Abstract
Background: A growing body of neuroimaging studies has reported common neural abnormalities among mental disorders in adults. However, it is unclear whether the distinct disorder-specific mechanisms operate during adolescence despite the overlap among disorders.
Methods: We studied a large cohort of more than 11 000 preadolescent (age 9–10 yr) children from the Adolescent Brain and Cognitive Development cohort. We adopted a regrouping approach to compare cortical thickness (CT) alterations and longitudinal changes between healthy controls (n = 4041) and externalizing (n = 1182), internalizing (n = 1959) and thought disorder (n = 347) groups. Genome-wide association study (GWAS) was performed on regional CT across 4468 unrelated European youth.
Results: Youth with externalizing or internalizing disorders exhibited increased regional CT compared with controls. Externalizing (p = 8 × 10−4, Cohen d = 0.10) and internalizing disorders (p = 2 × 10−3, Cohen d = 0.08) shared thicker CT in the left pars opercularis. The somatosensory and the primary auditory cortex were uniquely affected in externalizing disorders, whereas the primary motor cortex and higher-order visual association areas were uniquely affected in internalizing disorders. Only youth with externalizing disorders showed decelerated cortical thinning from age 10–12 years. The GWAS found 59 genome-wide significant associated genetic variants across these regions. Cortical thickness in common regions was associated with glutamatergic neurons, while internalizing-specific regional CT was associated with astrocytes, oligodendrocyte progenitor cells and GABAergic neurons.
Limitations: The sample size of the GWAS was relatively small.
Conclusion: Our study provides strong evidence for the presence of specificity in CT, developmental trajectories and underlying genetic underpinnings among externalizing and internalizing disorders during early adolescence. Our results support the neurobiological validity of the regrouping approach that could supplement the use of a dimensional approach in future clinical practice.
Introduction
During the past 2 decades, long-standing diagnostic categories of mental disorders, based on the Diagnostic and Statistical Manual of Mental Disorders (DSM) and International Classification of Diseases (ICD), have been increasingly challenged because of unclear boundaries, overlapping symptoms1 and high comorbidity2,3 among mental disorders. Furthermore, growing large-scale neuroimaging meta-analyses,4–8 especially from the ENIGMA Consortium, have identified common and shared neurobiological correlates across mental disorders. For instance, Goodkind and colleagues5 identified a transdiagnostic pattern of grey matter loss in the anterior insula and the dorsal anterior cingulate across severe mental illnesses in adults. Opel and colleagues8 identified a shared latent underlying factor for brain structural abnormalities across 6 major psychiatric disorders. Park and colleagues4 found a cortexwide dimension of morphological changes across 6 major psychiatric disorders. In this context, evidence for unique and specific neural abnormalities supporting natural boundaries between disorders as well as between specific disorders and healthy states has not been conclusively shown, raising uncertainty about the biological basis of diagnosable mental disorders.
In this context, according to Hyman,9 there are generally 2 ways to advance an alternative or refine the current categorical diagnostic system: a dimensional approach, conceptualizing psychopathology as a continuum or a spectrum10–12 and a regrouping approach, which regroups mental disorders into large families based on neurobiological and genetic hypotheses. Growing evidence10–12 highlights the advantages of dimensional approaches over current diagnostic categories in delineating the nature of psychopathology and addressing blurring boundaries, which would increase validity. Nevertheless, in the attempt to replace the current categorical system in research, treatment decisions, teaching and public communication where it finds clinical utility, describing alternate diagnostic units based on a dimensional approach will be a critical step.1,9,13 Repeatedly observed lower-order dimensions in psychopathology enable the regrouping of mental disorders with similar etiology or pathophysiology into broader diagnostic families. This promises to remove redundant boundaries and refine the bewildering variety of diagnostic labels, striking a trade-off between validity and utility.9 For example, most childhood mental disorders could be regrouped into externalizing symptoms (inattention, aggressive and disruptive behaviour), internalizing symptoms (depression, anxiety and fear) and thought disorders (delusions, hallucinations and obsessions).10 Single mental disorders within each of these broad diagnostic families likely share similar pathophysiology. For instance, major depressive disorder (MDD), social anxiety disorder and specific phobia are all internalizing disorders, likely all sharing defects in the regulation of negative emotions.14 However, such a regrouping approach requires biological validation from the level of neural circuits and genetics.
During childhood and adolescence, a period when most mental disorders originate, the brain undergoes major developmental changes in cortical morphology.2 A large body of research has linked abnormal cortical thickness (CT) development during this stage to multiple mental disorders15,16 and impaired cognitive performance.17 However, since most prior neuroimaging genome-wide association studies (GWAS)18–21 used adult samples from resources like the UK Biobank, little is known about the underlying genetic mechanisms of abnormal CT during adolescence, especially when it comes to associated genetic variants, genes, pathways and cell types.
