Abstract
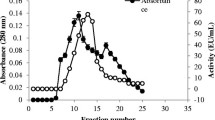
The chitinase producing bacterial strain was isolated from the vermicompost amended site of Mehsana district of Gujarat, India, and identified as Bacillus safensis MBCU6 using 16S rDNA sequencing. The chitinase was purified by ammonium sulfate precipitation followed by diethylaminoethanol sepharose CL-6B column chromatography. The purified enzyme could be demonstrated as a single band on sodium dodecyl sulfate polyacryalamide gel electrophoresis analysis as well as clear zone on zymogram, with estimated molecular mass of 58 kDa. The optimum pH and temperature of chitinase were pH 7.0 and 60°C, respectively. The purified chitinase exhibited high degree of antifungal activity particularly against pathogenic Macrophomina phaseolina (60%) and Rhizoctonia solani (73%) by dissolving their cell wall components. The purified enzyme could hydrolyze colloidal chitin to its oligomers. It infers that the chitinase produced by Bacillus safensis may play a significant role in the activity as a biopesticide and bioactive material production.
Similar content being viewed by others
Abbreviations
- DEAE:
-
diethylaminoethanol
- NAG:
-
N-aetylglucosamine
- SDS-PAGE:
-
sodium dodecyl sulfate poly-acryalamide gel electrophoresis
- TLC:
-
thin-layer chromatography
References
Ahmadian G., Degrassi G., Venturi V., Zeigler D.R., Soudi M. & Zanguinejad P. 2007. Bacillus pumilus SG2 isolated from saline conditions produces and secretes two chitinases. J. Appl. Microbiol. 103. 1081–1089.
Chang W.T., Chen M.L. & Wang S.L. 2010. An antifungal chiti-nase produced by B. subtilis using chitin waste as a carbon source. World J. Microbiol. Biotechnol. 26. 945–950.
Claus D. & Berkeley R.C.W. 1984. Genus Bacillus, pp. 1104–1139. In: Sneath P.H.A. (ed.), Bergey’s Manual of Systematic Bacteriology, Williams & Wilkins, Baltimore.
Chang W.T., Chen C. & Wang S.L. 2003. An antifungal chitinase produced by B. cereus using shrimp and crab shell powder as a carbon source. Curr. Microbiol. 47. 102–108.
Fravel D. 2005. Commercialization and implementation of bio-control. Annu. Rev. Phytopathol. 43. 337–359.
Ghasemi S., Ahmadian G., Jelodar N.B., Rahimian H., Ghandili S., Dehestani Ali. & Shariati P. 2010. Antifungal chitinases from B. pumilus SG2: preliminary report. World J. Microbiol. Biotechnol. 26. 1437–1443.
Gohel V., Singh A., Vimal M., Ashwini P. & Chhatpar H.S. 2006. Bioprospecting and antifungal potential of chitinolytic microorganisms. Afr. J. Biotechnol. 5. 54–72.
Halder S.K., Maity C., Jana A., Das A., Paul T., Mohapatra P.K.D., Pati B.R. & Mondal K.C. 2013. Proficient biodegra-dation of shrimp shell waste by Aeromonas hydrophila SBK1 for the concomitant production of antifungal chitinase and antioxidant chitosaccharides. Int. Biodeter. Biodegrad. 79
Halder S.K., Maity C., Jana A., Pati B.R. & Mondal K.C. 2012. Chitinolytic enzymes from the newly isolated Aeromonas hy-drophila SBK1: study of the mosquitocidal activity. BioCon-trol 57: 441–449.
Hammani I., Siala R., Jridi M., Ktari N., Nasir M. & Triki M.A. 2013. Partial purification and characterization of chiIO8, a novel antifungal chitinase produced by Bacillus cereus IO8. J. Appl. Microbiol. 115. 358–366.
Heydari A. & Pessarakli M. 2010. A review on biological control of fungal plant pathogens using microbial antagonists. J. Biol. Sci. 10. 273–290.
Imoto T. & Yagishita K. 1971. A simple activity measurement by lysozyme. Agric. Biol. Chem. 35. 1154–1156.
Kandra P., Challa M.M. & Jyothi H.K. 2012. Efficient use of shrimp waste: present and future trends. Appl. Microbiol. Biotechnol. 93:17–29.
Kishore G.K., Pande S. & Podile A.R. 2005. Biological control of late leaf spot of peanut (Arachis hypogaea) with chitinolytic bacteria. Phytopathology 95: 1157–1165.
Laemmli U.K. 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685.
Lee Y.S., Park I.H., Yoo J.S., Chung S.Y., Lee Y.C., Cho Y.S., Ahn S.C., Kim C.M. & Choi Y.L. 2007. Cloning, purification, and characterization of chitinase from Bacillus sp. DAU101. Bioresour. Technol. 98. 2734–2741.
Lien T.S., Yu S.T., Wu S.T. & Too J.R. 2007. Induction and purification of a thermophilic chitinase produced by Aeromonas sp. DYU-Too7 using glucosamine. Biotechnol. Bioprocess Eng. 12. 610–617.
Meena S., Gothwal R.K., Saxena J., Mohan M.K. & Ghosh P. 2013. Chitinase production by a newly isolated thermotolerant Paenibacillus sp. BISR-047. Ann. Microbiol. 64. 787–797.
Nawani N.N., Kapadnis B.P., Das A.D., Rao A.S. & Mahajan S.K. 2002. Purification and characterization of a thermophilic and acidophilic chitinase from Microbispora sp. V2. J. Appl. Microbiol. 93. 965–975.
