Abstract
Background
Transient receptor potential vanilloid 2 (TRPV2) is a member of the TRP superfamily of non-specific cation channels with functionally diverse roles. We herein investigated the effects of TRPV2 on the expression of programmed cell death-ligand 1 (PD-L1) and its binding ability to programmed cell death-1 (PD-1) in gastric cancer (GC).
Methods
Knockdown (KD) experiments were performed on human GC cell lines using TRPV2 small-interfering RNA. The surface expression of PD-L1 and its binding ability to PD-1 were analyzed by flow cytometry. Eighty primary tissue samples were assessed by immunohistochemistry (IHC), and the relationships between IHC results, clinicopathological factors, and patient prognosis were analyzed. The molecular mechanisms underlying the effects of TRPV2 on the intracellular ion environment were also investigated.
Results
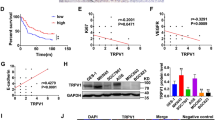
TRPV2-KD decreased the expression level of PD-L1 in NUGC4 and MKN7 cells, thereby inhibiting its binding to PD-1. A survival analysis revealed that 5-year overall survival rates were significantly lower in the TRPV2 high expression and PD-L1-positive groups. In IHC multivariate analysis of GC patients, high TRPV2 expression was identified as an independent prognostic factor. Furthermore, a positive correlation was observed between the expression of TRPV2 and PD-L1. An immunofluorescence analysis showed that TRPV2-KD decreased the intracellular concentration of calcium ([Ca2+]i). Treatment with ionomycin/PMA (phorbol 12-myristate 13-acetate), which increased [Ca2+]i, upregulated the protein expression of PD-L1 and promoted its binding to PD-1.
Conclusions
The surface expression of PD-L1 and its binding ability to PD-1 in GC were regulated by TRPV2 through [Ca2+]i, indicating the potential of TRPV2 as a biomarker and target of immune checkpoint blockage for GC.






Similar content being viewed by others
References
Smyth EC, Nilsson M, Grabsch HI, et al. Gastric cancer. Lancet. 2020;396(10251):635–48.
Catalano V, Labianca R, Beretta GD, et al. Gastric cancer. Crit Rev Oncol Hematol. 2009;71(2):127–64.
Bray F, Ferlay J, Soerjomataram I, et al. Global cancer statistics 2018 GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424.
Kang YK, Boku N, Satoh T, et al. Nivolumab in patients with advanced gastric or gastro-oesophageal junction cancer refractory to, or intolerant of, at least two previous chemotherapy regimens (ONO-4538-12, ATTRACTION-2): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2017;390(10111):2461–71.
Janjigian YY, Shitara K, Moehler M, et al. First-line nivolumab plus chemotherapy versus chemotherapy alone for advanced gastric, gastro-oesophageal junction, and oesophageal adenocarcinoma (CheckMate 649): a randomised, open-label, phase 3 trial. Lancet. 2021;398(10294):27–40.
Kang YK, Chen LT, Ryu MH, et al. Nivolumab plus chemotherapy versus placebo plus chemotherapy in patients with HER2-negative, untreated, unresectable advanced or recurrent gastric or gastro-oesophageal junction cancer (ATTRACTION-4): a randomised, multicentre, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2022;23(2):234–47.
Keir ME, Butte MJ, Freeman GJ, et al. PD-1 and its ligands in tolerance and immunity. Annu Rev Immunol. 2008;26:677–704.
Kulangara K, Guerrero L, Posch A, et al. Investigation of PD-L1 expression and response to pembrolizumab (pembro) in gastric cancer (GC) and cervical cancer (CC) using combined positive score (CPS) and tumor proportion score (TPS). J Clin Oncol. 2018;36(15 Suppl):4065.
Lei M, Siemers N, Pandya D, et al. Association of PD-L1 combined positive score and immune gene signatures with efficacy of nivolumab (NIVO) ± ipilimumab (IPI) in patients with metastatic gastroesophageal cancer (mGEC). Cancer Res. 2019;79(13 Suppl):2673.
Shitara K, Ozgurolu M, Bang YJ, et al. Pembrolizumab versus paclitaxel for previously treated, advanced gastric or gastrooesophageal junction cancer (KEYNOTE-061): a randomised, open-label, controlled, phase 3 trial. Lancet. 2018;392(10142):123–33.