In the present study, we hypothesized that there are specific CT alterations with distinct developmental trajectories and underlying genetic mechanisms for broad diagnostic groups of externalizing, internalizing and thought disorders during early adolescence. We tested our hypotheses in the Adolescent Brain and Cognitive Development (ABCD) Study (n = 11 878), a large adolescent longitudinal cohort. In addition, we performed GWAS, gene set enrichment analysis (GSEA) and cell type specificity analysis (CTSA) to explore underlying genetic variants, genes, pathways and cell types of CT alterations.
Methods
Participants
Participants were preadolescents aged 9–10 years (n = 11 878) recruited from 21 research sites across the United States from the ABCD Study (Release 3.0, November 2020). This longitudinal, multisite, population-representative cohort provides comprehensive clinical, behavioural, cognitive and multimodal neuroimaging data from baseline and from a 2-year follow-up (12 yr old) assessment. The sample filtering used in the analysis procedures is described in Appendix 1, Figure S1, available at jpn.ca/lookup/doi/10.1503/jpn.220202/tab-related-content.
Regrouping approach for broad diagnostic families
Single mental disorder diagnoses were determined using parental ratings in the computerized Kiddie Schedule for Affective Disorders and Schizophrenia (KSADS) based on DSM-5 criteria. For the current analyses, life-time diagnoses (past or present) of the 18 single mental disorders, including specific thought disorder symptoms (e.g., delusions and hallucinations, which do not consolidate into a diagnosis at this age), were used (Figure 1A and Appendix 1, Table S1). This regrouping of disorders is generally consistent with prior studies.22–24 Healthy controls (Table 1) were those who did not meet diagnostic criteria for any disorder based on the KSADS (including disorders that are not included in the 3 broad diagnostic families, such as eating disorders, sleep problems and other unspecified disorders). Demographic characteristics for patients of 3 diagnostic families and each single mental disorder are shown in Appendix 1, Table S1.
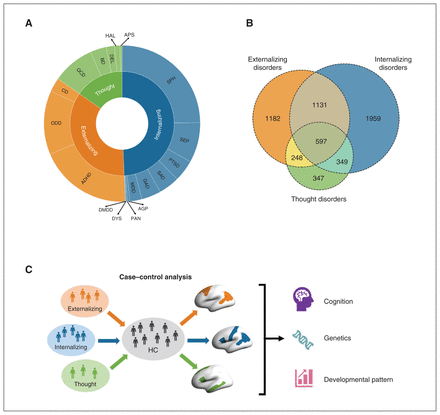
Components and comorbidity of externalizing, internalizing and thought disorders. A) Eighteen single mental disorders (outer circle) were classified into 3 broad diagnostic families (inner circle), i.e., externalizing, internalizing and thought disorders. B) The Venn diagram illustrates the large overlap among 3 diagnostic families. C) An overview of our current study. ADHD = attention deficit/hyperactivity disorder; AGP = agoraphobia; APS = associated psychotic symptoms; BD = bipolar disorder; CD = conduct disorder; DMDD = disruptive mood dysregulation disorder; DEL = delusions; DYS = dysthymia; GAD = generalized anxiety disorder; HAL = Hallucinations; HC = healthy controls; MDD = major depressive disorder; OCD = obsessive–compulsive disorder; ODD = oppositional defiant disorder; PAN = panic disorder; PTSD = posttraumatic stress disorder; SEP = separation anxiety disorder; SPH = specific phobia; SAD = social anxiety disorder.
Demographic characteristics of 3 subsets of broad diagnostic families without comorbidity and healthy controls from the Adolescent Brain and Cognitive Development Study
Structural image acquisition and quality control
Participants completed a high-resolution T1-weighted structural MRI scan (1 mm isotropic voxels) on 3 T scanners (Siemens Prisma, General Electric MR 750, Philips). Structural MRI data processing was completed using FreeSurfer version 5.3.0 according to standardized processing pipelines. 25 All scans underwent radiological review to identify incidental findings. We excluded 644 youth who did not pass visual inspection of T1 images and FreeSurfer quality control26 (imgincl_t1w_include = = 1) from the following analyses. The present study used post-processed CT data mapped to 34 cortical parcellations per hemisphere based on the Desikan–Killiany Atlas.27
NIH Toolbox cognition measures
The NIH Toolbox cognition batteries consist of 7 tasks measuring executive function, episodic memory, working memory, information processing, language abilities and 3 summary scores including crystal, fluid and total intelligence. Details of cognitive measures are listed in Appendix 1, Table S3.