Pandya U., Sudhir A., Gohel H., Subramanian R.B. & Saraf M. 2014. Zymographic identification and biochemical characterization of chitinase against phytofungal pathogens. J. Micro-biol. Biotechnol. Food Sci. 4. 44–47.
Pechsrichuang P., Yoohat K. & Yamabhai M. 2013. Production of recombinant B. subtilis chitosanase, suitable for biosynthesis of chitosan oligosachharides. Bioresour. Technol. 127. 407–414.
Perez-Garcia A., Romero D. & de Vicente A. 2011. Plant protection and growth stimulation by microorganisms: biotech-nological applications of Bacilli in agriculture. Curr. Opin. Biotechnol. 22. 187–193.
Prasanna L., Eijsink V.G.H., Meadow R. & Gaseidnes S. 2013. A novel strain of Brevibacillus laterosporus produces chitinases that contribute to its biocontrol poetential. Appl. Microbiol. Biotechnol. 97. 1601–1611.
Sampson M.N. & Gooday G.W. 1998. Involvement of chitinases of B. thuringiensis in insects. Microbiology 144: 2189–2194.
Shali A., Ghasemi S., Ahmadian G., Ranjbar G., Dehestani Ali., Khalesi N., Mortallebi E. & Vahed M. 2010. B. pumilus SG2 chitinases induced and regulated by chitin, show inhibitory activity against F. graminearum and B.sorokiniana. Phytoparasitica 38: 141–147.
Shanmugam V., Thakur H. & Gupta S. 2013. Use of chitinolytic Bacillus atrophaeus strain S2BC-2 antagonistic to Fus a r i u m spp. for control of rhizome rot of ginger. Ann. Microbiol. 63. 989–996.
Shimahara K. & Takiguchi Y. 1988. Preparation of crustacean chitin. Methods Enzymol. 161C: 417–423.
Songsirittigul C., Lapboonrueng S., Pechsrichuang P., Pesatcha P. & Yamabhai M. 2010. Expression and characterization of B. licheniformis chitinase (ChiA), suitable for bioconversion of chitin waste. Bioresour. Technol. 101. 4096–4103.
Tamura K., Dudley J., Nei M. & Kumar S. 2007. MEGA4: Molecular Evolutionary Genetics Analysis. Mol. Biol. Evolution. 24. 1596–1599.
Tronsmo A. & Harman G.E. 1993. Detection and quantification of N-acetyl-β-D-glucosaminidase, chitobiosidase, and endochitinase in solutions and on gels. Anal. Biochem. 208. 74–79.
Trudel J. & Asselin A. 1989. Detection of chitinase activity after polyacrylamide gel electrophoresis. Anal. Biochem. 178. 362–366.
Tsujibo H., Kubota T., Yamamoto M., Miyamoto K. & Inamori Y. 2003. Characterization of chitinase genes from an alka-liphilic actinomycete, Nocardiopsis prasina OPC-131. Appl. Environ. Microbiol. 69. 894–900.
Velusamy P. & Kim K.Y. 2011. Chitinolytic activity of Enterobacter sp. KB3 antagonistic to R. solani and its role in the degradation of living fungal hyphae. Int. Res. J. Microbiol. 2. 206–214.
Waghmare S.R. & Ghosh J.S. 2010. Chitobiose production by using novel thermostable chitinase from Bacillus licheniformis strain JS isolated from a mushroom bed. Carbohydr. Res. 345. 2630–2635.
Wang S.L. & Chang W.T. 1997. Purification and characterization of two bifunctional chitinase/lysozymes extracellularly produced by P. aeruginosa K-187 in a shrimp and crab shell powder medium. Appl. Environ. Microbiol. 63. 380–386.
Wang S.L., Liu C.P. & Liang T.W. 2012. Fermented and enzymatic production of chitin/chitosan oligosaccharides by extracellular chitinases from B. cereus TKU027. Carbohydr. Polym. 90. 1305–1313.
Wang S.L., Lin T.Y., Yen Y.H., Liao H.F. & Chen Y.J. 2006. Bioconversion of shellfish chitin wastes for the production of B. subtilis W-118 chitinase. Carbohydr. Res. 341. 2507–2515.
Wang S.L., Yieh T.C. & Shih I.L. 1999. Production of antifungal compounds by P. aeruginosa K-187 using shrimp and crab shell powder as a carbon source. Enzyme Microb. Technol. 25. 142–148.
Yamabhai M., Emrat S., Sukasem S., Pesatcha P., Jaruseranee N. & Buranabanyat B. 2008. Secretion of recombinant Bacillus hydrolytic enzymes using Escherichia coli expression systems. J. Biotechnol. 133. 50–57.
Yasir M., Aslam Z., Kim S.W., Lee S.W., Jeon C.O. & Chung R. 2009. Bacterial community composition and chitinase gene diversity of vermicompost with antifungal activity. Bioresour. Technol. 100. 4396–4403.
Acknowledgements
The financial support from Department of Science and Technology (DST), New Delhi, India, under Women Scientist Scheme (WOS-A) (Grant No. SR/WOS-A/LS-186/2010) is gratefully acknowledged. The authors thank Dr. Shailesh R. Waghmare for proof reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pandya, U., Saraf, M. Purification and characterization of antifungal chitinase from Bacillus safensis MBCU6 and its application for production of chito-oligosaccharides. Biologia 70, 863–868 (2015). https://doi.org/10.1515/biolog-2015-0112
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1515/biolog-2015-0112