Abstract
Background
The appropriate management of breast cancer risk in BRCA mutation carriers following ovarian cancer diagnosis remains unclear. We sought to determine the survival benefit and cost effectiveness of risk-reducing mastectomy (RRM) among women with BRCA1/2 mutations following stage II–IV ovarian cancer.
Design
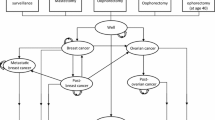
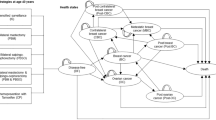
We constructed a decision model from a third-party payer perspective to compare annual screening with magnetic resonance imaging (MRI) and mammography to annual screening followed by RRM with reconstruction following ovarian cancer diagnosis. Survival, overall costs, and cost effectiveness were determined by decade at diagnosis using 2015 US dollars. All inputs were obtained from the literature and public databases. Monte Carlo probabilistic sensitivity analysis was performed with a $100,000 willingness-to-pay threshold.
Results
The incremental cost-effectiveness ratio (ICER) per year of life saved (YLS) for RRM increased with age and BRCA2 mutation status, with greater survival benefit demonstrated in younger patients with BRCA1 mutations. RRM delayed 5 years in 40-year-old BRCA1 mutation carriers was associated with 5 months of life gained (ICER $72,739/YLS), and in 60-year-old BRCA2 mutation carriers was associated with 0.8 months of life gained (ICER $334,906/YLS). In all scenarios, $/YLS and mastectomies per breast cancer prevented were lowest with RRM performed 5–10 years after ovarian cancer diagnosis.
Conclusion
For most BRCA1/2 mutation carriers following ovarian cancer diagnosis, RRM performed within 5 years is not cost effective when compared with breast cancer screening. Imaging surveillance should be advocated during the first several years after ovarian cancer diagnosis, after which point the benefits of RRM can be considered based on patient age and BRCA mutation status.


Similar content being viewed by others
References
Antoniou AC, Pharoah PDP, McMullan G, et al. A comprehensive model for familial breast cancer incorporating BRCA1, BRCA2 and other genes. Br J Cancer. 2002;86:76-83.
Chen S, Iversen ES, Friebel T, et al. Characterization of BRCA1 and BRCA2 mutations in a large United States sample. J Clin Oncol. 2006;24(6):863-871.
van der Kolk DM, de Bock GH, Leege BK, et al. Penetrance of breast cancer, ovarian cancer and contralateral breast cancer in BRCA1 and BRCA2 families: high cancer incidence at older age. Breast Cancer Res Treat. 2010;124:643-651.
Easton DF, Ford D, Bishop DT. Breast and ovarian cancer incidence in BRCA1-mutation carriers. Breast Cancer Linkage Consortium. Am J Hum Genet. 1995;56(1):265-271.
Rebbeck TR, Lynch HT, Neuhausen SL, et al. Prophylactic oophorectomy in carriers of BRCA1 or BRCA2 mutations. N Engl J Med. 2002;346(21):1616-1622.
Kauff ND, Satagopan JM, Robson ME, et al. Risk-reducing salpingo-oophorectomy in women iwht a BRCA1 or BRCA2 mutation. N Engl J Med. 2002;346(21):1609-1615.
Meijers-Heijboer H, van Geel B, van Putten WLJ, et al. Breast cancer after prophylactic bilateral mastectomy in women with a BRCA1 or BRCA2 mutation. N Engl J Med. 2001;345(3):159-164.
Hartmann LC, Sellers TA, Shchaid DJ, et al. Efficacy of bilateral prophylactic mastectomy in BRCA1 and BRCA2 gene mutation carriers. J Natl Cancer Inst. 2001;93(21):1633-1637.
National Comprehensive Cancer Network (NCCN). NCCN Clinical practice guidelines in oncology. 2017. http://www.nccn.org/professionals/physician_gls/f_guidelines.asp. Accessed 27 Feb 2017.