Shiozaki A, Ichikawa D, Otsuji E, et al. Cellular physiological approach for treatment of gastric cancer. World J Gastroenterol. 2014;20(33):11560–6.
Shiozaki A, Marunaka Y, Otsuji E. Roles of ion and water channels in the cell death and survival of upper gastrointestinal tract cancers. Front Cell Dev Biol. 2021;9:616933.
Kato S, Shiozaki A, Kudou M, et al. TRPV2 promotes cell migration and invasion in gastric cancer via the transforming growth factor-β signaling pathway. Ann Surg Oncol. 2022;29(5):2944–56.
Inoue H, Shiozaki A, Kosuga T, et al. Functions and clinical significance of CACNA2D1 in gastric cancer. Ann Surg Oncol. 2022. https://doi.org/10.1245/s10434-022-11752-5.
Katsurahara K, Shiozaki A, Kosuga T, et al. ANO9 regulates PD-L2 expression and binding ability to PD-1 in gastric cancer. Cancer Sci. 2021;112(3):1026–37.
Kurashima K, Shiozaki A, Kudou M, et al. LRRC8A influences the growth of gastric cancer cells via the p53 signaling pathway. Gastric Cancer. 2021;24(5):1063–75.
Mitsuda M, Shiozaki A, Kudou M, et al. Functional analysis and clinical significance of chloride channel 2 expression in esophageal squamous cell carcinoma. Ann Surg Oncol. 2021;28(9):5384–97.
Matsumoto Y, Shiozaki A, Kosuga T, et al. Expression and role of CFTR in human esophageal squamous cell carcinoma. Ann Surg Oncol. 2021;28(11):6424–36.
Fukami T, Shiozaki A, Kosuga T, et al. Anoctamin 5 regulates the cell cycle and affects prognosis in gastric cancer. World J Gastroenterol. 2022;28(32):4649–67.
Nakamura K, Shiozaki A, Kosuga T, et al. The expression of the alpha1 subunit of Na+/K+-ATPase is related to tumor development and clinical outcomes in gastric cancer. Gastric Cancer. 2021;24(6):1278–92.
Shiozaki A, Katsurahara K, Kudou M, et al. Amlodipine and verapamil, voltage-gated Ca2+ channel inhibitors, suppressed the growth of gastric cancer stem cells. Ann Surg Oncol. 2021;28(9):5400–11.
Sharma P, Allison JP. Immune checkpoint targeting in cancer therapy: toward combination strategies with curative potential. Cell. 2015;161(2):205–14.
Yuan J, Zhang J, Zhu Y, et al. Programmed death-ligand-1 expression in advanced gastric cancer detected with RNA in situ hybridization and its clinical significance. Oncotarget. 2016;7(26):39671–9.
Kawazoe A, Kuwata T, Kuboiki Y, et al. Clinicopathological features of programed death ligand 1 expression with tumor-infiltrating lymphocyte, mismatch repair, and Epstein-Barr virus status in a large cohort of gastric cancer patients. Gastric Cancer. 2017;20(3):407–15.
Zhang M, Dong Y, Liu H, et al. The clinicopathological and prognostic significance of PD-L1 expression in gastric cancer: a meta-analysis of 10 studies with 1,901 patients. Sci Rep. 2016;6:1–9.
Lan X, Zhao J, Song C, et al. TRPM8 facilitates proliferation and immune evasion of esophageal cancer cells. Biosci Rep. 2019;39(10):BSR20191878.
Liu BL, Cao QL, Zhao X, et al. Inhibition of TRPV1 by SHP-1 in nociceptive primary sensory neurons is critical in PD-L1 analgesia. JCI Insight. 2020;5(20):e137386.
Peralvarez-Marin A, Donate-Macian P, Gaudet R. What do we know about the transient receptor potential vanilloid 2 (TRPV2) ion channel? FEBS J. 2013;280(21):5471–87.
Zhou K, Zhang SS, Yan Y, et al. Overexpression of transient receptor potential vanilloid 2 is associated with poor prognosis in patients with esophageal squamous cell carcinoma. Med Oncol. 2014;31(7):17.