Statistical analysis
We implemented linear mixed models (LMM) using the R lme4 package28 to examine regional CT difference among the 3 diagnostic families in contrast to healthy controls (Figure 1C). In LMM, regional CT region of interest (ROI) was the dependent variable, and group (healthy controls defined as 0 and patients defined as 1) was the independent variable. All LMM also included crossed random effects for family and scanners and fixed-effects covariates for age, sex, race (White, Black, Hispanic, Asian, other/multiracial), parental marital status, pubertal level, parental education, body mass index and total intracranial volume. Because the occurrence of comorbidity tends to lag behind the initial disorder, 23,29 this progression is likely to follow a pattern of symptom accumulation in children and adolescents.29 Therefore, non-comorbid cases in each diagnostic family may exhibit an earlier state of psychopathology while comorbidities among different broad diagnostic families may represent a subsequent and mixed state of psychopathology, thus obscuring the original neural correlates of “core” pathology in externalizing, internalizing and thought disorders. Considering the high comorbidity among 3 diagnostic families (Figure 1B), to detect the anatomic alterations for the former state within the 3 diagnostic families, we conducted case–control analyses (1) excluding comorbid patients outside their primary diagnostic families (n = 1182 for externalizing disorders, n = 1959 for externalizing disorders and n = 347 for thought disorder) and (2) including comorbid cases (n = 3158 for externalizing disorders, n = 4036 for externalizing disorders and n = 1541 for thought disorders). We used the Benjamini–Hochberg30 false discovery rate (FDR) in all analyses for multiple comparisons, and the findings based on the adjusted p values (pFDR) are reported. The demographic characteristics of comorbid cases are described in Appendix 1, Table S1.
A shared alteration in the 3 diagnostic families was defined as a significant difference (pFDR < 0.05) in CT between any patient group and the healthy control group that was shared by at least 2 patient groups. A disorder-specific alteration (e.g., externalizing-specific) was defined by a significant difference between 1 patient group and the healthy control group, and as well as a nonsignificant difference (pFDR > 0.05) between each of the other 2 patient groups and the healthy control group. This definition was in line with our prior work.31 Notably, this definition of disorder specificity does not indicate exclusivity (i.e., absence of a corresponding change in another patient group), but only that a change with an effect size sufficient for detection at the group level examinations is specific to a group. Furthermore, to test if shared or disorder-specific CT alterations are implicated in cognitive performance at the population level, we also examined the association between these CT alterations and NIH Toolbox cognition measures across the whole ABCD sample.
Longitudinal analysis
We used the 2-year follow-up CT data to evaluate the longitudinal changes in 3 patient groups (n = 575 for externalizing disorders, n = 913 for internalizing disorders, n = 169 for thought disorders and n = 1881 for healthy controls). To this end, we performed 2 comparisons: (1) a case–control comparison for all 3 diagnostic families at the follow-up time point to investigate if baseline CT alterations persisted after 2 years and (2) a comparison of longitudinal CT changes (from 10 to 12 years of age) between the 3 diagnostic families and the healthy control group to identify progressive changes in CT. The LMM in the first comparison used 2-year follow-up covariates (the same as in the above case–control study), whereas the LMM in the second comparison used baseline covariates and included random intercepts defining subject as random factor, time variable (baseline defined as 0) and group × time interactions, which measured the case–control differences in cortical thinning from 10 to 12 years old. We used FDR correction for each of the tested associations.
Genome-wide association study
Before GWAS, we performed genetic ancestry inference, genotype imputation and strict quality control on genotype data, finally filtering out 4468 genetically unrelated preadolescents with European ancestry who passed structural image quality control (Appendix 1). We performed GWAS using plink version 2.032 to examine the genetic variants associated with CT ROIs contributing to the 3 diagnostic families. On the premise of additive genetic effects, general linear regression models were fitted to determine the association between common and disorder-specific CT alterations and allele dosages of single nucleotide polymorphisms (SNPs) in genetically unrelated preadolescents with European ancestry. Sex, age, mean cortical thickness, top 10 genetic principal components (PCs) scores, and study sites were included as covariates. Then, SNPs were annotated and mapped to genes (Appendix 1) using the FUMA33 online platform (version v1.3.7), which is an integrative tool for functional mapping and annotation of genetic associations.
Genes identified using FUMA were further merged separately across common and disorder-specific regions. We performed GSEA using the FUMA “GENE2FUNC” to test if these genes were enriched in specific gene sets including pathways (Kyoto Encyclopedia of Genes and Genomes34 [KEGG]), molecular functions (Gene Ontology35 [GO]), cellular components (GO), biological processes (GO) and other GWAS traits (GWAS Catalog36). Protein-coding genes were set as background genes. Bonferroni correction was performed per gene set using FUMA. We also performed CTSA37 to determine specific associations between cell types and each region of CT.