Ludwig KK, Neuner J, Butler A, Geurts JL, Kong AL. Risk reduction and survival benefit of prophylactic surgery in BRCA mutation carriers, a systematic review. Am J Surg. 2016;212(4):660-669.
George A, Kaye S, Banerjee S. Delivering widespread BRCA testing and PARP inhibition to patients with ovarian cancer. Nat Rev Clin Oncol. 2017;14(5):284–296.
Grann VR, Patel PR, Jacobson JS, et al. Comparative effectiveness of screening and prevention strategies among BRCA1/2-affected mutation carriers. Breast Cancer Res Treat. 2011;125:837-847.
Society of Gynecologic Oncology. SGO Clinical Practice Statement: Genetic Testing for Ovarian Cancer. Society of Gynecologic Oncology; 2014.
Howlander N, Noone AM, Krapcho M, et al. SEER Cancer Statistics Review, 1975–2012. Based on November 2014 SEER data submission, posted to the SEER web site, April 2015. http://seer.cancer.gov/csr/1975_2012/. Accessed 5 Jan 2016.
Chetrit A, Hirsh-Yechezkel G, Ben-David Y, Lubin F, Friedman E, Sadetzki S. Effect of BRCA1/2 mutations on long-term survival of patients with invasive ovarian cancer: the national Israeli study of ovarian cancer. J Clin Oncol. 2008;26(1):20-25.
Alsop K, Fereday S, Meldrum C, et al. BRCA mutation frequency and patterns of treatment response in BRCA mutation-positive women with ovarian cancer: a report from the Australian Ovarian Cancer Study Group. J Clin Oncol. 2012;30(21):2654-2663.
Ledermann JA, Harter P, Gourley C, et al. Overall survival in patients with platinum-sensitive recurrent serous ovarian cancer receiving olaparib maintenance monotherapy: an updated analysis from a randomised, placebo-controlled, double-blind, phase 2 trial. Lancet Oncol. 2016;17(11):1579-1589.
Mirza MR, Monk BJ, Herrstedt J, et al. Niraparib maintenance therapy in platinum-sensitive, recurrent ovarian cancer. N Engl J Med. 2016;375(22):2154-2164.
Sander GD, Neumann PJ, Basu A, et al. Second panel on cost-effectiveness in health and medicine. JAMA. 2016;316(10):1093-1103.
Neumann PJ, Cohen JT, Weinstein MC. Updating cost-effectiveness: the curious resilience of the $50,000-per-QALY threshold. N Engl J Med. 2014;371(9):796-797.
Havrilesky LJ, Alvarez Secord A, Darcy KM, Armstrong DK, Kulasingam S. Cost effectiveness of intraperitoneal compared with intravenous chemotherapy for women with optimally resected stage III ovarian cancer: a Gynecologic Oncology Group Study. J Clin Oncol. 2008;26(25):4144-4150.
Heemskerk-Gerritsen BA, Menke-Pluijmers MB, Jager A, et al. Substantial breast cancer risk reduction and potential survival benefit after bilateral mastectomy when compared with surveillance in healthy BRCA1 and BRCA2 mutation carriers: a prospective analysis. Ann Oncol. 2013;24(8):2029-2035.
Havrilesky LJ, Chino JP, Myers ER. How much is another randomized trial of lymph node dissection in endometrial cancer worth? A value of information analysis. Gynecol Oncol. 2013;131(1):140-146.
Yang D, Khan S, Sun Y, et al. Association of BRCA1 and BRCA2 mutations with survival, chemotherapy sensitivity and gene mutator phenotype in patients with ovarian cancer. JAMA. 2011;306(14):1557-1565.
van der Burg ME, Onstenk W, Boere IA, et al. Long-term results of a randomised phase III trial of weekly versus three-weekly paclitaxel/platinum induction therapy followed by standard or extended three-weekly paclitaxel/platinum in European patients with advanced epithelial ovarian cancer. Eur J Cancer. 2014;50(15):2592-2601.
