Abstract
Background
At our institution, a planned sentinel node biopsy (SNB) procedure is occasionally canceled after preoperative lymphoscintigraphy. This study reports the frequency of this, the reasons, and the management and outcomes of these patients.
Methods
All patients with clinically localized cutaneous melanoma treated at Melanoma Institute Australia between 2000 and 2009 whose planned SNB procedure was not undertaken after lymphoscintigraphy were included in this retrospective study.
Results
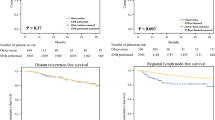
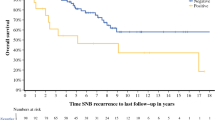
Of the 3148 patients in whom the procedure had been planned, 203 patients (6.4 %) did not have a SNB. The main reason for not proceeding with SNB (in 84 % of cases) was the lymphoscintigraphic demonstration of multiple drainage fields and/or multiple sentinel nodes (SNs). Patients who did not proceed to SNB were significantly older than those who did, more often had melanomas of the head or neck, and had more SNs and more nodal drainage fields. Of the 203 patients, 181 (89 %) were followed with high-resolution ultrasound of their SNs, which identified 33 % of the nodal recurrences before they were clinically apparent. Patients whose SNB was canceled had significantly worse recurrence-free survival and regional node disease-free survival, but melanoma-specific survival was similar. Compared to SN-positive patients, node-positive patients without SNB had significantly more involved nodes when a delayed lymphadenectomy was performed, but melanoma-specific survival was not significantly different after a median follow-up of 42 months.
Conclusions
Lymphoscintigraphy with ultrasound follow-up of previously identified SNs is an acceptable management strategy for patients in whom a SNB procedure is likely to be challenging.


Similar content being viewed by others
References
Wong SL, Balch CM, Hurley P, et al. Sentinel lymph node biopsy for melanoma: American Society of Clinical Oncology and Society of Surgical Oncology joint clinical practice guideline. Ann Surg Oncol. 2012;19:3313–24.
Morton DL, Thompson JF, Cochran AJ, et al. Final trial report of sentinel-node biopsy versus nodal observation in melanoma. N Engl J Med. 2014;370:599–609.
Nieweg OE, Jansen L, Kroon BB. Technique of lymphatic mapping and sentinel node biopsy for melanoma. Eur J Surg Oncol. 1998;24:520–4.
Thompson JF, Uren RF. Lymphatic mapping in management of patients with primary cutaneous melanoma. Lancet Oncol. 2005;6:877–85.
Uren RF, Howman-Giles R, Chung D, Thompson JF. Guidelines for lymphoscintigraphy and F18 FDG PET scans in melanoma. J Surg Oncol. 2011;104:405–19.
Verwer N, Scolyer RA, Uren RF, et al. Treatment and prognostic significance of positive interval sentinel nodes in patients with primary cutaneous melanoma. Ann Surg Oncol. 2011;18:3292–9.
Nieweg OE, Tanis PJ, Kroon BB. The definition of a sentinel node. Ann Surg Oncol. 2001;8:538–41.
Scolyer RA, Murali R, McCarthy SW, Thompson JF. Pathologic examination of sentinel lymph nodes from melanoma patients. Semin Diagn Pathol. 2008;25:100–11.
Nieweg OE. Current status of sentinel lymph node biopsy in patients with melanoma. Rozhl Chir. 2014;93:485–90.
Balch CM, Gershenwald JE, Soong SJ, et al. Final version of 2009 AJCC melanoma staging and classification. J Clin Oncol. 2009;27:6199–206.
Breslow A. Thickness, cross-sectional areas and depth of invasion in the prognosis of cutaneous melanoma. Ann Surg. 1970;172:902–8.
Thompson JF, Soong SJ, Balch CM, et al. Prognostic significance of mitotic rate in localized primary cutaneous melanoma: an analysis of patients in the multi-institutional American Joint Committee on Cancer melanoma staging database. J Clin Oncol. 2011;29:2199–205.
Balch CM, Soong SJ, Gershenwald JE, et al. Age as a prognostic factor in patients with localized melanoma and regional metastases. Ann Surg Oncol. 2013;20:3961–8.
Joosse A, Collette S, Suciu S, et al. Superior outcome of women with stage I/II cutaneous melanoma: pooled analysis of four european organisation for research and treatment of cancer phase III trials. J Clin Oncol. 2012;30:2240–7.