Additional analysis
The Child Behaviour Checklist (CBCL), completed by the child’s parent or caregiver, is a well-established and widely used scale to assess emotional, behavioural and social problems in children and adolescents.39 The CBCL has various syndrome- or DSM-oriented scales (Appendix 1) capturing 2 broad-band domains: externalizing and internalizing problems, which are similar to the dimensions of psychopathology. To compare with a regrouping approach, we also examined the associations between 20 CBCL subscales and whole brain regional CT (68 ROIs) to explore the profile of CT alterations related to dimensions of psychopathology. We used FDR correction for multiple comparisons (1360 comparisons: 20 CBCL scales × 68 regions). Finally, we compared CT alterations between each of the 18 single mental disorder groups and the healthy control group separately and performed hierarchical clustering on 18 single disorder groups using the z-score of CT alterations across 68 ROIs to test if they clustered regionally within externalizing, internalizing and thought disorders.
Results
Common and unique cortical thickness alterations
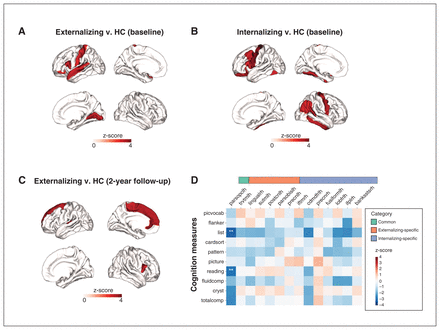
Our case–control analyses, excluding comorbid cases, showed that CT was significantly higher (pFDR < 0.05) in externalizing (5 regions) and internalizing (7 regions) disorders than healthy controls (Appendix 1, Table S2). No regions were significantly altered in either direction in thought disorders. Increases in CT converged across externalizing and internalizing disorders in the left pars opercularis. Regions showing externalizing-specific changes included the left transverse temporal gyrus (Heschl gyrus), superior temporal gyrus (STG), postcentral gyrus and pars orbitalis, while those showing internalizing-specific changes included the bilateral precentral gyrus, right inferior temporal gyrus (ITG), inferior parietal gyrus, left fusiform gyrus and caudal middle frontal gyrus (Figure 2A and B). In contrast, our case–control analyses including comorbid cases (Appendix 1, Table S8 and Figure S3A–C) identified only 5 significant regions (4 for externalizing, 1 for internalizing and 1 for thought), 1 of which was shared by externalizing and internalizing disorders, and 3 of which overlapped those in non-comorbid case–control analyses (left pars opercularis, pars orbitalis and superior temporal gyrus). The left pars opercularis was affected in both externalizing and internalizing disorders, highlighting the transdiagnostic nature of CT changes in this region. Also, CT in the left superior temporal gyrus and pars orbitalis was significantly higher only in externalizing disorders, which was also consistent with the non-comorbid case–control analyses. Correlation analysis (Figure 2C) showed that CT of the left pars opercularis (common region) was associated with lower capacities in reading and working memory. Additional analyses found no significant (pFDR < 0.05) associations between any CBCL subscale and regional CT (Appendix 1, Table S9 and Figure S16).
Regions with significant (false discovery rate–corrected p value [pFDR] < 0.05) thickness alterations at baseline in A) externalizing disorders compared with healthy controls (HC) and B) internalizing disorders compared with healthy controls. C) Regions with significant (pFDR < 0.05) thickness alterations at baseline in externalizing disorders at 2-year follow-up. D) Associations between baseline cortical thickness (CT) in the common, externalizing-specific and internalizing-specific regions (X axis) and NIH Toolbox cognition measures (Y axis). The colour bars represent the z-score of the regression coefficient from linear mixed models. **pFDR < 0.05. For abbreviations of brain regions and NIH Toolbox cognition measures, see Appendix 1, Tables S2 and S3, available at jpn.ca/lookup/doi/10.1503/jpn.220202/tab-related-content.
Cortical thickness developmental pattern
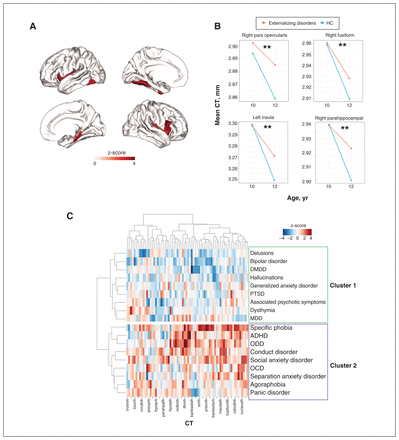
For cross-sectional CT alterations at 12 years (2-yr follow-up), no regions survived FDR correction across the 3 diagnostic families (Appendix 1, Table S5). As to longitudinal changes in regional CT (i.e., within-subject changes from 10 to 12 years old), externalizing, internalizing, thought disorders and healthy control groups all showed notable cortical thinning from 10 to 12 years of age (Figure 3A–D). However, only externalizing disorders showed a decelerated cortical thinning compared with the healthy control group. This deceleration affected 4 regions, including the right pars opercularis, right parahippocampal gyrus, left insula and left fusiform cortex (Figure 4A and B, and Appendix 1, Table S6).
