Abstract
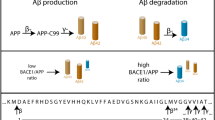
Alzheimer’s disease (AD) is defined by deposits of the 42-residue amyloid-β peptide (Aβ42) in the brain. Aβ42 is a minor metabolite of the amyloid precursor protein (APP), but its relative levels are increased by mutations on APP and presenilins 1 and 2 linked to familial AD. β-secretase (BACE-1), an aspartyl protease, cleaves approx 10% of the APP in neuronal cells on the N-terminal side of Aβ to produce the C-terminal fragment (CTFβ), which is cleaved by γ-secretase to produce mostly Aβ of 40 residues (90%) and approx10% Aβ42. A third enzyme, α-secretase, cleaves APP after Aβ16 to secrete sAPPα and CTFα, the major metabolites of APP. Moreover, previous studies have demonstrated that phorbol esters stimulate processing of APP by α-secretase. Because α-secretase and BACE-1 cleave APP within the secretory pathway, it is likely that the two enzymes compete for the APP substrate. This type of competition can explain the failure to saturate the minor BACE-1 pathway by overexpressing APP in the cell. In this study, we demonstrate that inhibition of constitutive α-secretase processing in a human neuroblastoma cell line does not increase the yield of Aβ, suggesting that the APP substrate targeted for α-secretase processing is not diverted to the BACE-1 pathway. However, when phorbol ester-induced α-secretase was similarly inhibited, we detected an increase in BACE-1 processing and AB yield. We explain these results compartmentalization of BACE-1 and α-secretase with processing depending on sorting of APP to the two compartments. The simplest explanation for the detection of competition between the two pathways upon phorbol ester stimulation is the partial failure of this compartmentalization by phorbol ester-induced release of secretory vesicles.
Similar content being viewed by others
References
Anderson J. P., Esch F. S., Keim P. S., Sambamurti K., Lieberburg I., and Robakis N. K. (1991) Exact cleavage site of Alzheimer amyloid precursor in neuronal PC-12 cells. Neurosci. Lett. 128, 126–128.
Arribas J. and Massague J. (1995) Transforming growth factor-alpha and beta-amyloid precursor protein share a secretory mechanism. J. Cell Biol. 128, 433–441.
Borchelt D. R., Thinakaran G., Eckman C. B., Lee M. K., Davenport F., Ratovitsky T., et al. (1996) Familial Alzheimer’s disease-linked presenilin 1 variants elevate Abeta1-42/1-40 ratio in vitro and in vivo. Neuron 17, 1005–1013.
Buxbaum J. D., Gandy S. E., Cicchetti P., Ehrlich M. E., Czernik A. J., Fracasso R. P., et al. (1990) Processing of Alzheimer beta/A4 amyloid precursor protein: modulation by agents that regulate protein phosphorylation. Proc. Natl. Acad. Sci. U. S. A. 87, 6003–6006.
Buxbaum J. D., Liu K. N., Luo Y., Slack J. L., Stocking K. L., Peschon J. J., et al. (1998) Evidence that tumo necrosis factor alpha converting enzyme is involved in regulated alpha-secretase cleavage of the Alzheimer amyloid protein precursor. J. Biol. Chem. 273, 27765–27767.
Checler F. (1995) Processing of the beta-amyloid precursor protein and its regulation in Alzheimer’s disease. J. Neurochem. 65, 1431–1444.
Esch F. S., Keim P. S., Beattie E. C., Blacher R. W., Culwell A. R., Oltersdorf T., et al. (1990) Cleavage of amyloid beta peptide during constitutive processing of its precursor. Science 248, 1122–1124.
Farzan M., Schnitzler C. E., Vasilieva N., Leung D., and Choe H. (2000) BACE2, a beta-secretase homolog, cleaves at the beta site and within the amyloid-beta region of the amyloid-beta precursor protein. Proc. Natl. Acad. Sci. U. S. A. 97, 9712–9717.
Fassbender K., Simons M., Bergmann C., Stroick M., Lutjohann D., Keller P., et al. (2001) Simvastatin strongly reduces levels of Alzheimer’s disease beta-amyloid peptides Abeta 42 and Abeta 40 in vitro and in vivo. Proc. Natl. Acad. Sci. U. S. A. 98, 5856–5861.
Gabuzda D., Busciglio J., and Yankner B. A. (1993) Inhibition of beta-amyloid production by activation of protein kinase C. J. Neurochem. 61, 2326–2329.
Hardy J. and Selkoe D. J. (2002) The amyloid hypothesis of Alzheimer’s disease: progress and problems on the road to therapeutics. Science 297, 353–356.
Hartmann D., de Strooper B., Serneels L., Craessaerts K., Herreman A., Annaert W., et al. (2002) The disintegrin/metalloprotease ADAM 10 is essential for Notch signalling but not for alpha-secretase activity in fibroblasts. Hum. Mol. Genet. 11, 2615–2624.
Hartmann D., Tournoy J., Saftig P., Annaert W., and De Strooper B. (2001) Implication of APP secretases in notch signaling. J. Mol. Neurosci. 17, 171–181.
Hooper N. M. and Turner A. J. (2002) The search for alpha-secretase and its potential as a therapeutic approach to Alzheimer s disease. Curr. Med. Chem. 9, 1107–1119.
