Abstract
Background
Accumulation of basophils has been reported in several malignancies. In gastric cancer, the relation between tumor-infiltrating basophils and patient overall survival and chemotherapeutic responsiveness still remains obscure.
Objective
We aimed to investigate the postoperative prognostic and predictive significance of basophils to survival outcomes and chemotherapeutic responsiveness in resectable gastric cancer.
Methods
The study enrolled two independent patient data sets with 448 gastric cancer patients overall. Basophils were evaluated with the use of immunohistochemistry (IHC) staining, and the correlation with clinicopathological characteristics, survival outcomes, and responsiveness to fluorouracil-based adjuvant chemotherapy (ACT) were investigated. Additionally, IHC was applied to characterize immune contexture in gastric cancer.
Results
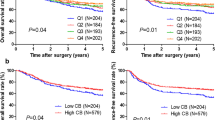
In either the discovery or validation data sets, accumulated basophils indicated poorer prognosis, and tumor-infiltrating basophils were identified as an independent adverse prognostic factor by multivariate analysis. Furthermore, tumor-infiltrating basophils determined significantly inferior therapeutic responsiveness to fluorouracil-based ACT in patients with stage III tumors. In addition, the abundance of basophils was correlated with an immunoevasive contexture characterized by M2-polarized macrophage infiltration. Moreover, our findings indicated elevated interleukin-4 expression but decreased interferon-γ expression in the high-basophils subgroup.
Conclusions
Tumor-infiltrating basophils in gastric cancer were identified as an independent adverse prognosticator, and also predicted inferior chemotherapeutic responsiveness, which identified those patients in need of much more individualized postoperative adjuvant therapy and more stringent follow-up. Furthermore, the infiltration of basophils was associated with immunoevasive tumor microenvironment, which might be a potential immunotherapeutic target for gastric cancer.





Similar content being viewed by others
References
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65(2):87–108. https://doi.org/10.3322/caac.21262.
Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. Feb 4 2021. https://doi.org/10.3322/caac.21660
Chen W, Zheng R, Baade PD, et al. Cancer statistics in China, 2015. CA Cancer J Clin. 2016;66(2):115–32. https://doi.org/10.3322/caac.21338.
Fuchs CS, Mayer RJ. Gastric carcinoma. New England J Med. 1995;333(1):32–41. https://doi.org/10.1056/nejm199507063330107.
De Vita F, Orditura M, Matano E, et al. A phase II study of biweekly oxaliplatin plus infusional 5-fluorouracil and folinic acid (FOLFOX-4) as first-line treatment of advanced gastric cancer patients. Br J Cancer. 2005;92(9):1644–9.
Longley DB, Harkin DP, Johnston PG. 5-Fluorouracil: mechanisms of action and clinical strategies: review article. Nature Reviews Cancer. 2003;3:330. https://doi.org/10.1038/nrc1074.
Karasuyama H, Mukai K, Obata K, Tsujimura Y, Wada T. Nonredundant roles of basophils in immunity. Ann Rev Immunol. 2011;29(1):45–69. https://doi.org/10.1146/annurev-immunol-031210-101257.
Khodoun MV, Orekhova T, Potter C, Morris S, Finkelman FD. Basophils initiate IL-4 production during a memory T-dependent response. J Exp Med. 2004;200(7):857.
Denzel A, Maus UA, Gomez MR, et al. Basophils enhance immunological memory responses. Nat Immunol. 2008;9(7):733–42. https://doi.org/10.1038/ni.1621.
Mukai K, Matsuoka K, Taya C, et al. Basophils play a critical role in the development of IgE-mediated chronic allergic inflammation independently of T cells and mast cells. Immunity. 2005;23(2):191–202. https://doi.org/10.1016/j.immuni.2005.06.011.
Sokol CL, Barton GM, Farr AG, Medzhitov R. A mechanism for the initiation of allergen-induced T helper type 2 responses. Nat Immunol. 2008;9(3):310–8. https://doi.org/10.1038/ni1558.
Obata K, Mukai K, Tsujimura Y, et al. Basophils are essential initiators of a novel type of chronic allergic inflammation. Blood. 2007;110(3):913–20. https://doi.org/10.1182/blood-2007-01-068718.
Tsujimura Y, Obata K, Mukai K, et al. Basophils play a pivotal role in immunoglobulin-G-mediated but not immunoglobulin-E-mediated systemic anaphylaxis. Immunity. 2008;28(4):581–9. https://doi.org/10.1016/j.immuni.2008.02.008.
Ohnmacht C, Schwartz C, Panzer M, Schiedewitz I, Naumann R, Voehringer D. Basophils orchestrate chronic allergic dermatitis and protective immunity against helminths. Immunity. 2010;33(3):364–74. https://doi.org/10.1016/j.immuni.2010.08.011.