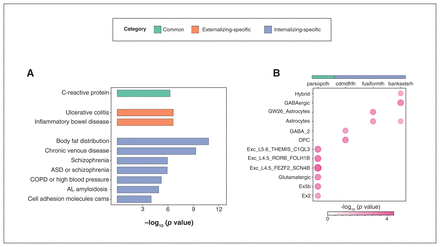
Genetic analyses following genome-wide association study. A) Gene set enrichment analysis of common, externalizing-specific and internalizing-specific genes. Y axis: phenotypes or traits; X axis: –log10 (p value). B) Cell type specificity analysis of cortical thickness (CT) of common and internalizing-specific regions. X axis: regional CT; Y axis: cell type; Value: –log10 (p value). AL amyloidosis = immunoglobulin light chain (AL) amyloidosis; ASD = autism spectrum disorder; COPD = chronic obstructive pulmonary disease; GABA = γ-aminobutyric acid; OPC = oligodendrocyte progenitor cell. For explanations of cell types, please see Appendix 1, Table S16, available at jpn.ca/lookup/doi/10.1503/jpn.220202/tab-related-content.
A) Regions with significantly (false discovery rate–corrected p value [pFDR] < 0.05) decelerated cortical thinning from age 10 to 12 years in externalizing disorders (compared with healthy controls [HC]). The colour bar represents the z-score of the regression coefficient of group × time interaction from linear mixed models (LMM). B) Mean cortical thickness (CT) of the above regions at ages 10 and 12 years in externalizing disorders and healthy controls. C) Hierarchical clustering of 18 single mental disorders based on regional CT alterations. The colour bar represents the z-score of the regression coefficient from LMM. X axis: 68 CT regions of interest; Y axis: 18 single mental disorders. The hierarchical clustering identified 2 clusters of single mental disorders (cluster 1 and cluster 2). ADHD = attention-deficit/hyperactivity disorder; DMDD = disruptive mood dysregulation disorder; MDD = major depressive disorder; OCD = obsessive–compulsive disorder; ODD = oppositional defiant disorder; PTSD = posttraumatic stress disorder.
Hierarchical clustering of 18 single mental disorders
The clustering analysis (Figure 4C) showed that 18 single diagnostic descriptors formed 2 broad clusters based on regional CT alterations. Cluster 1 included most thought disorders and 1 subset of internalizing disorders — posttraumatic stress disorder (PTSD), major depressive disorder (MDD) and generalized anxiety disorder. Cluster 2 included externalizing disorders and 1 subset of internalizing disorders — social anxiety disorder, specific phobia and panic disorder. Essentially, the single disorders from internalizing disorders were separated into 2 clusters, while the single disorders from externalizing disorders and thought disorders segregated into distinct clusters, again indicating specificity. In general, cluster 2 (particularly for specific phobia, attention-deficit/hyperactivity disorder [ADHD], oppositional defiant disorder [ODD] and conduct disorder) showed much higher regional CT than cluster 1.
Genetic underpinnings of regional cortical thickness alterations
We performed GWAS using CT of common and disorder-specific regions (altogether 11 significant ROIs) in non-comorbid case–control analyses using 4468 unrelated individuals of European ancestry. Under the classic genome-wide threshold of p < 5 × 10−8, we identified 59 genome-wide significant independent SNPs after clumping, across 6 regions (Appendix 1, Table S7). This includes 1 common region (left pars opercularis), 1 externalizing-specific region (left postcentral gyrus) and 4 internalizing-specific regions (right inferior parietal lobule, left caudal middle frontal and bilateral precentral gyrus). For Manhattan plots and Q-Q plots of these regions see Appendix 1, Figures S4–S15. After Bonferroni correction for multiple comparisons, 52 SNPs remained significant (p < 3.57 × 10−9, 5 × 10−8; 14 ROIs). All SNPs in LD with these genome-wide significant SNPs were mapped to genes using positional, expression quantitative trait loci (eQTL) and 3D chromatin interaction mapping in FUMA. In all, 13, 52 and 446 genes were mapped to common, externalizing-specific and internalizing-specific regions, respectively (Appendix 1, Tables S10–12). The GSEA identified terms/phenotypes significantly (pBonferroni < 0.05) associated with common, externalizing-specific and internalizing-specific genes (Figure 3A and Appendix 1, Tables S13–15). Common genes were significantly associated with C-reactive protein. Externalizing-specific genes were significantly associated with ulcerative colitis and inflammatory bowel disease, while internalizing-specific genes were significantly associated with body fat distribution, chronic venous disease, schizophrenia and immunoglobulin light chain amyloidosis. The CTSA detected cell types significantly (pBonferroni < 0.05) linked with CT in 4 regions (Figure 3B and Appendix 1, Table S16), including the left pars opercularis (common region), and left caudal middle frontal and left fusiform gyrus (2 internalizing-specific regions). Cortical thickness of the left pars opercularis was mainly associated with excitatory neurons and glutamatergic neurons. For internalizing-specific regions, CT of the left caudal middle frontal gyrus was associated with oligodendrocyte progenitor cell (OPC) and GABAergic neurons, while CT of the left fusiform gyrus was associated with astrocytes.