Koike H., Tomioka S., Sorimachi H., Saido T. C., Maruyama K., Okuyama A., et al. (1999) Membrane-anchored metalloprotease MDC9 has an alpha-secretase activity responsible for processing the amyloid precursor protein. Biochem. J. 343(Pt, 2), 371–375.
Lahiri D. K., Farlow M. R., and Sambamurti K. (1998) The secretion of amyloid beta-peptides is inhibited in the tacrine-treated human neuroblastoma cells. Brain Res. Mol. Brain Res. 62, 131–140.
Lammich S., Kojro E., Postina R., Gilbert S., Pfeiffer R., Jasionowski M., et al. (1999) Constitutive and regulated alpha-secretase cleavage of Alzheimer’s amyloid precursor protein by a disintegrin metalloprotease. Proc. Natl. Acad. Sci. U. S. A. 96, 3922–3927.
Marlow L., Cain M., and Sambamurti K. (2003) Regulation of beta-secretase processing of the Alzheimer’s amyloid protein precursor (APP). J. Mol. Neurosci. 20, 233–239.
Mills J. and Reiner P. B. (1999) Regulation of amyloid precursor protein cleavage. J. Neurochem. 72, 443–460.
Nitsch R. M., Farber S. A., Growdon J. H., and Wurtman R. J. (1993) Release of amyloid beta-protein precursor derivatives stimulated by activation of muscarinic acetyl choline receptors. Science 258, 304–307.
Pinnix I., Musunuru U, Tun H., Sridharan A., Golde T., Eckman C., et al. (2001) A novel gamma-secretase assay based on detection of the putative C-terminal fragment-gamma of amyloid beta protein precursor. J. Biol. Chem. 276, 481–487.
Refolo L. M., Malester B., LaFrancois J., Bryant-Thomas T., Wang R., Tint G. S., et al. (2000) Hypercholesterolemia accelerates the Alzheimer’s amyloid pathology in a transgenic mouse model. Neurobiol. Dis. 7, 321–331.
Refolo L. M., Pappolla M. A., LaFrancois J., Malester B., Schmidt S. D., Thomas-Bryant T, et al. (2001) A cholesterol-lowering drug reduces beta-amyloid pathology in a transgenic mouse model of Alzheimer’s disease. Neurobiol. Dis. 8, 890–899.
Refolo L. M., Salton S. R., Anderson J. P., Mehta P., and Robakis N. K. (1989) Nerve and epidermal growth factors induce the release of the Alzheimer amyloid precursor from PC 12 cell cultures. Biochem. Biophys. Res. Commun. 164, 664–670.
Sambamurti K., Greig N. H., and Lahiri D. K. (2002a) Advances in the cellular and molecular biology of the beta-amyloid protein in Alzheimer’s disease. Neuromol. Med. 1, 1–31.
Sambamurti K., Hardy J., Refolo L. M., and Lahiri D. K. (2002b) Targeting APP metabolism for the treatment of Alzheimer’s disease. Drug Dev. Res. 56, 211–227.
Sambamurti K., Sevlever D., Koothan T., Refolo L. M., Pinnix I., Gandhi S., et al. (1999) Glycosylphosphatidylinositol-anchored proteins play an important role in the biogenesis of the Alzheimer’s amyloid beta-protein. J. Biol. Chem. 274, 26810–26814.
Scheuner D., Eckman C., Jensen M., Song X., Citron M., Suzuki N., et al. (1996) Secreted amyloid beta-protein similar to that in the senile plaques of Alzheimer’s disease is increased in vivo by the presenilin 1 and 2 and APP mutations linked to familial Alzheimer’s disease. Nat. Med. 2, 864–870.
Seubert P., Oltersdorf T., Lee M. G., Barbour R., Blomquist C., Davis D. L., et al. (1993) Secretion of beta-amyloid precursor protein cleaved at the amino terminus of the beta-amyloid peptide. Nature 361, 260–263.
Simons M., Keller P., De Strooper B., Beyreuther K., Dotti C. G., and Simons K. (1998) Cholesterol depletion inhibits the generation of beta-amyloid in hippocampal neurons. Proc. Natl. Acad. Sci. U. S. A. 95, 6460–6464.
Sinha S., Anderson J. P., Barbour R., Basi G. S., Caccavello R., Davis D., et al. (1999) Purification and cloning of amyloid precursor protein beta-secretase from human brain. Nature 402, 537–540.
Slack B. E., Ma L. K., and Seah C. C. (2001) Constitutive shedding of the amyloid precursor protein ectodomain is up-regulated by tumour necrosis factor-alpha converting enzyme. Biochem. J. 357, 787–794.
Tun H., Marlow L., Pinnix I., Kinsey R., and Sambamurti K. (2002) Lipid rafts play an important role in A beta biogenesis by regulating beta-secretase pathway. J. Mol. Neurosci. 19, 31–35.
Wolozin B. (2003) Cyp46 (24S-cholesterol hydroxylase): a genetic risk factor for Alzheimer disease. Arch. Neurol. 60, 16–18.
Xu H., Greengard P., and Gandy S. (1995) Regulated formation of Golgi secretory vesicles containing Alzheimer beta-amyloid precursor protein. J. Biol. Chem. 270, 23243–23245.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gandhi, S., Refolo, L.M. & Sambamurti, K. Amyloid precursor protein compartmentalization restricts β-amyloid production. J Mol Neurosci 24, 137–143 (2004). https://doi.org/10.1385/JMN:24:1:137
Issue Date:
DOI: https://doi.org/10.1385/JMN:24:1:137