Abstract
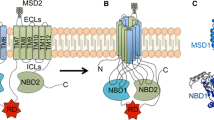
The autosomal recessive disease cystic fibrosis (CF) is caused by mutations in the gene coding for the CF transmembrane conductance regulator (CFTR) protein, a cAMP-activated chloride channel expressed at the apical membrane of epithelial cells. Although about 1000 different mutations have been identified, most CF patients carry the F508del mutation in at least one CFTR allele. F508del-CFTR is synthesized but is substantially retained as a core-glycosylated intermediate in the endoplasmic reticulum (ER), probably because of misfolding. The mutant protein is recognized by molecular chaperones involved in cellular quality control and rapidly targeted for proteasomal degradation. Although maturation of wild-type CFTR (wt-CFTR) is also inefficient at varying levels, depending on the cell type, there is increasing evidence that the two proteins acquire at least partially distinct conformations. However, the structural cues responsible for the conformational differences and the cellular mechanisms determining the endpoint for each conformer remain largely unclear. Some knowledge is emerging on CFTR membrane folding and on the role of this process of molecular chaperones, such as Hsp70/Hdj-1 and calnexin. The key players in the final decision on whether the protein will enter the secretory pathway or be degraded, however, remain unidentified. Here we discuss existing data on the interaction of molecular chaperones and CFTR, as well as their putative role on the degradation and processing of this polytopic membrane protein.
Similar content being viewed by others
References
Bannykh S. I., Bannykh G. I., Fish K. N., Moyer B. D., Riordan J. R., and Balch W.E. (2000) Traffic pattern of cystic fibrosis transmembrane regulator through the early exocytic pathway. Traffic 1, 852–870.
Bebok Z., Mazzochi C., King S. A, Hong J. S., and Sorscher E. J. (1998) The mechanism underlying cystic fibrosis transmembrane conductance regulator transport from the endoplasmic reticulum to the proteasome includes Sec61beta and a cytosolic, deglycosylated intermediary. J. Biol. Chem. 273, 29873–29878.
Bradbury N. A., Jilling T., Berta G., Sorcher E. J., Bridges R. J., and Kirk K. L. (1992) Regulation of plasma membrane recycling by CFTR. Science 256, 530–532.
Cabral C. M., Liu Y., and Sifers R. N. (2001) Dissecting glycoprotein quality control in the secretory pathway. Trends Biochem. Sci. 26, 619–624.
Collins F. S. (1992) Cystic fibrosis: molecular biology and therapeutic implications. Science 256, 774–779.
Dalemans W., Barbry P., Champigny G., Jallat S., Dott K., Dreyer D., et al. (1991) Altered chloride ion channel kinetics associated with the delta F508 cystic fibrosis mutation. Nature 354, 526–528.
Denning G. M., Ostedgaard L. S., and Welsh M. J. (1992) Abnormal localization of cystic fibrosis transmembrane conductance regulator in primary cultures of cystic fibrosis airway epithelia. J. Cell. Biol. 118, 551–559.
Ellgaard L. and Helenius A. (2001) ER quality control: towards an understanding at the molecular level. Curr. Opin. Cell. Biol. 13, 431–437.
Ellgaard L., Molinari M., and Helenius A. (1999) Setting the standards: quality control in the secretory pathway. Science 286, 1882–1888.
Farinha C. M. (2002) “Processing and intracellular trafficking of wild-type and mutant CFTR,” Ph.D. thesis, Faculty of Sciences, University of Lisboa, Lisboa, Portugal.
Farinha C. M., Nogueira P., Mendes F., Penque D., and Amaral M. D. (2002) The human DnaJ homologue (Hdj)-1/heat-shock protein (Hsp) 40 co-chaperone is required for the in vivo stabilization of the cystic fibrosis transmembrane conductance regulator by Hsp70. Biochem. J. 366, 797–806.