Discussion
Our study underscored both commonality and specificity in the CT alterations, their developmental patterns and the underlying genetic mechanisms among externalizing and internalizing disorders, supporting the utility of a diagnostic regrouping approach when studying neurobiological validity. Our regrouping approach is more aligned to a dimensional approach in theory but relies on traditional categories, meaning that it contributes to both utility (it can inform decisions such as treatment type and whether to treat and/or hospitalize) and validity (it contributes to ongoing efforts for clear classification of the dimensions and categories surrounding mental illness). Prior dimensional studies23,24 also adopted the notion of a “(broad) diagnostic family,” suggesting a shared theoretical background between our regrouping approach and a dimensional approach. This approach seeks to answer the question of whether a group of disorders or a diagnostic family with shared dimensions of psychopathology can share similar anatomy and pathophysiology, and thus have neurobiological validity. The methods used in the present study could be perceived as a necessary transitional effort to bringing traditional categories and newer dimensional approaches together in order to balance the validity and utility of study results and conclusions.9 Additional analysis found no significant associations between CBCL subscales and whole brain regional CT. Our results are also consistent with those of Mewton and colleagues,40 who found no significant associations between regional CT, externalizing, internalizing, thought disorders and p factor in the ABCD cohort. Hence, a dimensional approach may not capture all the neural abnormalities underlying psychopathology in preadolescence, but might do so if it is supplemented by a regrouping approach.
Externalizing and internalizing disorders are both characterized by increased CT. The left pars opercularis is the common region shared by externalizing and internalizing disorders, often seen as the Broca area. This region is involved in inhibitory control and impairments related to externalizing disorders like ADHD.41 The pars opercularis is also located in the ventrolateral prefrontal cortex, which is involved in emotional regulation through direct projections to the ventral medial prefrontal cortex–amygdala pathway.42 A thicker pars opercularis may result in impaired emotional regulation, which in turn leads to internalizing disorders.43 Furthermore, greater CT in this region was associated with worse working memory and reading abilities, implying that shared CT alterations among mental disorders may underscore poor cognitive performance.
Externalizing-specific regions included the left postcentral gyrus, which is responsible for somatosensory processing, while internalizing-specific regions included the bilateral precentral gyrus, which is the site of the primary motor cortex. On the one hand, previous literature has associated the aberration of somatosensory processing with ADHD,44 and abnormalities in the postcentral gyrus could result in somatosensory dysfunction, which has been linked to externalizing disorders and their symptoms.16,45 On the other hand, internalizing symptoms such as anxiety could affect perceptual motor performance,46 and children with depression or anxiety disorders often exhibit poor motor skills and performance.47 Additionally, psychomotor delay is a core symptom of MDD, meaning the alterations of cortical morphology in the precentral gyrus found in these internalizing disorders (anxiety disorders15 and MDD48), may lead to impaired motor abilities and psychomotor delay. For visual and auditory cortices, we also found distinct alterations in externalizing and internalizing disorders. Externalizing patients showed more alterations in primary auditory areas such as the STG and Heschl gyrus. Functional alterations in these areas have previously been linked with ADHD49 and conduct disorder.50 Moreover, hypersensitivity or hyposensitivity to auditory stimuli are common symptoms in ADHD. In terms of internalizing patients, alterations occurred more in higher-order visual association areas like the fusiform gyrus and the ITG, which are both involved in facial recognition and perception.51,52
Our case–control analyses excluding comorbid cases identified 11 significant regions. The case–control analyses including comorbid cases detected only 5 significant regions. Although there were 3 overlapping regions between both case–control analyses (Appendix 1, Tables S2 and S8), the differences before and after including comorbid cases suggested that including comorbid cases likely reduces the ability to discover the underlying neural correlates of earlier states of psychopathology. As Jablensky13 pointed out, the current diagnostic system fragments psychopathology into a variety of “disorders,” of which many are merely symptoms. Such redundant and arbitrary diagnostic labels not only result in many inflated and spurious comorbidities featuring multifaceted but essentially unitary syndromes, but also obscure more reliable comorbidity among disorders with independent and distinguishable etiology.