Perrigoue JG, Saenz SA, Siracusa MC, et al. MHC class II-dependent basophil-CD4+ T cell interactions promote TH2 cytokine-dependent immunity. Nat Immunol. 2009;10(7):697–705.
Yoshimoto T, Yasuda K, Tanaka H, et al. Basophils contribute to TH2-IgE responses in vivo via IL-4 production and presentation of peptide-MHC class II complexes to CD4+ T cells. Nat Immunol. 2009;10(7):706–12. https://doi.org/10.1038/ni.1737.
Sokol CL, Chu N-Q, Yu S, Nish SA, Laufer TM, Medzhitov R. Basophils function as antigen-presenting cells for an allergen-induced T helper type 2 response. Nat Immunol. 2009;10(7):713–20. https://doi.org/10.1038/ni.1738.
Matsushima T, Handa H, Yokohama A, et al. Prevalence and clinical characteristics of myelodysplastic syndrome with bone marrow eosinophilia or basophilia. Blood. 2003;101(9):3386. https://doi.org/10.1182/blood-2002-03-0947.
Wimazal F, Germing U, Kundi M, et al. Evaluation of the prognostic significance of eosinophilia and basophilia in a larger cohort of patients with myelodysplastic syndromes. Cancer. 2010;116(10):2372–81. https://doi.org/10.1002/cncr.25036.
Anthony HM. Blood basophils in lung cancer. Br J Cancer. 1982;45(2):209–16.
Sektioglu IM, Carretero R, Bulbuc N, et al. Basophils promote tumor rejection via chemotaxis and infiltration of CD8+ T cells. Cancer Res. 2017;77(2):291–302. https://doi.org/10.1158/0008-5472.CAN-16-0993.
Cao Y, Liu H, Li H, et al. Association of O6-methylguanine-dna methyltransferase protein expression with postoperative prognosis and adjuvant chemotherapeutic benefits among patients with stage II or III gastric cancer. JAMA Surg. 2017;152(11):e173120. https://doi.org/10.1001/jamasurg.2017.3120.
Zhang H, Liu H, Shen Z, et al. Tumor-infiltrating neutrophils is prognostic and predictive for postoperative adjuvant chemotherapy benefit in patients with gastric cancer. Ann Surg. 2018;267(2):311–8. https://doi.org/10.1097/sla.0000000000002058.
Wang JT, Li H, Zhang H, et al. Intratumoral IL17-producing cells infiltration correlate with antitumor immune contexture and improved response to adjuvant chemotherapy in gastric cancer. Ann Oncol. 2019;30(2):266–73. https://doi.org/10.1093/annonc/mdy505.
Zhang H, Li R, Cao Y, et al. Poor clinical outcomes and immunoevasive contexture in intratumoral IL-10-producing macrophages enriched gastric cancer patients. Ann Surg. 2020. https://doi.org/10.1097/SLA.0000000000004037.
Liu X, Cao Y, Li R, et al. Poor clinical outcomes of intratumoral dendritic cell-specific intercellular adhesion molecule 3-grabbing non-integrin-positive macrophages associated with immune evasion in gastric cancer. Eur J Cancer. 2020;128:27–37. https://doi.org/10.1016/j.ejca.2020.01.002.
Shou Z-X, Jin X, Zhao Z-S. Upregulated expression of ADAM17 is a prognostic marker for patients with gastric cancer. Ann Surg. 2012;256(6):1014–22.
Clifford JL, Menter DG, Yang X, et al. Expression of protein mediators of type I interferon signaling in human squamous cell carcinoma of the skin. Cancer Epidemiol Biomarkers Prevent. 2000;9(9):993–7.
Ishii G, Ochiai A, Neri S. Phenotypic and functional heterogeneity of cancer-associated fibroblast within the tumor microenvironment. Adv Drug Deliv Rev. 2016;99(Pt B):186–96. https://doi.org/10.1016/j.addr.2015.07.007.
Seder RA, Paul WE, Dvorak AM, et al. Mouse splenic and bone marrow cell populations that express high-affinity Fc epsilon receptors and produce interleukin 4 are highly enriched in basophils. Proc Natl Acad Sci United States of America. 1991;88(7):2835–9.
Piccinni MP, Macchia D, Parronchi P, et al. Human bone marrow non-B, non-T cells produce interleukin 4 in response to cross-linkage of Fc epsilon and Fc gamma receptors. Proc Natl Acad Sci United States of America. 1991;88(19):8656–60.
Schroeder JT, MacGlashan DW, Lichtenstein LM. Human basophils: mediator release and cytokine production. Adv Immunol. 2001;77:93–122.
Ohnmacht C, Voehringer D. Basophils protect against reinfection with hookworms independently of mast cells and memory Th2 cells. J Immunol. 2009;184(1):344. https://doi.org/10.4049/jimmunol.0901841.
