https://doi.org/10.1016/j.aquaculture.2005.04.028
Hseu JR, Lu FI, Su HM, et al., 2003. Effect of exogenous tryptophan on cannibalism, survival and growth in juvenile grouper, Epinephelus coioides. Aquaculture, 218(1–4):251–263. https://doi.org/10.1016/S0044-8486(02)00503-3
Justo CC, Aida K, Hanyu I, 1991. Effects of photoperiod and temperature on molting, reproduction and growth of the freshwater prawn Macrobrachium rosenbergii. Nippon Suisan Gakk, 57(2):209–217. https://doi.org/10.2331/suisan.57.209
Kamaruding NA, Ismail N, Ikhwanuddin M, 2017. Characterization of molting stages in the giant freshwater prawn, Macrobrachium rosenbergii using setagenesis of pleopod. Songklanakarin J Sci Technol, 40(2):397–401. https://doi.org/10.14456/sjst-psu.2018.35
Kato H, Rhue MR, Nishimura T, 1989. Role of free amino acids and peptides in food taste. In: Teranishi R, Buttery RG, Shahidi F (Eds.), Flavor Chemistry. Trends and Developments. American Chemical Society, Washington, p.158–174. https://doi.org/10.1021/bk-1989-0388.ch013
Keenan CP, Blackshaw A, 1999. Mud crab aquaculture and biology: Proceedings of an International Scientific Forum held in Darwin. Australian Centre for International Agricultural Research, Canberra, Australia.
Lachaise F, le Roux A, Hubert M, et al., 1993. The molting gland of crustaceans: localization, activity, and endocrine control (a review). J Crustacean Biol, 13(2):198–234. https://doi.org/10.1163/193724093X00020
Laranja JLQ Jr, Quinitio ET, Catacutan MR, et al., 2010. Effects of dietary l-tryptophan on the agonistic behavior, growth and survival of juvenile mud crab Scylla serrata. Aquaculture, 310(1–2):84–90. https://doi.org/10.1016/j.aquaculture.2010.09.038
Liu TY, Boykins RA, 1989. Hydrolysis of proteins and peptides in a hermetically sealed microcapillary tube: high recovery of labile amino acids. Anal Biochem, 182(2):383–387. https://doi.org/10.1016/0003-2697(89)90612-X
Luvizotto-santos R, Lee JT, Branco ZP, et al., 2003. Lipids as energy source during salinity acclimation in the euryhaline crab Chasmagnathus granulata dana, 1851 (crustaceagrapsidae). J Exp Zool Part A: Compar Exp Biol, 295A(2):200–205. https://doi.org/10.1002/jez.a.10219
Marshall S, Warburton K, Paterson B, et al., 2005. Cannibalism in juvenile blue-swimmer crabs Portunus pelagicus (Linnaeus, 1766): effects of body size, moult stage and refuge availability. Appl Anim Behav Sci, 90(1):65–82. https://doi.org/10.1016/j.applanim.2004.07.007
McCallum ML, Weston SD, Tilahun Y, 2018. Angular substrate preference and molting behavior of the Giant River Prawn, Macrobrachium rosenbergii and its implications for cannibalism management. BioRxiv, preprint. https://doi.org/10.1101/354472
McNamara JC, Rosa JC, Greene LJ, et al., 2004. Free amino acid pools as effectors of osmostic adjustment in different tissues of the freshwater shrimp Macrobrachium olfersii (Crustacea, Decapoda) during long-term salinity acclimation. MarFreshw Behav Physiol, 37(3):193–208. https://doi.org/10.1080/10236240400006208
Mente E, Coutteau P, Houlihan DF, et al., 2002. Protein turnover, amino acid profile and amino acid flux in juvenile shrimp Litopenaeus vannamei: effects of dietary protein source. J Exp Biol, 205(20):3107–3122.
Nair KKC, Bransilav M, Rosenthal H, et al., 1999. Experimental studies on the cannibalistic habit of Macrobrachium rosenbergii (de Man). The Fourth Indian Fisheries Forum Proceeding, 24:227–232.