Kudou M, Shiozaki A, Yamazato Y, et al. The expression and role of TRPV2 in esophageal squamous cell carcinoma. Sci Rep. 2019;9(1):16055.
Liu G, Xie C, Sun F, et al. Clinical significance of transient receptor potential vanilloid 2 expression in human hepatocellular carcinoma. Cancer Genet Cytogenet. 2010;197(1):54–9.
Yamada T, Ueda T, Shibata Y, et al. TRPV2 activation induces apoptotic cell death in human T24 bladder cancer cells: a potential therapeutic target for bladder cancer. Urology. 2010;76(2):509.e1-7.
Monet M, Lehen’kyi V, Gackiere F, et al. Role of cationic channel TRPV2 in promoting prostate cancer migration and progression to androgen resistance. Cancer Res. 2010;70(3):1225–35.
Shiozaki A, Kudou M, Ichikawa D, et al. Esophageal cancer stem cells are suppressed by tranilast, a TRPV2 channel inhibitor. J Gastroenterol. 2018;53(2):197–207.
Darakhshan S, Pour AB. Tranilast: a review of its therapeutic applications. Pharmacol Res. 2015;91:15–28.
Ando N, Kato H, Igaki H, et al. A randomized trial comparing postoperative adjuvant chemotherapy with cisplatin and 5-fluorouracil versus preoperative chemotherapy for localized advanced squamous cell carcinoma of the thoracic esophagus (JCOG9907). Ann Surg Oncol. 2012;19(1):68–74.
Shiozaki A, Kudou M, Fujiwara H, et al. Clinical safety and efficacy of neoadjuvant combination chemotherapy of tranilast in advanced esophageal squamous cell carcinoma: phase I/II study (TNAC). Medicine (Baltimore). 2020;99(50):e23633.
Miyazaki H, Shiozaki A, Niisato N, et al. Chloride ions control the G1/S cell-cycle checkpoint by regulating the expression of p21 through a p53-independent pathway in human gastric cancer cells. Biophys Res Commun. 2008;366(2):506–12.
Ohsawa R, Miyazaki H, Niisato N, et al. Intracellular chloride regulates cell proliferation through the activation of stress-activated protein kinases in MKN28 human gastric cancer cells. J Cell Physiol. 2010;223(3):764–70.
Shiozaki A, Otsuji E, Marunaka Y. Intracellular chloride regulates the G(1)/S cell cycle progression in gastric cancer cells. World J Gastrointest Oncol. 2011;3(8):119–22.
Alam MR, Rahman MM, Li A. The link between intracellular calcium signaling and exosomal PD-L1 in cancer progression and immunotherapy. Genes Diseases. 2023. https://doi.org/10.1016/j.gendis.2023.01.026.
Savina A, Fader CM, Damiani MT, et al. Rab11 promotes docking and fusion of multivesicular bodies in a calcium dependent manner. Traffic. 2005;6(2):131–43.
Poggio M, Hu T, Pai CC, et al. Suppression of exosomal PD-L1 induces systemic anti-tumor immunity and memory. Cell. 2019;177(2):414–27.
Acknowledgments
The present study was supported by Grants-in-Aid for Scientific Research (C) [20K09016, 20K09084, 22K08832, and 21K08689] and Grants-in-Aid for Young Scientists [20K17659, 21K16456, and 22K16518] from the Japan Society for the Promotion of Science.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Atsushi Shiozaki, Tomoyuki Fukami, Hiroki Shimizu, Toshiyuki Kosuga, Michihiro Kudou, Kenichi Takemoto, Keita Katsurahara, Keiji Nishibeppu, Takuma Ohashi, Tomohiro Arita, Hirotaka Konishi, Shuhei Komatsu, Takeshi Kubota, Hitoshi Fujiwara, and Eigo Otsuji have no conflicts of interest or financial ties to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.




Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shiozaki, A., Fukami, T., Shimizu, H. et al. Effects of TRPV2 on the Expression of PD-L1 and Its Binding Ability to PD-1 in Gastric Cancer. Ann Surg Oncol 30, 8704–8716 (2023). https://doi.org/10.1245/s10434-023-14084-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-023-14084-0