Scoggins CR, Ross MI, Reintgen DS, et al. Gender-related differences in outcome for melanoma patients. Ann Surg. 2006;243:693–8.
Balch CM, Soong SJ, Gershenwald JE, et al. Prognostic factors analysis of 17,600 melanoma patients: validation of the American Joint Committee on Cancer melanoma staging system. J Clin Oncol. 2001;19:3622–34.
Callender GG, Egger ME, Burton AL, et al. Prognostic implications of anatomic location of primary cutaneous melanoma of 1 mm or thicker. Am J Surg. 2011;202:659–64.
Steyerberg E. Selection of main effects. In: Clinical prediction models. New York: Springer Science + Business Media; 2009. p. 191–210.
Reynolds HM, Dunbar PR, Uren RF, Blackett SA, Thompson JF, Smith NP. Three-dimensional visualisation of lymphatic drainage patterns in patients with cutaneous melanoma. Lancet Oncol. 2007;8:806–12.
Thompson JF, Uren RF, Shaw HM, et al. Location of sentinel lymph nodes in patients with cutaneous melanoma: new insights into lymphatic anatomy. J Am Coll Surg. 1999;189:195–204.
Uren RF, Howman-Giles RB, Shaw HM, Thompson JF, McCarthy WH. Lymphoscintigraphy in high-risk melanoma of the trunk: predicting draining node groups, defining lymphatic channels and locating the sentinel node. J Nucl Med. 1993;34:1435–40.
Porter GA, Ross MI, Berman RS, Lee JE, Mansfield PF, Gershenwald JE. Significance of multiple nodal basin drainage in truncal melanoma patients undergoing sentinel lymph node biopsy. Ann Surg Oncol. 2000;7:256–61.
Ahmadzadehfar H, Hinz T, Wierzbicki A, et al. Significance of multiple nodal basin drainage in patients with truncal melanoma. Q J Nucl Med Mol Imaging. 2014;55:1687.
Federico AC, Chagpar AB, Ross MI, et al. Effect of multiple-nodal basin drainage on cutaneous melanoma. Arch Surg. 2008;143:632–7,
Faries MB, Thompson JF, Cochran A, et al. The impact on morbidity and length of stay of early versus delayed complete lymphadenectomy in melanoma: results of the Multicenter Selective Lymphadenectomy Trial (I). Ann Surg Oncol. 2010;17:3324–9.
Balch CM, Thompson JF, Gershenwald JE, et al. Age as a predictor of sentinel node metastasis among patients with localized melanoma: an inverse correlation of melanoma mortality and incidence of sentinel node metastasis among young and old patients. Ann Surg Oncol. 2014;21:1075–81.
White RL Jr, Ayers GD, Stell VH, et al. Factors predictive of the status of sentinel lymph nodes in melanoma patients from a large multicenter database. Ann Surg Oncol. 2011;18:3593–3600.
Voit CA, van Akkooi AC, Schäfer-Hesterberg G, et al. Rotterdam criteria for sentinel node (SN) tumor burden and the accuracy of ultrasound (US)-guided fine-needle aspiration cytology (FNAC): can US-guided FNAC replace SN staging in patients with melanoma? J Clin Oncol. 2009;27:4994–5000.
Voit CA, van Akkooi AC, Eggermont AM, et al. Fine needle aspiration cytology of palpable and nonpalpable lymph nodes to detect metastatic melanoma. J Natl Cancer Inst. 2011;103:1771–7.
Kruger U, Kretschmer L, Thoms KM, et al. Lymph node ultrasound during melanoma follow-up significantly improves metastasis detection compared with clinical examination alone: a study on 433 patients. Melanoma Res. 2011;21:457–63.
Prayer L, Winkelbauer H, Gritzmann N, Winkelbauer F, Helmer M, Pehamberger H. Sonography versus palpation in the detection of regional lymph-node metastases in patients with malignant melanoma. Eur J Cancer. 1990;26:827–30.
Binder M, Kittler H, Steiner A, Dorffner R, Wolff K, Pehamberger H. Lymph node sonography versus palpation for detecting recurrent disease in patients with malignant melanoma. Eur J Cancer. 1997;33:1805–8.