Paul WE, Zhu J. How are TH2-type immune responses initiated and amplified? Nat Rev Immunol. 2010;10(4):225–35. https://doi.org/10.1038/nri2735.
Ho IC, Tai T-S, Pai S-Y. GATA3 and the T-cell lineage: essential functions before and after T-helper-2-cell differentiation. Nat Rev Immunol. 2009;9(2):125–35. https://doi.org/10.1038/nri2476.
De Monte L, Wörmann S, Brunetto E, et al. Basophil recruitment into tumor-draining lymph nodes correlates with Th2 inflammation and reduced survival in pancreatic cancer patients. Cancer Res. 2016;76(7):1792. https://doi.org/10.1158/0008-5472.CAN-15-1801-T.
Parker WB, Cheng YC. Metabolism and mechanism of action of 5-fluorouracil. Pharmacol Ther. 1990;48(3):381–95. https://doi.org/10.1016/0163-7258(90)90056-8.
Major PP, Egan E, Herrick D, Kufe DW. 5-Fluorouracil incorporation in DNA of human breast carcinoma cells. Cancer Res. 1982;42(8):3005.
Longley DB, Harkin DP, Johnston PG. 5-Fluorouracil: mechanisms of action and clinical strategies. Nat Rev Cancer. 2003;3(5):330–8. https://doi.org/10.1038/nrc1074.
Todaro M, Alea MP, Di Stefano AB, et al. Colon cancer stem cells dictate tumor growth and resist cell death by production of interleukin-4. Cell Stem Cell. 2007;1(4):389–402. https://doi.org/10.1016/j.stem.2007.08.001.
Nevala WK, Vachon CM, Leontovich AA, Scott CG, Thompson MA, Markovic SN. Evidence of systemic Th2-driven chronic inflammation in patients with metastatic melanoma. Clin Cancer Res. 2009;15(6):1931. https://doi.org/10.1158/1078-0432.CCR-08-1980.
Guenova E, Watanabe R, Teague JE, et al. TH2 cytokines from malignant cells suppress TH1 responses and enforce a global TH2 bias in leukemic cutaneous T-cell lymphoma. Clin Cancer Res. 2013;19(14):3755–63. https://doi.org/10.1158/1078-0432.CCR-12-3488.
Wenzel L, Osann K, Hsieh S, Tucker JA, Monk BJ, Nelson EL. Psychosocial telephone counseling for survivors of cervical cancer: results of a randomized biobehavioral trial. J Clin Oncol. 2015;33(10):1171–9. https://doi.org/10.1200/JCO.2014.57.4079.
Todaro M, Lombardo Y, Francipane MG, et al. Apoptosis resistance in epithelial tumors is mediated by tumor-cell-derived interleukin-4. Cell Death Differ. 2008;15(4):762–72.
Nagai H, Miyaki D, Matsui T, et al. Th1/Th2 balance: an important indicator of efficacy for intra-arterial chemotherapy. Cancer Chemother Pharmacol. 2008;62(6):959–63. https://doi.org/10.1007/s00280-008-0685-y.
Zhang H, Wang X, Shen Z, Xu J, Qin J, Sun Y. Infiltration of diametrically polarized macrophages predicts overall survival of patients with gastric cancer after surgical resection. Gastric Cancer. 2015;18(4):740–50. https://doi.org/10.1007/s10120-014-0422-7.
Acknowledgments
The authors thank Dr. Lingli Chen (Department of Pathology, Zhongshan Hospital, Fudan University, Shanghai, China) and Dr. Peipei Zhang (Department of Pathology, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China) for their excellent help with the pathological technology.
Funding
This study was funded by grants from the National Natural Science Foundation of China (31770851, 81871930, 81902402, 81902901, 81972219) and Shanghai Sailing Program (17YF1402200, 18YF1404600, 19YF1407500).
Author information
Authors and Affiliations
Contributions
XH, YC, YG and KJ undertook acquisition of data, analysis and interpretation of data, statistical analysis, and drafting of the manuscript. JW, XL, KL, HF, KJ, YF, CL, HL, HZ, HL, JX provided technical and material support. RL and HH undertook study concept and design, analysis and interpretation of data, and drafting of the manuscript, obtained funding, and provided study supervision. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Disclosures
Xudong He, Yifan Cao, Yun Gu, Hanji Fang, Jieti Wang, Xin Liu, Kunpeng Lv, Kuan Yu, Yuchao Fei, Chao Lin, Hao Liu, Heng Zhang, He Li, Jiejie Xu, Ruochen Li, and Hongyong He declare they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
He, X., Cao, Y., Gu, Y. et al. Clinical Outcomes and Immune Metrics in Intratumoral Basophil-Enriched Gastric Cancer Patients. Ann Surg Oncol 28, 6439–6450 (2021). https://doi.org/10.1245/s10434-021-09815-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-021-09815-0