Our results, paired with previous work,53 provide evidence for the neural underpinnings of comorbidities among externalizing, internalizing and thought disorders. During the 2-year follow-up, only externalizing disorders showed decelerated cortical thinning from ages 10 to 12 years compared with healthy controls, among all 3 diagnostic families. This is consistent with previous literature showing reduced cortical thinning in externalizing disorders or externalizing symptoms in adolescence.16,54 These results suggest that externalizing and internalizing disorders may follow different developmental patterns in CT during adolescence.
There has been a growing realization that traditional diagnostic categories may preclude identification of neurobiological mechanisms, in addition to contributing to high comorbidity and clinical heterogeneity.55,56 Recent studies have identified subtypes based on brain imaging features for ADHD,57 depression,58 psychosis59 and schizophrenia,60 to tackle clinical heterogeneity within patients with the same diagnostic label. However, rather than contributing to a more transdiagnostic framework, such subtyping and/or clustering is still constrained by traditional diagnostic categories. On the other hand, our hierarchical clustering approach collates traditional diagnostic categories into more neurobiologically informed transdiagnostic groups. Our clustering results, based on regional CT alterations for 18 single mental disorders, supported our regrouping approach to some extent. For instance, single disorders from among externalizing disorders were clustered (within cluster 2), but internalizing and thought disorders showed more heterogeneity in the patterns of regional CT alterations. Obsessive–compulsive disorder (OCD) did not cluster with other thought disorders, but did cluster with separation anxiety disorder and panic disorder, suggesting that OCD was more similar to the latter 2 disorders in terms of CT alteration patterns during adolescence. Because different neural modalities have different neurobiological mechanisms, clustering based on only 1 measure (e.g., CT, surface area) may show different patterns of clusters. Integrating multiple neural modalities with a multimodal clustering approach61 can be a promising future step to obtain more stable and neurobiologically consistent patient groups.
We also compared subcortical volume (36 ROIs) among 3 diagnostic families and healthy controls (Appendix 1, Tables S17 and S18) but found no significant associations. Previous studies62–64 have found abnormal subcortical volume, including smaller hippocampal, putamen and amygdala, as well as a larger pallidum, across different mental disorders in mainly adult samples. Nevertheless, the changes in subcortical structures between childhood and young adulthood have been found to be slower than in the cortex.65 Therefore, our results suggest that alterations in subcortical structures related to psychopathology may not be fully apparent during late childhood or early adolescence. More longitudinal analyses in the middle and late stage of adolescence need to be conducted to study how the development of subcortical structures captures the psychopathology.
The GWAS and follow-up analyses identified specific pathways and cell types contributing to externalizing and internalizing disorders, providing genetic underpinnings of common and disorder-specific changes in CT. The GSEA showed that genes associated with CT in the common region (left pars opercularis) were linked to C-reactive protein. C-reactive protein is a commonly used biomarker of peripheral inflammation and has been strongly linked to both internalizing disorders like depression66 and PTSD67 and externalizing disorders like ADHD.68 For genes linked to externalizing-specific regions, phenotypes related to bowel diseases were enriched. A previous study found that children with ADHD were more prone to bowel problems,69 with evidence supporting the link between the gut microbiome, the “gut–brain axis” and ADHD.70
The CTSA uncovered the significant associations between gene expression in excitatory neurons, glutamatergic neurons and CT in the common region. Glutamate is the most abundant excitatory neurotransmitter in the human brain and plays a critical role in synaptogenesis as well as synaptic plasticity. Abnormal function of the glutamatergic system has been implicated in many mental disorders.71 The CTSA also associated astrocytes, OPCs and GABAergic neurons to CT in internalizing-specific regions. Astrocytes are supporting cells that provide homeostatic control and trophic support within the brain and have been proposed as a promising target for mood disorder interventions.72 Previous studies have linked the astrocytes with inflammation in depression. 73,74 The OPCs play an important role in myelinogenesis by generating oligodendrocytes, which are the main myelin-forming cells of the central nervous system and a crucial component in the maintenance of axonal integrity.75 Also, oligodendrocyte alterations have been associated with MDD.76 Our results show that CT alterations associated with internalizing disorders during adolescence may be caused by inadequate myelination, mediated by OPCs and oligodendrocytes. Furthermore, GABAergic deficits have been highlighted as a central and causal role in the etiology of depressive disorders.77 These results shed light on the hidden genetic links between glial cell types, neurotransmitters and regional CT, which then highlight the neural and genetic mechanisms of CT alterations related to psychopathology, especially with internalizing disorders. Our analyses suggest that specific structural alterations are likely to occur during the developmental period. This specificity is underwritten by distinct genetic mechanisms that influence externalizing and internalizing disorders.