Rossi CR, Seno A, Vecchiato A, et al. The impact of ultrasound scanning in the staging and follow-up of patients with clinical stage I cutaneous melanoma. Eur J Cancer. 1997;33:200–3.
Blum A, Schlagenhauff B, Stroebel W, Breuninger H, Rassner G, Garbe C. Ultrasound examination of regional lymph nodes significantly improves early detection of locoregional metastases during the follow-up of patients with cutaneous melanoma: results of a prospective study of 1288 patients. Cancer. 2000;88:2534–9.
Voit C, Mayer T, Kron M, et al. Efficacy of ultrasound B-scan compared with physical examination in follow-up of melanoma patients. Cancer. 2001;91:2409–16.
Garbe C, Paul A, Kohler-Spath H, et al. Prospective evaluation of a follow-up schedule in cutaneous melanoma patients: recommendations for an effective follow-up strategy. J Clin Oncol. 2003;21:520–9.
Schmid-Wendtner MH, Paerschke G, Baumert J, Plewig G, Volkenandt M. Value of ultrasonography compared with physical examination for the detection of locoregional metastases in patients with cutaneous melanoma. Melanoma Res. 2003;13:183–8.
Hofmann U, Szedlak M, Rittgen W, Jung EG, Schadendorf D. Primary staging and follow-up in melanoma patients—monocenter evaluation of methods, costs and patient survival. Br J Cancer. 2002;87:151–7.
Leiter U, Marghoob AA, Lasithiotakis K, et al. Costs of the detection of metastases and follow-up examinations in cutaneous melanoma. Melanoma Res. 2009;19:50–7.
Bafounta ML, Beauchet A, Chagnon S, Saiag P. Ultrasonography or palpation for detection of melanoma nodal invasion:a meta-analysis. Lancet Oncol. 2004;5:673–80.
Balch CM, Gershenwald JE, Soong SJ, et al. Multivariate analysis of prognostic factors among 2,313 patients with stage III melanoma: comparison of nodal micrometastases versus macrometastases. J Clin Oncol. 2010;28:2452–9.
Mohrle M, Schippert W, Rassner G, Garbe C, Breuninger H. Is sentinel lymph node biopsy of therapeutic relevance for melanoma? Dermatology. 2004;209:5–13.
Gutzmer R, Al Ghazal M, Geerlings H, Kapp A. Sentinel node biopsy in melanoma delays recurrence but does not change melanoma-related survival: a retrospective analysis of 673 patients. Br J Dermatol. 2005;153:1137–41.
Starz H, Balda BR. Benefit of sentinel lymphadenectomy for patients with nonulcerated cutaneous melanomas in the breslow range between 0.76 and 1 mm: a follow-up study of 148 patients. Int J Cancer. 2007;121:689–93.
Koskivuo I, Talve L, Vihinen P, Maki M, Vahlberg T, Suominen E. Sentinel lymph node biopsy in cutaneous melanoma: a case–control study. Ann Surg Oncol. 2007;14:3566–74.
Leiter U, Buettner PG, Bohnenberger K, et al. Sentinel lymph node dissection in primary melanoma reduces subsequent regional lymph node metastasis as well as distant metastasis after nodal involvement. Ann Surg Oncol. 2010;17:129–37.
Satzger I, Meier A, Hoy L, et al. Sentinel node dissection delays recurrence and prolongs melanoma-related survival: an analysis of 673 patients from a single center with long-term follow-up. Ann Surg Oncol. 2011;18:514–20.
van der Ploeg AP, Haydu LE, Spillane AJ, et al. Outcome following sentinel node biopsy plus wide local excision versus wide local excision only for primary cutaneous melanoma: analysis of 5840 patients treated at a single institution. Ann Surg. 2014;260:149–57.
Acknowledgment
N.A.I. received funding from K. F. Hein Fonds, Stichting Fundatie van de Vrijvrouwe van Renswoude, and Stichting dr. Hendrik Muller’s Vaderlandsch Fonds.
Disclosure
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
Rights and permissions
About this article
Cite this article
Ipenburg, N.A., Nieweg, O.E., Uren, R.F. et al. Outcome of Melanoma Patients Who Did Not Proceed to Sentinel Node Biopsy After Preoperative Lymphoscintigraphy. Ann Surg Oncol 24, 117–126 (2017). https://doi.org/10.1245/s10434-016-5458-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-016-5458-y