Okuma E, Abe H, 1994. Simultaneous determination of d- and l-amino acids in the nervous tissues of crustaceans using precolumn derivatization with (+)-1-(9-fluorenyl)ethyl chloroformate and reversed-phase ion-pair high-performance liquid chromatography. J Chromatogr B: Biomed Sci Appl, 660(2):243–250. https://doi.org/10.1016/0378-4347(94)00304-1
Peebles B, 1978. Molting and mortality in Macrobrachium rosenbergii. J World Aquacult Soc, 9(1–4):39–46. https://doi.org/10.1111/j.1749-7345.1978.tb00229.x
Romano N, Zeng CS, 2017. Cannibalism of decapod crustaceans and implications for their aquaculture: a review of its prevalence, influencing factors, and mitigating methods. Rev Fish Sci Aquacult, 25(1):42–69. https://doi.org/10.1080/23308249.2016.1221379
Schmidt M, Mellon D Jr, 2010. Neuronal processing of chemical information in crustaceans. In: Breithaupt T, Thiel M (Eds.), Chemical Communication in Crustaceans. Springer, New York, p. 123–147. https://doi.org/10.1007/978-0-387-77101-4_7
Sefiani M, le Caer JP, Soyez D, 1996. Characterization of hyperglycemic and molt-inhibiting activity from sinus glands of the penaeid shrimp Penaeus vannamei. Gen Comp Endocrinol, 103(1):41–53. https://doi.org/10.1006/gcen.1996.0092
Shinji J, Okutsu T, Jayasankar V, et al., 2012. Metabolism of amino acids during hyposmotic adaptation in the whiteleg shrimp, Litopenaeus vannamei. Amino acids, 43(5):1945–1954. https://doi.org/10.1007/s00726-012-1266-2
Skinner DM, 1985. Interacting factors in the control of the crustacean molt cycle. Am Zoologist, 25(1):275–284. https://doi.org/10.1093/icb/25.1.275
Wang L, Xu RJ, Hu B, et al., 2010. Analysis of free amino acids in Chinese teas and flower of tea plant by high performance liquid chromatography combined with solidphase extraction. Food Chem, 123(4):1259–1266. https://doi.org/10.1016/j.foodchem.2010.05.063
Waterman TH, 1960. The Physiology of Crustacea. Academic Press, New York, p.670–681.
Webster SG, Keller R, 1986. Purification, characterisation and amino acid composition of the putative moult-inhibiting hormone (MIH) of Carcinus maenas (Crustacea, Decapoda). J Comp Physiol B, 156(5):617–624. https://doi.org/10.1007/BF00692738
Wu GY, Wu ZL, Dai ZL, et al., 2013. Dietary requirements of “nutritionally non-essential amino acids” by animals and humans. Amino Acids, 44(4):1107–1113. https://doi.org/10.1007/s00726-012-1444-2
Wu GY, Bazer FW, Dai ZL, et al., 2014. Amino acid nutrition in animals: protein synthesis and beyond. Annu Rev Anim Biosci, 2:387–417. https://doi.org/10.1146/annurev-animal-022513-114113
Yano H, Aso K, Tsugita A, 1990. Further study on gas phase acid hydrolysis of protein: improvement of recoveries for tryptophan, tyrosine, and methionine. J Biochem, 108(4):579–582. https://doi.org/10.1093/oxfordjournals.jbchem.a123245
Yasuda A, Yasuda Y, Fujita T, et al., 1994. Characterization of crustacean hyperglycemic hormone from the crayfish (Procambarus clarkii): multiplicity of molecular forms by stereoinversion and diverse functions. Gen Comp Endocrinol, 95(3):387–398. https://doi.org/10.1006/gcen.1994.1138
Zarubin TP, Chang ES, Mykles DL, 2009. Expression of recombinant eyestalk crustacean hyperglycemic hormone from the tropical land crab, Gecarcinus lateralis, that inhibits Y-organ ecdysteroidogenesis in vitro. Mol Biol Rep, 36(6):1231. https://doi.org/10.1007/s11033-008-9302-8
Acknowledgments
Abu Seman JUNETA-NOR is grateful for the financial support received from the Ministry of Higher Education of Malaysia for the Postgraduate Scholarship Programme (MyPhD-MyBrain15). Mohamad Nor AZRA is grateful for support from the same organization to his Post-Doctoral Program in the Institute of Tropical Aquaculture and Fisheries, Universiti Malaysia Terengganu, Malaysia. Special thanks also goes to late Associate Professor Dr. Safiah JASMANI on her support during initial draft methodology in this work.
Author information
Authors and Affiliations
Contributions
Abu Seman JUNETA-NOR and Mhd IKHWANUDDIN conceived and designed the study, drafted the paper, and took responsibility for the integrity of the data and the accuracy of the data analysis. Noordiyana Mat NOORDIN, Hong-yu MA, and Norainy Mohd HUSIN developed the statistical methods and interpreted the data. Mohamad Nor AZRA revised the manuscript. All authors agree to be accountable for all aspects of the work and have read and approved the manuscript.
Corresponding authors
Additional information
Compliance with ethics guidelines
Abu Seman JUNETA-NOR, Noordiyana Mat NOORDIN, Mohamad Nor AZRA, Hong-yu MA, Norainy Mohd HUSIN, and Mhd IKHWANUDDIN declare that they have no conflict of interest.
All institutional and national guidelines for the care and use of laboratory animals were followed.
Project supported by the Fundamental Research Grant Scheme from the Ministry of Higher Education, Malaysia (No. 59324)
Rights and permissions
About this article
Cite this article
Juneta-Nor, A.S., Noordin, N.M., Azra, M.N. et al. Amino acid compounds released by the giant freshwater prawn Macrobrachium rosenbergii during ecdysis: a factor attracting cannibalistic behaviour. J. Zhejiang Univ. Sci. B 21, 823–834 (2020). https://doi.org/10.1631/jzus.B2000126
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B2000126