Abstract
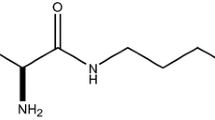
A Phase I, double-blind, placebo-controlled, single-dose, escalation study of the purine derivative, AIT-082 (Neotrofin™, NeoTherapeutics, Inc.) was conducted in healthy elderly volunteers. This trial was designed to evaluate single-dose safety, tolerability, and pharmacokinetics. Potential cognitive domains that might benefit from AIT-082 were preliminarily investigated. AIT-082 is currently being developed as a potential treatment for Alzheimer’s disease (AD). Preclinical studies indicate that AIT-082 has memory-enhancing properties, stimulates neuritogenesis, and upregulates neurotrophic factors. Subjects received a single oral dose of AIT-082 or placebo on a weekly basis for 5 wk. All patients received a placebo dose at baseline. Six subjects received increasing doses of AIT-082 over the next 4 wk at doses of 0.6, 2.0, 6.0, and 20.0 mg of AIT-082 per kilogram of body weight. Two subjects received placebo throughout the trial. Nine subjects were recruited. One subject was withdrawn after the third treatment visit owing to poor venous access. There were no serious adverse events. The drug was well-tolerated. The time to peak drug concentration was approx 85 min with an elimination half-life of approx 17.6 h. Performance on the Number Comparison, Symbol Digit, and Trails A tests improved with AIT-082 dosing compared to baseline (placebo). In conclusion, AIT-082 was rapidly absorbed by the oral route with a half-life suitable for once daily dosing. No problems with tolerability or safety were demonstrated.
Similar content being viewed by others
References
Army Individual Test Battery (1944) Manual of Directions and Scoring. War Department, Adjutant General’s Office, Washington, DC.
Benton A.L., Varney N.R., and Hamsher K.D. (1978) Visuospatial judgment. A clinical test. Arch Neurol. 35, 364–367.
Cooper J. A., Sagar H. J., Jordan N., Harvey N. S., and Sullivan E. V. (1991) Cognitive impairment in early, untreated Parkinson’s disease and its relationship to motor disability. Brain 114, 2095–2122.
Craik F. I. (1990) Changes in memory with normal aging: a functional view. Adv. Neurol. 51, 201–205.
Folstein M. F., Folstein S. E., and McHugh P. R. (1975) “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatric Res. 12, 189–198.
Glasky A., Glasky M. S., Ritzmann R., and Rathbone M. (1997) AIT-082, a novel purine derivative with neuroregenerative properties. Exp. Opin. Invest. Drugs 6, 1413–1417.
Glasky A. J., Melchior C. L., Pirzadeh B., Heydari N., and Ritzmann R. F. (1994) Effect of AIT-082, a purine analog, on working memory in normal and aged mice. Pharmacol. Biochem. Behav. 47, 325–329.
Glasky A. J., Ritzmann R. F., Rathbone M. P., Middlemiss P. J., and Crocker C. (1996) Neurotrophins, growth factors and mimetic agents as neuroprotectors in the treatment of Alzheimer’s disease, in Alzheimer Disease: From Molecular Biology to Therapy (Becker, R. and Giacobini, E. eds.), Birkhauser Boston, Boston, pp. 119–124.
Glasky M. S., Conner J. M., and Tuszynski M. H. (2001) Oral Administration of AIT-082 Stimulates BDNF and NGF Expression in Vivo in Rats, Society for Neuroscience’s 31st Annual Meeting, San Diego, CA, 137.10.
Hachinski V. C., Iliff L. D., Zilhka E., Du Boulay G. H., McAllister V. L., Marshall J., et al. (1975) Cerebral blood flow in dementia. Arch. Neurol. 32, 632–637.
Middlemiss P. J., Glasky A. J., Rathbone M. P., Werstuik E., Hindley S., and Gysbers J. (1995) AIT-082, a unique purine derivative, enhances nerve growth factor mediated neurite outgrowth from PC12 cells. Neurosci. Lett. 199, 131–134.
Peterson L. R. and Peterson M. J. (1959) Short-term retention of individual verbal items. J. Exp. Psychol. 58, 193–198.
Ramirez J. J., Glasky A.J., Parakh T., George N.M., Becton A. (1998) AIT-082 accelerates septodentate sprouting after unilateral entorhinal cortex lesion in rats, 28th Annual Meeting of the Society for Neuroscience, Los Angeles, CA, 770.6.
Randolph C., Braun A. R., Goldberg T. E., and Chase T. N. (1993) Semantic fluency in Alzheimer’s, Parkinson’s, and Huntington’s disease: Dissociation of storage and retrieval failures. Neuropsychology 7, 82–88.
Rathbone M. P., Middlemiss P. J., Gysbers J., Diamond J., Holmes M., Pertens E., et al. (1998) Physiology and pharmacology of natural and synthetic nonadenine-based purines in the nervous system. Drug Dev. Res. 45, 356–372.
Rey A. (1964) L’examen clinique en psychologie. Presses Universitaires de France, Paris.
Salthouse T. A. and Coon V. E. (1993) Influence of task-specific processing speed on age differences in memory. J. Gerontol. Psycholog. Sci. 48, 245–255.
Smith A. (1984) Symbol Digit Modalities Test Manual — Revised. Western Psychological Services, Los Angeles, CA.
Taylor E. M., Wilson, R.A., Chen, M., and Dulak, K.R. (2001) AIT-082 Stimulated the Proliferation of Neural Stem Cells in the Dentate Gyrus of Adult Mice, Society for Neuroscience’s 31st Annual Meeting, San Diego, CA, 894.3.
Wang X., Foreman, M. M., and Maines, M. D. (2001) Neotrofin Selectively Increases Heme Oxygenase-1 in Neurons, Society for Neuroscience’s 31st Annual Meeting, San Diego, CA, 549.10.
Wechsler D. (1987) WMS-R Wechsler Memory Scale — Revised Manual. The Psychological Corporation, Harcourt Brace Jovanovich, Inc., New York, NY.
Author information
Authors and Affiliations
Consortia
Corresponding author
Rights and permissions
About this article
Cite this article
Grundman, M., Farlow, M., Peavy, G. et al. A phase I study of AIT-082 in healthy elderly volunteers. J Mol Neurosci 18, 283–293 (2002). https://doi.org/10.1385/JMN:18:3:283
Issue Date:
DOI: https://doi.org/10.1385/JMN:18:3:283