Abstract
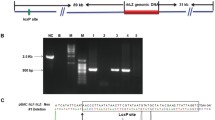
To develop a gene therapy strategy for treating bovine mastitis, a new mammary-specific vector containing human lysozyme (hLYZ) cDNA and kanamycin resistance gene was constructed for intramammary expression and clinical studies. After one time acupuncture or intracisternal infusion of healthy cows with 400 μg of the p215C3LYZ vector, over 2.0 μg/ml of rhLYZ could be detected by enzymatic assay for about 3 weeks in the milk samples. Western blotting showed that rhLYZ secreted into milk samples from the vector-injected cows had molecular weight similar to that of the natural hLYZ in human colostrums. Twenty days after the primary injection, the quarters were re-injected with the same vector by quarter acupuncture and even higher concentrations of rhLYZ could be detected. Indirect competitive ELISA of milk samples showed that the vector injection did not induce detectable humoral immune response against hLYZ. Clinical studies showed that twice acupuncture of quarters with the p215C3LYZ vector had overt therapeutic effect on clinical and subclinical mastitis previously treated with antibiotics, including disappearance of clinical symptoms and relatively high microbiological cure rates. These data provide a solid rationale for using the vector to develop gene therapy for treating bovine mastitis.
Similar content being viewed by others
References
Chen, X., Niyonsaba, F., Ushio, H., Okuda, D., Nagaoka, I., Ikeda, S., Okumura, K., Ogawa, H., 2005. Synergistic effect of antibacterial agents human β-defensins, cathelicidin LL-37 and lysozyme against Staphylococcus aureus and Escherichia coli. J. Dermatol. Sci., 40(2):123–132. [doi:10.1016/j.jdermsci.2005.03.014]
Erskine, R., Cullor, J., Schaellibaum, M., Yancey, B., Zecconi, A., 2004. Bovine Matitis Pathogens and Trends in Resistance to Antibacterial Drugs. NMC Annual Meeting Proceedings, p.400–403.
Fan, W., Plaut, K., Bramley, A.J., Barlow, J.W., Kerr, D.E., 2002. Adenoviral-mediated transfer of a lysostaphin gene into the goat mammary gland. J. Dairy Sci., 85(7):1709–1716.
Fleming, A., 1922. On a remarkable bacteriolytic element found in tissues and secretions. Proceedings of the Royal Society, B93:306.
Gruet, P., Maincent, P., Berthelot, X., Kaltsatos, V., 2001. Bovine mastitis and intramammary drug delivery: review and perspectives. Advanced Drug Delivery Reviews, 50(3):245–259. [doi:10.1016/S0169-409X(01)00160-0]
Hurley, I.P., Coleman, R.C., Ireland, H.E., Williams, J.H.H., 2004. Measurement of bovine IgG by indirect competitive ELISA as a means of detecting milk adulteration. J. Dairy Sci., 87(3):543–549.
Ito, Y., Yamada, H., Imoto, T., 1992. Colorimetric assay for lysozyme using Micrococcus luteus labeled with a blue dye, Remazol brilliant blue R, as a substrate. Chem. and Pharm. Bull., 40(6):1523–1526.
Lander, R.J., Winters, M.A., Meacle, F.J., Buckland, B.C., Lee, A.L., 2002. Fractional precipitation of plasmid DNA from lysate by CTAB. Biotechnol. Bioeng., 79(7):776–784. [doi:10.1002/bit.10335]
Li, G.C., Sun, H.C., Sun, Q., Chen, G., Zhang, Q., Shi, W.Q., Li, H.D., 2004. Transgenic mice expressing human lysozyme gene in milk. Chin. J. Microbiol. Immunol., 24(2):142–145 (in Chinese).
Maga, E.A., Anderson, G.B., Murray, J.D., 1995. The effect of mammary gland expression of human lysozyme on the properties of milk from transgenic mice. J. Dairy Sci., 78(12):2645–2652.
Middleton, J.R., Hardin, D., Steevens, B., Randle, R., Tyler, J.W., 2004. Use of somatic cell counts and california mastitis test results from individual quarter milk samples to detect subclinical intramammary infection in dairy cattle from a herd with a high bulk tank somatic cell count. J. Am. Vet. Med. Assoc., 224(3):419–423. [doi:10.2460/javma.2004.224.419]
Murphy, D.B., Epstein, S.L., 1998. Guidance for Human Somatic Cell Therapy and Gene Therapy. Food and Drug Administration, Rockville, MD.
Norman, J.A., Hobart, P., Manthorp, M., Felgner, P., 1997. Development of improved vectors for DNA-based immunization and other gene therapy applications. Vaccine, 15(8):801–803. [doi:10.1016/S0264-410X(96)00247-2]
Sambrook, J., Russell, D.W., 2002. Molecular Cloning, a Laboratory Manual. Cold Spring Harbor Laboratory Press, NY, USA.
Sordillo, L.M., Shafer-Weaver, K., DeRosa, D., 1997. Immunology of the mammary gland. J. Dairy Sci., 80:1851–1865.
Steinhoff, U.M., Senft, B., Seyfert, H.M., 1994. Lysozyme-encoding bovine cDNAs from neutrophile granulocytes and mammary gland are derived from a different gene than stomach lysozyme. Gene, 143(2):271–276. [doi:10.1016/0378-1119(94)90109-0]
Sun, H.C., Yu, F., Su, J.H., Wu, C.H., Zhang, Q., Li, G.C., 2004. Preliminary studies on gene therapy for dairy cow mastitis using human lysozyme gene. Chinese Journal of Animal and Veterinary Sciences, 35(2):227–232 (in Chinese).
Wall, R.J., Powell, A.M., Paape, M.J., Kerr, D.E., Bannerman, D.D., Pursel, V.G., Wells, K.D., Talbot, N., Hawk, H.W., 2005. Genetically enhanced cows resist intramammary Staphylococcus aureus infection. Nat. Biotechnol., 23(4):445–451. [doi:10.1038/nbt1078]
Author information
Authors and Affiliations
Corresponding author
Additional information
Project (Nos. BJ2001315 and BE2004611) supported by the Department of Science and Technology of Jiangsu Province, China
Rights and permissions
About this article
Cite this article
Sun, Hc., Xue, Fm., Qian, K. et al. Intramammary expression and therapeutic effect of a human lysozyme-expressing vector for treating bovine mastitis. J. Zhejiang Univ. - Sci. B 7, 324–330 (2006). https://doi.org/10.1631/jzus.2006.B0324
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1631/jzus.2006.B0324