Zhong Q, Peng HL, Zhao X, Zhang L, Hwang WT. Effects of BRCA1- and BRCA2-related mutations on ovarian and breast cancer survival: a meta-analysis. Clin Cancer Res. 2015;21(1):211-220.
Aghajanian C, Goff B, Nycum LR, Wang YV, Husain A, Blank SV. Final overall survival and safety analysis of OCEANS, a phase 3 trial of chemotherapy with or without bevacizumab in patients with platinum-sensitive recurrent ovarian cancer. Gynecol Oncol. 2015;139(1):10-16.
Lakhani SR, Van De Vijver MJ, Jacquemier J, et al. The pathology of familial breast cancer: predictive value of immunohistochemical markers estrogen receptor, progesterone receptor, HER-2, and p53 in patients with mutations in BRCA1 and BRCA2. J Clin Oncol. 2002;20(9):2310-2318.
Spurdle AB, Couch FJ, Parsons MT, et al. Refined histopathological predictors of BRCA1 and BRCA2 mutation status: a large-scale analysis of breast cancer characteristics from the BCAC, CIMBA, and ENIGMA consortia. Breast Cancer Res. 2014;16(6):1.
Dent R, Trudeau M, Pritchard KI, et al. Triple-negative breast cancer: clinical features and patterns of recurrence. Clin Cancer Res. 2007;13(15):4429-4434.
Arias E. United States Life Tables, 2011. Natl Vital Stat Rep. 2015;64(11):1–63.
Halfhill TR. Tom’s Inflation Calculator. 2016. http://www.halfhill.com/inflation_js.html. Accessed 23 May 2015.
Plevritis SK, Kurian AW, Sigal BM, et al. Cost-effectiveness of screening BRCA1/2 mutation carrierw with breast magnetic resonance imaging. JAMA. 2006;295:2374-2384.
Zendejas B, Moriarty JP, O’Byrne J, Degnim AC, Farley DR, Boughey JC. Cost-effectiveness of contralateral prophylactic mastectomy versus routine surveillance in patients with unilateral breast cancer. J Clin Oncol. 2011;29(22):2993-3000.
Fitzpatrick AM, Gao LL, Smith BL, et al. Cost and outcome analysis of breast reconstruction paradigm shift. Ann Plast Surg. 2014;73(2):141-149.
Mattos D, Gfrerer L, Reish RG, et al. Lifetime costs of prophylactic mastectomies and reconstruction versus surveillance. Plast Reconstr Surg. 2015;136(6):730e-740e.
Yabroff KR, Lamont EB, Mariotto A, et al. Cost of care for elderly cancer patients in the United States. J Natl Cancer Inst. 2008;100(9):630-641.
Vencken PM, Kriege M, Hooning M, et al. The risk of primary and contralateral breast cancer after ovarian cancer in BRCA1/BRCA2 mutation carriers: implications for counseling. Cancer. 2013;119(5):955-962.
Gangi A, Cass I, Paik D, et al. Breast cancer following ovarian cancer in brca mutation carriers. JAMA Surg. 2014;149(12):1306-1313.
Domchek SM, Jhaveri K, Patil S, et al. Risk of metachronous breast cancer after BRCA mutation-associated ovarian cancer. Cancer. 2013;119(7):1344-1348.
Minion LE, Coleman RL, Alvarez RD, Herzog TJ. Endpoints in clinical trials: what do patients consider important? A survey of the Ovarian Cancer National Alliance. Gynecol Oncol. 2016;140(2):193-198.
Walsh T, Casadei S, Lee MK, et al. Mutations in 12 genes for inherited ovarian, fallopian tube, and peritoneal carcinoma identified by massively parallel sequencing. Proc Natl Acad Sci U S A. 2011;108(44):18032-18037.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gamble, C., Havrilesky, L.J., Myers, E.R. et al. Cost Effectiveness of Risk-Reducing Mastectomy versus Surveillance in BRCA Mutation Carriers with a History of Ovarian Cancer. Ann Surg Oncol 24, 3116–3123 (2017). https://doi.org/10.1245/s10434-017-5995-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-017-5995-z