Abstract
Background
The role of systemic therapy for Stage IA pancreatic ductal adenocarcinoma (PDAC) is unclear. The aim of our study was to evaluate the impact of adjuvant chemotherapy (AC) on survival in patients with early stage disease.
Methods
The National Cancer Database was queried from 2006 to 2017 for resected pT1N0M0 (Stage 1A) PDAC. Exclusion criteria included neoadjuvant therapy, radiation, or those who suffered a 90-day mortality.
Results
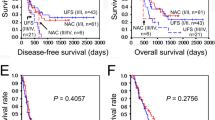
Of the 1526 patients included in the study, 42.2% received AC and 57.8% underwent surgery alone. Patients who received AC were younger, had fewer comorbidities, and were more likely to have private insurance, compared with those treated with surgery alone. Patients who received AC had longer median overall survival (OS) compared with those who underwent surgery alone (105.7 months vs 72.0 months, p < 0.01). Subset analyses based on individual “good” prognostic features (size ≤ 1.0 cm, lymphovascular invasion negative, well/moderately differentiated, margin negative resection) demonstrated improved OS with AC. Following propensity score matching based on key clinicopathologic features, AC remained associated with improved median OS (83.7 months vs 59.8 months, p < 0.01). However, in the cohort with body/tail tumors (101.2 months vs 95.0 months, p = 0.19) and those with all “good” prognostic features (95.9 months vs 90.6 months, p = 0.15), AC was not associated with improved survival.
Conclusions
In resected, Stage IA PDAC, AC is associated with improved overall survival in the vast majority of patients; however, in select cohorts the role of AC is unclear. Further study is needed to tailor treatment to individual patients with PDAC.




Similar content being viewed by others
References
SEER Cancer Stat Facts: Pancreatic Cancer. National Cancer Institute. Bethesda, MD, https://seer.cancer.gov/statfacts/html/pancreas.html (https://seer.cancer.govhttps://seer.cancer.gov/statfacts/html/pancreas.html). Accessed 2 Feb 2021.
Rhim AD, Mirek ET, Aiello NM, et al. EMT and dissemination precede pancreatic tumor formation. Cell. 2012;148(1–2):349–61.
Mizrahi JD, Surana R, Valle JW, Shroff RT. Pancreatic cancer. Lancet. 2020;395(10242):2008–20.
Sohal DP, Walsh RM, Ramanathan RK, Khorana AA. Pancreatic adenocarcinoma: treating a systemic disease with systemic therapy. J Natl Cancer Inst. 2014;106(3):dju011.
AJCC Cancer Staging Manual, Eighth Edition. 2018.
Cong L, Liu Q, Zhang R, et al. Tumor size classification of the 8(th) edition of TNM staging system is superior to that of the 7(th) edition in predicting the survival outcome of pancreatic cancer patients after radical resection and adjuvant chemotherapy. Sci Rep. 2018;8(1):10383.
Sohal DPS, Duong M, Ahmad SA, et al. Efficacy of perioperative chemotherapy for resectable pancreatic adenocarcinoma: a phase 2 randomized clinical trial. JAMA Oncol. 2021;7(3):421–7.
Pancreatic Adenocarcinoma: Version 2.2021.
Neoptolemos JP, Dunn JA, Stocken DD, et al. Adjuvant chemoradiotherapy and chemotherapy in resectable pancreatic cancer: a randomised controlled trial. Lancet. 2001;358(9293):1576–85.
Ueno H, Kosuge T, Matsuyama Y, et al. A randomised phase III trial comparing gemcitabine with surgery-only in patients with resected pancreatic cancer: Japanese Study Group of Adjuvant Therapy for Pancreatic Cancer. Br J Cancer. 2009;101(6):908–15.
Oettle H, Neuhaus P, Hochhaus A, et al. Adjuvant chemotherapy with gemcitabine and long-term outcomes among patients with resected pancreatic cancer: the CONKO-001 randomized trial. JAMA. 2013;310(14):1473–81.
Neoptolemos JP, Stocken DD, Bassi C, et al. Adjuvant chemotherapy with fluorouracil plus folinic acid vs gemcitabine following pancreatic cancer resection: a randomized controlled trial. JAMA. 2010;304(10):1073–81.
Neoptolemos JP, Palmer DH, Ghaneh P, et al. Comparison of adjuvant gemcitabine and capecitabine with gemcitabine monotherapy in patients with resected pancreatic cancer (ESPAC-4): a multicentre, open-label, randomised, phase 3 trial. Lancet. 2017;389(10073):1011–24.
Abrams RA, Winter KA, Safran H, et al. Results of the NRG Oncology/RTOG 0848 adjuvant chemotherapy question-erlotinib+gemcitabine for resected cancer of the pancreatic head: a phase II randomized clinical trial. Am J Clin Oncol. 2020;43(3):173–9.
Conroy T, Hammel P, Hebbar M, et al. FOLFIRINOX or gemcitabine as adjuvant therapy for pancreatic cancer. N Engl J Med. 2018;379(25):2395–406.
Shaib WL, Narayan AS, Switchenko JM, et al. Role of adjuvant therapy in resected stage IA subcentimeter (T1a/T1b) pancreatic cancer. Cancer. 2019;125(1):57–67.
Mungo B, Croce C, Oba A, et al. Controversial role of adjuvant therapy in node-negative invasive intraductal papillary mucinous neoplasm. Ann Surg Oncol. 2021;28(3):1533–42.