Fu L. and Sztul E. (2003) Traffic-independent function of the Sar1p/COPII machinery in proteasomal sorting of the cystic fibrosis transmembrane conductance regulator. J. Cell. Biol. 160, 157–163.
Gilbert A., Jadot M., Leontieva E., Wattiaux-De Coninck S., and Wattiaux R. (1998) Delta F508 CFTR localizes in the endoplasmic reticulum-Golgi intermediate compartment in cystic fibrosis cells. Exp. Cell. Res. 242, 144–152.
Hosokawa N., Wada I., Hasegawa K., Yorihuzi T., Tremblay L. O., Herscovics A., and Nagata K. (2001) A novel ER alpha-mannosidase-like protein accelerates ER-associated degradation. EMBO Rep. 2, 415–422.
Jakob C. A., Bodmer D., Spirig U., Battig P., Marcil A., Dignard D., et al. (2001) Htm1p, a mannosidase-like protein, is involved in glycoprotein degradation in yeast. EMBO Rep. 2, 423–430.
Jakob C. A., Burda P., Roth J., and Aebi M. (1998) Degradation of misfolded endoplasmic reticulum glycoproteins in Saccharomyces cerevisae is determined by a specific oligosaccharide structure. J. Cell Biol. 142, 1223–1233.
Jensen T. J., Loo M. A., Pind S., Williams D. B., Goldberg A. L., and Riordan J. R. (1995) Multiple proteolytic systems, including the proteasome, contribute to CFTR processing. Cell 83, 129–135.
Johnston J. A., Ward C. L., and Kopito R. R. (1998) Aggresomes: a cellular response to misfolded proteins. J. Cell Biol. 143, 1883–1898.
Kälin N., Claass A., Sommer M., Puchelle E., and Tümmler B. (1999) DeltaF508 CFTR protein expression in tissues from patients with cystic fibrosis. J. Clin. Invest. 103, 1379–1389.
Kamhi-Nesher S., Shenkman M., Tolchinsky S., Fromm S. V., Ehrlich R., and Lederkremer G. Z. (2001) A novel quality control compartment derived from the endoplasmic reticulum. Mol. Biol. Cell. 12, 1711–1723.
Klein I., Sarkadi B., and Varadi A. (1999) An inventory of the human ABC proteins. Biochim. Biophys. Acta 1461, 237–262 (see also www.humanabc.org).
Loo M. A., Jensen T. J., Cui L., Hou Y., Chang X. B., and Riordan, J. R. (1998) Perturbation of Hsp90 interaction with nascent CFTR prevents its maturation and accelerates its degradation by the proteasome. EMBO J. 17, 6879–6887.
Lu Y., Xiong X., Helm A., Kimani K., Bragin A., and Skach W. R. (1998) Co- and posttranslational translocation mechanisms direct cystic fibrosis transmembrane conductance regulator N terminus transmembrane assembly. J. Biol. Chem. 273, 568–576.
Lukacs G. L., Chang X. B., Bear C., Kartner N., Mohamed A., Riordan J. R., and Grinstein S. (1993) The delta F508 mutation decreases the stability of cystic fibrosis transmembrane conductance regulator in the plasma membrane. Determination of functional half-lives on transfected cells. J. Biol. Chem. 268, 21592–21598.
Meacham G. C., Lu Z., King S., Sorscher E., Tousson A., and Cyr, D. M. (1999) The Hdj-2/Hsc70 chaperone pair facilitates early steps in CFTR biogenesis. EMBO J. 18, 1492–1505.
Meacham G. C., Patterson C., Zhang W., Younger J. M., and Cyr D. M. (2001) The Hsc70 co-chaperone CHIP targets immature CFTR for proteasomal degradation. Nat. Cell Biol. 3, 100–105.
Nehls S., Snapp E. L., Cole N. B., Zaal K. J. M., Kenworthy A. K., Roberts T. H., et al. (2000) Dynamics and retention of misfolded proteins in native ER membranes. Nat. Cell Biol. 2, 288–295.