Limitations
A limitation of our study is that the sample size of thought disorders was relatively small compared with the samples of externalizing and internalizing disorders. This may account for why we found no significant difference between the thought disorders and the healthy control groups. Additionally, the sample size in our GWAS was also relatively small and was restricted to adolescents of European ancestry. Therefore, our genetic results need further validation in larger adolescent samples. Cross-ancestry GWAS needs to be performed to determine whether our results can be replicated in other populations.
Conclusion
We identified common and unique regional CT increases in externalizing and internalizing disorders in a large preadolescent sample (n = 11 878). The longitudinal analysis suggested that externalizing disorders showed a unique deceleration in cortical thinning. Furthermore, genetic analyses also identified shared and unique genetic underpinnings of externalizing and internalizing disorders. Cortical thickness in common regions was associated with glutamatergic neurons while internalizing-specific regional CT was associated with astrocytes, OPCs and GABAergic neurons. Together, these findings support the neurobiological validity of a regrouping approach to supplement the dimensional approach to preadolescent psychopathology.
Acknowledgements
K. Zhang is Sponsored by Shanghai Pujiang Program. J. Feng is supported by the 111 Project (No. B18015), the key project of Shanghai Science and Technology (No. 16JC1420402), National Key R&D Program of China (No. 2018YFC1312900), and National Natural Science Foundation of China (NSFC 91630314). L. Palaniyappan acknowledges salary support from the Tanna Schulich Chair of Neuroscience and Mental Health, a salary award from the Fonds de recherche du Québec-Santé and research support from the Monique H. Bourgeois Chair in Developmental Disorders and Graham Boeckh Foundation (Douglas Research Centre, McGill University). J. Zhang is supported by Science and Technology Innovation 2030 - Major Projects (Grant No.2021ZD0200204), Science and Technology Innovation 2030 - Brain Science and Brain-Inspired Intelligence Project (Grant No. 2021ZD0200204), Shanghai Municipal Science and Technology Major Project (No.2018SHZDZX01), ZJLab, and NSFC 61973086. The authors acknowledge Ms. Farida Zaher, Douglas Research Centre, for stylistic changes to prepare the final version of the manuscript.
Footnotes
↵* These authors contributed equally to the work.
Competing interests: B.J. Sahakian reports grants from Lundbeck Foundation and Leverhulme Trust; royalties from PopReach; consulting fees from Cambridge Cognition; and honoraria from the Alberta Psychiatric Association. She is a member of the Human Brain Project Science and Infrastructure Advisory Board, the academic representative for the National Institute for Health and Care Excellence guidelines on harmful gambling, and a member of the EBRAINS Science and Technology Committee. T.W. Robbins reports consultancies with Cambridge Cognition, Arcadia, Greenfield Bioventures, Heptares, Takeda, Lundbeck, Merck, Sharp and Dohme; and royalties from Cambridge Cognition. Research Grants with Shionogi and GlaxoSmithKline and editorial honoraria with Springer Nature and Elsevier. L. Palaniyappan reports personal fees from Janssen Canada, Otsuka Canada, SPMM Course Limited UK and the Canadian Psychiatric Association; book royalties from Oxford University Press; and investigator-initiated educational grants from Janssen Canada, Sunovion and Otsuka Canada outside the submitted work. He is also co-editor-in-chief of JPN; he was not involved in the review or decision to accept this manuscript for publication. None of the above-listed companies or funding agencies had any influence on the content of this article. No other competing interests were declared.
Contributors: N. Kuang, G. Schumann, J. Feng, L. Palaniyappan and J. Zhang designed the study. G. Yu, Z. Liu, X. Wu, B. Becker, K. Zhang, H. Fan, S. Peng, J. Kang, G. Dong, X.-M. Zhao, B.J. Sahakian and T.W. Robbins analyzed the data. G. Yu, Z. Liu, X. Wu, N. Kuang, J. Kang, J. Feng, B.J. Sahakian and L. Palaniyappan wrote the article. All of the authors revised it critically for important intellectual content, gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Data sharing: The data from the ABCD Study are available by request (https://abcdstudy.org/scientists/data-sharing/). The GWAS summary statistics of regional CT of 14 ROIs can be downloaded from https://drive.google.com/drive/folders/1a64gvTM5AQAYLdUw7GQxT-GQN1msXw3H?usp=sharing. The tables from Appendix 1 can be downloaded from https://docs.google.com/spreadsheets/d/1q9l1rHJxI2vOT51pWP1CSiriJQID25GM/edit?usp=sharing&ouid=102360571478483263984&rtpof=true&sd=true.
- Received November 6, 2022.
- Revision received February 13, 2023.
- Revision received March 28, 2023.
- Revision received May 2, 2023.
- Accepted May 17, 2023.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/