National Cancer Database. American College of Surgeons. https://www-facs-org.proxy.libraries.uc.edu/quality-programs/cancer/ncdb. 2020.
Winer LK, Cortez AR, Ahmad SA, et al. Evaluating the impact of ESPAC-1 on shifting the paradigm of pancreatic cancer treatment. J Surg Res. 2021;259:442–50.
Blackford AL, Canto MI, Klein AP, Hruban RH, Goggins M. Recent trends in the incidence and survival of stage 1A pancreatic cancer: a surveillance, epidemiology, and end results analysis. J Natl Cancer Inst. 2020;112(11):1162–9.
Mellby LD, Nyberg AP, Johansen JS, et al. Serum biomarker signature-based liquid biopsy for diagnosis of early-stage pancreatic cancer. J Clin Oncol. 2018;36(28):2887–94.
Guler GD, Ning Y, Ku CJ, et al. Detection of early stage pancreatic cancer using 5-hydroxymethylcytosine signatures in circulating cell free DNA. Nat Commun. 2020;11(1):5270.
Tzeng CW, Tran Cao HS, Lee JE, et al. Treatment sequencing for resectable pancreatic cancer: influence of early metastases and surgical complications on multimodality therapy completion and survival. J Gastrointest Surg. 2014;18(1):16–24 (discussion 24-15).
Olecki EJ, Stahl K, Peng JS, Dixon M, Gusani NJ, Shen C. Undertreatment of pancreatic cancer: role of surgical pathology. Ann Surg Oncol. 2021;28(3):1581–92.
Conroy T, Desseigne F, Ychou M, et al. FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N Engl J Med. 2011;364(19):1817–25.
Von Hoff DD, Ervin T, Arena FP, et al. Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N Engl J Med. 2013;369(18):1691–703.
de Rooij T, Tol JA, van Eijck CH, et al. Outcomes of distal pancreatectomy for pancreatic ductal adenocarcinoma in the Netherlands: a nationwide retrospective analysis. Ann Surg Oncol. 2016;23(2):585–91.
Nassour I, Adam MA, Kowalsky S, et al. Neoadjuvant therapy versus upfront surgery for early-stage left-sided pancreatic adenocarcinoma: a propensity-matched analysis from a national cohort of distal pancreatectomies. J Surg Oncol. 2021;123(1):245–51.
Winer LK, Dhar VK, Wima K, et al. The impact of tumor location on resection and survival for pancreatic ductal adenocarcinoma. J Surg Res. 2019;239:60–6.
Samowitz WS, Curtin K, Schaffer D, Robertson M, Leppert M, Slattery ML. Relationship of Ki-ras mutations in colon cancers to tumor location, stage, and survival: a population-based study. Cancer Epidemiol Biomark Prev. 2000;9(11):1193–7.
Slattery ML, Wolff E, Hoffman MD, Pellatt DF, Milash B, Wolff RK. MicroRNAs and colon and rectal cancer: differential expression by tumor location and subtype. Genes Chromosomes Cancer. 2011;50(3):196–206.
Lee B, Lipton L, Cohen J, et al. Circulating tumor DNA as a potential marker of adjuvant chemotherapy benefit following surgery for localized pancreatic cancer. Ann Oncol. 2019;30(9):1472–8.
Seppala TT, Zimmerman JW, Sereni E, et al. Patient-derived organoid pharmacotyping is a clinically tractable strategy for precision medicine in pancreatic cancer. Ann Surg. 2020;272(3):427–35.
Valle JW, Palmer D, Jackson R, et al. Optimal duration and timing of adjuvant chemotherapy after definitive surgery for ductal adenocarcinoma of the pancreas: ongoing lessons from the ESPAC-3 study. J Clin Oncol. 2014;32(6):504–12.
Conroy T, Hammel P, Turpin A, Belletier C, Wei A, Mitry E, Lopez A, Francois E, Artru P, Biagi J, Lecomte T, Assenat E, Faroux R, Ychou M, Bouche O, Lambert A, Monard L, Rat P, Castan F, Bachet J-B. LBA57 Unicancer PRODIGE 24/CCTG PA6 trial: updated results of a multicenter international randomized phase III trial of adjuvant mFOLFIRINOX (mFFX) versus gemcitabine (gem) in patients (pts) with resected pancreatic ductal adenocarcinomas (PDAC). Ann Oncol. 2021;32:1334.
Acknowledgment
The authors have no acknowledgements.
Funding
The authors have no grants or financial support to report.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
The authors have no disclosures to report.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
10434_2022_11580_MOESM1_ESM.eps
Supplementary Figure 1 Utilization of Chemotherapy in Stage IA Pancreatic Cancer. Use of adjuvantchemotherapy among all patients undergoing surgical resection and classification of type of chemotherapeutic regimen (single vs. multi-agent) among patients who received adjuvant chemotherapy. Adjuvant Chemotherapy (AC)
Rights and permissions
About this article
Cite this article
Turner, K.M., Delman, A.M., Ammann, A.M. et al. Is There a Benefit to Adjuvant Chemotherapy in Resected, Early Stage Pancreatic Ductal Adenocarcinoma?. Ann Surg Oncol 29, 4610–4619 (2022). https://doi.org/10.1245/s10434-022-11580-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-022-11580-7