Parodi A. J. (2000) Protein glucosylation and its role in protein folding. Annu. Rev. Biochem. 69, 69–93.
Pasyk E. A. and Foskett J. K. (1995) Mutant (delta F508) cystic fibrosis transmembrane conductance regulator Cl- channel is functional when retained in endoplasmic reticulum of mammalian cells. J. Biol. Chem. 270, 12347–12350.
Penque D., Mendes F., Beck S., Farinha C., Pacheco P., Nogeuira P., et al. (2000) Cystic fibrosis F508del patients have apically localized CFTR in a reduced number of airway cells. Lab. Invest. 80, 857–868.
Pind S., Riordan J. R., and Williams D. B. (1994) Participation of the endoplasmic reticulum chaperone calnexin (p88, IP90) in the biogenesis of the cystic fibrosis transmembrane conductance regulator. J. Biol. Chem. 269, 12784–12788.
Ramalho A. S., Beck S., Meyer M., Penque D., Cutting G., and Amaral M. D. (2002) Five percent of normal CFTR mRNA ameliorates the severity of pulmonary disease in cystic fibrosis. Am. J. Respir. Cell. Mol. Biol. 27, 619–627.
Riordan, J. R. (1999) Cystic fibrosis as a diseae of misprocessing of the cystic fibrosis transmembrane conductance regulator glycoprotein. Am. J. Hum. Genet. 64, 1499–1504.
Riordan J. R. (1993) The cystic fibrosis transmembrane conductance regulator. Annu. Rev. Physiol. 55, 609–630.
Riordan J. R., Rommens J. M., Kerem B., Alon N., Rozmahel R., Grzelczak Z., et al. (1989) Identification of the cystic fibrosis gene: cloning and characterization of complementary DNA. Science 245, 1066–1073.
Sharma M., Benharouga M., Hu W., and Lukacs G. L. (2001) Conformational and temperature-sensitive stability defects of the delta F508 cystic fibrosis transmembrane conductance regulator in post-endoplasmic reticulum compartments. J. Biol. Chem. 276, 8942–8950.
Sheppard D. N. and Welsh M. J. (1999) Structure and function of the CFTR chloride channel. Physiol. Rev. 79, S23-S45.
Skach W. R. (2000) Defects in processing and trafficking of the cystic fibrosis transmembrane conductance regulator. Kidney Int. 57, 825–831.
Vashist S., Kim W., Belden W. J., Spear E. D., Barlowe C., and Ng D. T. W. (2001) Distinct retrieval and retention mechanisms are required for the quality control of endoplasmic reticulum protein folding. J. Cell Biol. 155, 355–367.
Wang F., Zeltwanger S., Hu S., and Hwang T. C. (2000) Deletion of phenylalanine 508 causes attenuated phosphorylation-dependent activation of CFTR chloride channels J. Physiol. 524, 637–648.
Ward C. L., Omura S., and Kopito R. R. (1995) Degradation of CFTR by the ubiquitin-proteasome pathway. Cell 83, 121–127.
Wei X., Eisman R., Xu J., Harsch A. D., Mulberg A. E., Bevins C. L., et al. (1996) Turnover of the cystic fibrosis transmembrane conductance regulator (CFTR): slow degradation of wild-type and delta F508 CFTR in surface membrane preparations of immortalized airway epithelial cells. J. Cell. Physiol. 168, 373–384.
Yang Y., Janich S., Cohn J. A., and Wilson J. M. (1993) The common variant of cystic fibrosis transmembrane conductance regulator is recognized by hsp70 and degraded in a pre-Golgi nonlysosomal compartment. Proc. Natl. Acad. Sci. USA 90, 9480–9484.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Amaral, M.D. CFTR and chaperones. J Mol Neurosci 23, 41–48 (2004). https://doi.org/10.1385/JMN:23:1-2:041
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/JMN:23:1-2:041